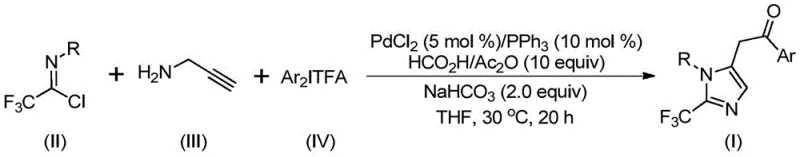
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial API Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups which are pivotal for enhancing metabolic stability and lipophilicity in drug candidates. Patent CN111423381A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing critical limitations in existing synthetic routes. This technology leverages a transition metal palladium-catalyzed carbonylation tandem reaction, utilizing cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts. The significance of this innovation lies in its ability to operate under exceptionally mild conditions, specifically at 30°C, while maintaining high reaction efficiency and broad substrate compatibility. For R&D directors and process chemists, this represents a paradigm shift away from hazardous diazo chemistry toward safer, more controllable catalytic cycles that facilitate the rapid generation of diverse molecular libraries for drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on the use of highly reactive and potentially unstable synthons. Literature reports frequently cite the use of trifluorodiazoethane as a primary building block for introducing the CF3 moiety into heterocyclic frameworks. While effective in certain contexts, trifluorodiazoethane poses significant safety hazards due to its explosive nature and thermal instability, requiring specialized equipment and rigorous safety protocols that drive up operational costs and complicate scale-up efforts. Furthermore, alternative methods using trifluoroethylimide acid halides have not been widely explored or optimized, leaving a gap in practical, safe, and efficient synthetic strategies. These conventional approaches often suffer from narrow substrate scope, harsh reaction conditions, and poor atom economy, making them less attractive for the commercial manufacturing of complex pharmaceutical intermediates where purity and safety are paramount concerns for regulatory compliance.
The Novel Approach
The novel methodology disclosed in the patent overcomes these hurdles by employing a sophisticated palladium-catalyzed carbonylation cascade that merges three distinct components into a single imidazole ring system. By utilizing trifluoroethylimidoyl chloride and propargylamine alongside diaryliodonium salts, the process achieves a highly efficient construction of the 2-trifluoromethyl imidazole core without the need for dangerous diazo reagents. This approach not only enhances operational safety but also simplifies the reaction setup, as it proceeds smoothly in common aprotic solvents like tetrahydrofuran at a mild temperature of 30°C. The versatility of this method is underscored by its tolerance for a wide array of functional groups, allowing for the synthesis of diversified derivatives simply by modifying the aryl groups on the starting materials. This flexibility is crucial for medicinal chemists aiming to optimize structure-activity relationships (SAR) in lead compounds.
Mechanistically, this transformation is a marvel of modern organometallic chemistry, involving a sequence of well-coordinated steps that ensure high fidelity in product formation. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, generating a trifluoroacetamidine intermediate. This species then undergoes isomerization followed by palladium-catalyzed aminopalladation of the alkyne moiety to form a key alkenyl palladium intermediate. Subsequent isomerization yields an alkyl palladium species, which then engages in a carbonylation step driven by carbon monoxide released in situ from the formic acid and acetic anhydride mixture. The resulting acyl palladium intermediate undergoes oxidative addition with the diaryliodonium salt to form a transient tetravalent palladium complex, finally concluding with a reductive elimination step that releases the desired 2-trifluoromethyl substituted imidazole and regenerates the active catalyst. This intricate cycle highlights the precision required in catalyst design and reaction condition optimization to achieve such high yields.
Understanding the impurity profile is critical for any pharmaceutical intermediate, and this catalytic system offers inherent advantages in controlling side reactions. The use of specific ligands like triphenylphosphine in conjunction with palladium chloride helps stabilize the active catalytic species, minimizing the formation of palladium black and other inactive aggregates that could lead to incomplete conversions or difficult-to-remove metal residues. Furthermore, the mild reaction temperature of 30°C suppresses thermal decomposition pathways that often plague high-temperature heterocycle syntheses. The choice of sodium bicarbonate as a base provides a buffered environment that facilitates the initial nucleophilic attack without promoting excessive hydrolysis of the sensitive imidoyl chloride or the diaryliodonium salt. Post-reaction processing is straightforward, involving simple filtration and silica gel treatment followed by column chromatography, ensuring that the final product meets stringent purity specifications required for downstream API synthesis.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
To implement this synthesis effectively, precise control over stoichiometry and reaction parameters is essential. The patent outlines a robust protocol where palladium chloride, triphenylphosphine, sodium bicarbonate, and a mixture of acetic anhydride and formic acid are combined with the three main substrates in an organic solvent. The molar ratios are carefully balanced, typically employing a slight excess of the trifluoroethylimidoyl chloride and diaryliodonium salt relative to propargylamine to drive the reaction to completion. The detailed standardized synthesis steps below provide a clear roadmap for replicating this high-efficiency transformation in a laboratory or pilot plant setting, ensuring consistent quality and yield across different batches.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid mixture, trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt in an organic solvent such as THF.
- Stir the reaction mixture at 30°C for 16 to 24 hours to ensure complete conversion of starting materials.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 2-trifluoromethyl substituted imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented process offers substantial strategic benefits that directly impact the bottom line and operational resilience. The reliance on commercially available and inexpensive starting materials such as aromatic amines (precursors to imidoyl chlorides), propargylamine, and aryl boronic acids (precursors to iodonium salts) ensures a stable and diversified supply base. Unlike specialized diazo reagents which may have limited suppliers and long lead times, the precursors for this method are commodity chemicals produced at scale by multiple global vendors. This reduces the risk of supply chain disruptions and provides procurement managers with greater leverage in negotiating favorable pricing terms. Additionally, the elimination of hazardous reagents simplifies logistics and storage requirements, further lowering the total cost of ownership for the manufacturing process.
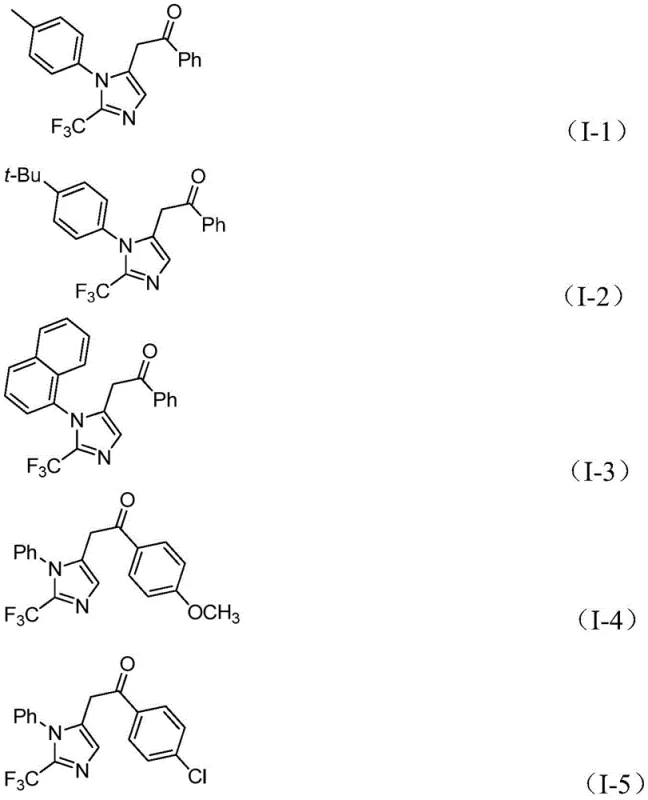
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the use of low-cost catalysts and reagents. Palladium chloride, while a precious metal, is used in catalytic amounts (5 mol%), and the ligand triphenylphosphine is inexpensive and widely available. The avoidance of expensive and dangerous trifluorodiazoethane removes a major cost driver associated with safety infrastructure and specialized handling equipment. Furthermore, the high reaction efficiency and yields reported (up to 97%) minimize raw material waste and reduce the burden on purification processes, leading to substantial cost savings in solvent usage and waste disposal. The mild reaction conditions also translate to lower energy consumption, as heating to high temperatures or cryogenic cooling is not required.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply chain. Operating at 30°C in THF allows for the use of standard glass-lined or stainless steel reactors without the need for exotic materials resistant to extreme corrosion or pressure. The broad functional group tolerance means that variations in raw material quality (within specification) are less likely to cause batch failures, ensuring consistent output. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream API manufacturers. The ability to source precursors from multiple geographic regions further mitigates the risk of regional supply shocks.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or tonnage levels is facilitated by its simplicity and safety profile. The absence of gas cylinders for carbon monoxide (since CO is generated in situ from formic acid/acetic anhydride) removes a significant safety barrier to scale-up. The in situ generation of CO also improves atom economy and reduces the release of toxic gases into the environment. Waste streams are primarily organic solvents and inorganic salts, which can be managed through standard wastewater treatment and solvent recovery systems. This alignment with green chemistry principles supports corporate sustainability goals and ensures compliance with increasingly stringent environmental regulations governing chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the practical implementation and advantages of the technology for potential partners and licensees. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production pipelines.
Q: What are the safety advantages of this new synthesis method compared to traditional routes?
A: Traditional methods often rely on hazardous trifluorodiazoethane. This patented process utilizes stable trifluoroethylimidoyl chlorides and diaryliodonium salts, significantly reducing explosion risks and handling hazards associated with diazo compounds.
Q: What yields can be expected for diverse substrates in this palladium-catalyzed reaction?
A: The patent demonstrates excellent substrate compatibility with yields ranging from 65% to 97% across various electron-donating and electron-withdrawing groups, including methyl, tert-butyl, methoxy, chloro, bromo, and nitro substituents.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method operates under mild conditions (30°C) using commercially available reagents and standard solvents like THF. The patent explicitly states the process can be expanded to the gram level, providing a viable pathway for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing palladium-catalyzed processes to meet stringent purity specifications, ensuring that every batch of 2-trifluoromethyl imidazole intermediate delivered meets the rigorous demands of the global pharmaceutical industry. With state-of-the-art rigorous QC labs and a commitment to quality assurance, we provide a secure and reliable source for these critical building blocks, enabling our clients to accelerate their drug development timelines with confidence.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your supply chain with high-quality, cost-effective chemical solutions.
