Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access fluorinated heterocycles, which are pivotal motifs in modern drug design. Patent CN111423381B introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing critical challenges in synthetic efficiency and substrate versatility. This technology leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts as key building blocks. The significance of this innovation lies in its ability to operate under remarkably mild conditions, specifically at 30°C, while maintaining high reaction efficiency and broad functional group tolerance. For R&D directors and process chemists, this represents a substantial leap forward in accessing complex nitrogen-containing five-membered heterocycles that are essential for enhancing the bioavailability and metabolic stability of active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with significant operational hazards and economic inefficiencies. Literature reports indicate that prevailing methods often rely on direct reactions between synthons containing trifluoromethyl groups and suitable substrates, frequently employing reagents such as trifluorodiazoethane. These conventional approaches are characterized by the use of highly unstable and potentially explosive diazo compounds, which necessitate stringent safety protocols and specialized equipment, thereby inflating production costs. Furthermore, many existing protocols suffer from limited substrate compatibility, often failing to tolerate sensitive functional groups or requiring harsh reaction conditions that degrade the molecular integrity of complex intermediates. The reliance on expensive or difficult-to-handle trifluoromethyl sources creates a bottleneck in the supply chain, limiting the scalability required for commercial API manufacturing and restricting the structural diversity accessible to medicinal chemists during lead optimization phases.
The Novel Approach
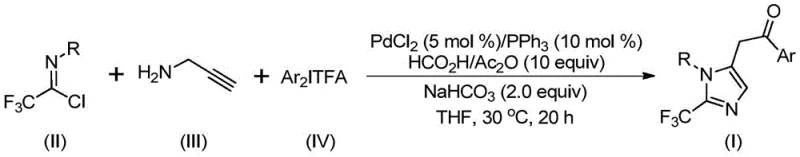
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a sophisticated yet operationally simple multicomponent coupling strategy. By employing cheap and easily obtained trifluoroethyliminato chloride, propargylamine, and diaryl iodide as starting materials, the process circumvents the need for hazardous diazo reagents entirely. The reaction proceeds via a palladium-catalyzed carbonylation mechanism where formic acid and acetic anhydride serve as a safe and effective carbon monoxide alternative. As illustrated in the general reaction scheme below, this methodology enables the efficient construction of the imidazole core with high regioselectivity. The mild reaction temperature of 30°C not only reduces energy consumption but also minimizes the formation of thermal degradation byproducts, ensuring a cleaner crude profile. This strategic shift in synthetic design significantly broadens the utility of the method, allowing for the synthesis of diversely substituted imidazole compounds through simple substrate modification without compromising yield or purity.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation offers profound insights into how high selectivity is achieved under such mild conditions. During the reaction, an intermolecular carbon-nitrogen bond promoted by alkali is initially formed to generate a trifluoroacetamidine compound, which subsequently undergoes isomerization. The palladium catalyst then facilitates the activation of the alkyne moiety in the propargylamine through palladation, yielding an alkenyl palladium intermediate. This intermediate undergoes further isomerization to form a more stable alkyl palladium species, setting the stage for the critical carbonylation step. Under the action of carbon monoxide released in situ from the formic acid and acetic anhydride mixture, an acyl palladium intermediate is generated. The cycle concludes with the oxidative addition of the diaryl iodonium salt to form a tetravalent palladium intermediate, followed by reductive elimination to release the final 2-trifluoromethyl-substituted imidazole product and regenerate the active catalyst. This intricate dance of organometallic steps ensures that the trifluoromethyl group is precisely installed at the 2-position of the imidazole ring.
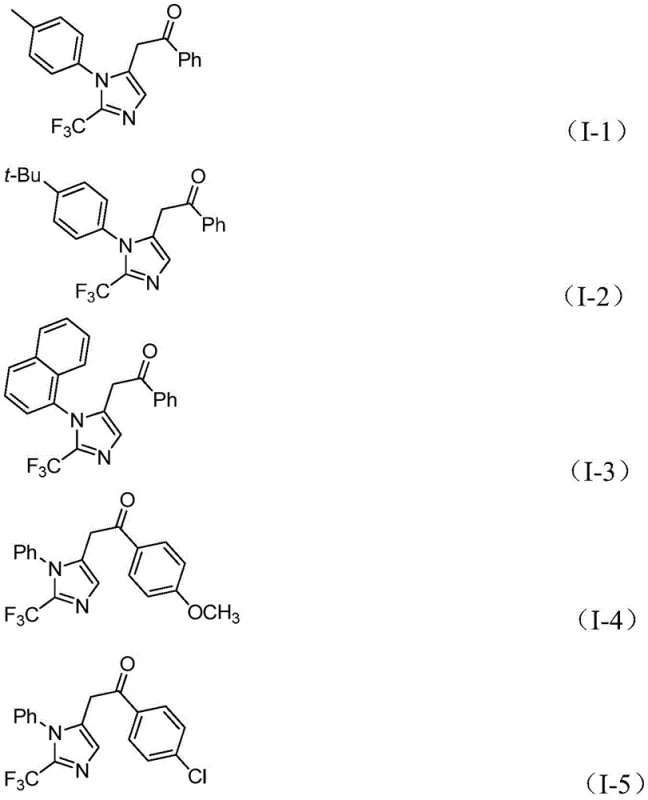
From an impurity control perspective, the choice of reagents and conditions plays a pivotal role in defining the purity profile of the final API intermediate. The use of sodium bicarbonate as a mild base helps to neutralize acidic byproducts without promoting unwanted side reactions such as hydrolysis of the sensitive imidoyl chloride or the final ketone product. The specific molar ratios, such as the preferred 1.5:1:1.5 ratio of trifluoroethylimidoyl chloride to propargylamine to diaryl iodonium salts, are optimized to drive the reaction to completion while minimizing the accumulation of unreacted starting materials. The patent data demonstrates exceptional substrate compatibility, with yields reaching as high as 97% for electron-deficient aryl groups and maintaining robust performance with sterically hindered substrates like naphthyl groups. The structural diversity achievable is evident in the specific examples provided, showcasing the method's capability to produce various derivatives including those with methoxy, chloro, bromo, and nitro substituents, all of which are valuable handles for downstream medicinal chemistry modifications.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
To implement this synthesis effectively, precise adherence to the optimized reaction parameters is essential for maximizing yield and minimizing impurities. The process begins with the careful selection of the organic solvent, with tetrahydrofuran (THF) being identified as the preferred medium due to its ability to effectively dissolve all reactants and promote high conversion rates. The catalyst system, comprising palladium chloride and triphenylphosphine, must be freshly prepared or handled under inert conditions to prevent oxidation which could deactivate the catalytic cycle. Detailed standardized synthesis steps, including specific workup procedures like filtration and silica gel mixing followed by column chromatography, are critical for isolating the high-purity product required for pharmaceutical applications. The following guide outlines the procedural framework derived directly from the patent examples to ensure reproducibility and success in your laboratory or pilot plant.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture.
- Stir the reaction at 30°C for 16-24 hours, then filter and purify via column chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative advantages in terms of cost structure and logistical reliability. The primary driver for cost reduction lies in the substitution of expensive and hazardous specialty reagents with commodity chemicals that are widely available in the global market. Trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts are all commercially accessible at scale, eliminating the need for custom synthesis of dangerous precursors like trifluorodiazoethane. This shift fundamentally alters the cost basis of the raw materials, leading to substantial cost savings in the overall manufacturing budget. Furthermore, the simplicity of the post-treatment process, which involves standard filtration and chromatography rather than complex distillation or crystallization sequences, reduces the operational expenditure associated with purification and waste management. The elimination of high-pressure carbon monoxide cylinders in favor of a liquid CO source (formic acid/acetic anhydride) also simplifies facility requirements and enhances workplace safety, indirectly lowering insurance and compliance costs.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive palladium chloride compared to more exotic noble metal catalysts, coupled with the high atom economy of the multicomponent reaction. By avoiding the need for cryogenic conditions or high-pressure reactors, the capital expenditure for equipment is significantly minimized. The high yields reported, often exceeding 90% for optimized substrates, mean that less raw material is wasted, directly improving the cost-per-kilogram metric. Additionally, the mild reaction temperature of 30°C drastically reduces energy consumption for heating or cooling, contributing to a lower carbon footprint and reduced utility bills. The ability to recycle solvents like THF further enhances the economic efficiency of the process, making it highly competitive for large-volume production runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on feedstock chemicals that have multiple global suppliers, reducing the risk of single-source bottlenecks. The stability of the starting materials allows for longer storage times and easier transportation compared to unstable diazo compounds, facilitating better inventory management and just-in-time manufacturing strategies. The robustness of the reaction against variations in substrate electronics means that supply disruptions for one specific aromatic amine can often be mitigated by switching to alternative analogs without re-optimizing the entire process. This flexibility ensures continuous production schedules and reliable delivery timelines for downstream customers, which is critical for maintaining the integrity of the pharmaceutical supply chain.
- Scalability and Environmental Compliance: The patent explicitly highlights the potential for industrial large-scale production, noting that the method can be expanded from milligram to gram levels seamlessly. The use of benign solvents and the absence of toxic heavy metal waste streams (beyond the standard palladium residue which can be recovered) align with increasingly strict environmental regulations. The simplified workup procedure generates less hazardous waste, easing the burden on waste treatment facilities and reducing disposal costs. The process design inherently supports green chemistry principles by maximizing resource efficiency and minimizing the generation of auxiliary substances, making it an attractive option for companies aiming to meet sustainability goals while scaling up complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this synthesis route. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing manufacturing workflows.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method utilizes cheap and readily available starting materials like trifluoroethylimidoyl chloride instead of hazardous trifluorodiazoethane. It operates under mild conditions (30°C) with high substrate compatibility and excellent yields up to 97%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method can be expanded to the gram level and provides possibilities for industrial large-scale production due to simple operation and easy post-treatment procedures.
Q: What catalyst system is employed in this transformation?
A: The reaction employs a palladium catalytic system consisting of palladium chloride (PdCl2) and triphenylphosphine (PPh3) ligand, utilizing formic acid and acetic anhydride as a carbon monoxide source.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN111423381B for accelerating drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity 2-trifluoromethyl imidazole intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex fluorinated chemistry allows us to offer customized solutions that align perfectly with your specific project requirements and timeline constraints.
We invite you to collaborate with us to leverage this cutting-edge technology for your next pharmaceutical project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can optimize your supply chain and reduce your overall time-to-market. Let us be your partner in turning innovative chemical concepts into commercial reality.
