Revolutionizing Phosphoramide Production: A Safer Copper-Catalyzed Reductive Coupling Strategy for Commercial Scale
The landscape of organophosphorus chemistry is undergoing a significant transformation with the emergence of safer and more efficient synthetic methodologies, as detailed in the recent patent CN115677760A. This intellectual property introduces a groundbreaking reductive coupling strategy for the synthesis of phosphoramide compounds, a class of molecules critical to the development of advanced pharmaceutical intermediates and agrochemical agents. Traditionally, the construction of the P(=O)-N bond has been fraught with challenges involving hazardous reagents and苛刻 reaction conditions. However, this new protocol leverages a copper-catalyzed system to directly couple aromatic nitro compounds with phosphine oxyhydrogen compounds. For R&D directors and procurement specialists seeking a reliable phosphoramide supplier, understanding this technological shift is paramount, as it promises not only enhanced safety profiles but also a streamlined pathway for the commercial scale-up of complex phosphoramides. The method fundamentally alters the synthetic logic by replacing oxidative protocols with a reductive manifold, thereby opening new avenues for cost reduction in pharmaceutical intermediates manufacturing while maintaining high purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phosphoramides has relied heavily on the nucleophilic substitution of phosphoryl chlorides with amines or the oxidative dehydrogenation coupling of phosphine oxides with amines. These conventional routes present severe limitations that hinder industrial adoption. The use of phosphoryl chlorides, such as diphenylphosphinous chloride, introduces significant safety hazards due to their instability and corrosiveness, requiring specialized handling equipment and rigorous waste management protocols. Furthermore, oxidative coupling methods, while avoiding chlorides, necessitate the use of large quantities of strong oxidants to activate the substrates. This reliance on oxidants not only increases the risk of thermal runaway and explosion but also limits functional group tolerance, often leading to the degradation of sensitive moieties within the molecule. Additionally, the strong coordination ability of amine substrates in oxidative systems can saturate the active sites of transition metal catalysts, leading to catalyst deactivation and inconsistent yields. These factors collectively contribute to high production costs, extended lead times for high-purity phosphoramides, and a complex impurity profile that complicates downstream purification.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN115677760A offers a paradigm shift by employing a reductive coupling mechanism. This innovative approach utilizes simple, inexpensive, and commercially abundant aromatic nitro compounds as the nitrogen source, bypassing the need for pre-formed amines entirely. By reacting these nitro compounds with phosphine oxyhydrogen compounds in the presence of a copper catalyst, a ligand, and a silane reagent, the system achieves direct P-N bond formation under mild reducing conditions. 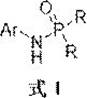 This eliminates the requirement for dangerous oxidants and unstable chlorides, fundamentally enhancing the safety profile of the operation. The use of nitro compounds, which exhibit weak coordination properties, prevents the catalyst poisoning often observed with amine substrates, ensuring sustained catalytic activity and superior reaction efficiency. This novel route represents a robust solution for the commercial scale-up of complex phosphoramides, offering a cleaner, safer, and more economically viable alternative to legacy technologies.
This eliminates the requirement for dangerous oxidants and unstable chlorides, fundamentally enhancing the safety profile of the operation. The use of nitro compounds, which exhibit weak coordination properties, prevents the catalyst poisoning often observed with amine substrates, ensuring sustained catalytic activity and superior reaction efficiency. This novel route represents a robust solution for the commercial scale-up of complex phosphoramides, offering a cleaner, safer, and more economically viable alternative to legacy technologies.
Mechanistic Insights into Copper-Catalyzed Reductive Coupling
The core of this technological advancement lies in the intricate interplay between the copper catalyst, the silane reductant, and the unique electronic properties of the substrates. The reaction initiates with the activation of the phosphine oxyhydrogen compound by the copper center, facilitated by the silane reagent which serves as the terminal reductant. Unlike oxidative pathways that generate water or other oxidized byproducts, this reductive cycle consumes the oxygen from the nitro group to form the P=O bond while simultaneously reducing the nitrogen center to form the P-N linkage. The specific choice of ligands, such as 1,10-phenanthroline or 2,2'-bipyridine, plays a critical role in stabilizing the copper species and modulating its redox potential to match the reduction requirements of the nitro group without over-reducing it to an amine prematurely. 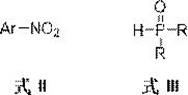 This precise control allows for the direct conversion of the nitro group into the phosphoramide functionality in a single pot. The mechanism avoids the formation of free amine intermediates in the bulk solution, which would otherwise compete for the catalyst or undergo side reactions. This mechanistic elegance ensures that the reaction proceeds with high atom economy and minimal byproduct formation, a key consideration for R&D teams focused on impurity control and process robustness.
This precise control allows for the direct conversion of the nitro group into the phosphoramide functionality in a single pot. The mechanism avoids the formation of free amine intermediates in the bulk solution, which would otherwise compete for the catalyst or undergo side reactions. This mechanistic elegance ensures that the reaction proceeds with high atom economy and minimal byproduct formation, a key consideration for R&D teams focused on impurity control and process robustness.
Furthermore, the tolerance of this catalytic system towards various functional groups is exceptional, owing to the mild nature of the reducing environment. The weak coordination of the aromatic nitro substrate ensures that the copper active sites remain accessible throughout the catalytic cycle, preventing the saturation issues that plague amine-based couplings. This results in consistent turnover numbers and allows for the use of lower catalyst loadings, typically ranging from 2% to 20% molar ratio relative to the nitro compound. The reaction conditions, operating between 100°C and 150°C in solvents like toluene or dioxane, are compatible with a wide array of substituents on both the aromatic ring and the phosphorus center. Whether the substrate contains electron-withdrawing groups like fluorine or chlorine, or electron-donating groups like methyl or methoxy, the system maintains high efficiency. This broad scope is vital for generating diverse libraries of high-purity OLED materials or pharmaceutical intermediates, enabling rapid structure-activity relationship studies without the bottleneck of incompatible synthesis routes.
How to Synthesize P,P-Diphenyl-N-(4-methylphenyl)phosphoramide Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to specific procedural guidelines that maximize yield and safety. The process begins with the careful selection of high-quality starting materials, specifically the aromatic nitro compound and the phosphine oxyhydrogen compound, ensuring they meet stringent purity specifications to minimize trace metal contamination. The reaction is conducted under an inert atmosphere, such as nitrogen or argon, to prevent oxidation of the sensitive phosphorus species and the copper catalyst. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety compliance across different batches.
- Mix aromatic nitro compounds, phosphine oxyhydrogen compounds, copper catalyst, ligand, silane reagent, and organic solvent in a reaction vessel.
- Stir the mixture under an inert gas environment at temperatures between 100°C and 150°C for 2 to 8 hours.
- Remove the solvent via reduced pressure distillation and purify the crude product using column chromatography to obtain the target phosphoramide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this reductive coupling technology translates into tangible strategic advantages that extend beyond mere chemical curiosity. The shift from hazardous phosphoryl chlorides and strong oxidants to stable nitro compounds and silanes drastically simplifies the logistics of raw material sourcing. Nitro compounds are commodity chemicals with established global supply chains, ensuring consistent availability and shielding the production schedule from the volatility associated with specialty reagents. This stability in raw material supply is crucial for maintaining continuous manufacturing operations and meeting the demanding delivery timelines of multinational clients. Moreover, the elimination of corrosive and toxic reagents reduces the burden on waste treatment facilities and lowers the overall environmental compliance costs, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The economic impact of this method is profound, primarily driven by the substitution of expensive and difficult-to-handle amines or phosphoryl chlorides with cheap aromatic nitro compounds. By integrating the reduction and coupling steps into a single operation, the process achieves significant step economy, effectively removing the need for a separate amine synthesis and isolation stage. This consolidation reduces solvent consumption, energy usage, and labor hours, leading to substantial cost savings in the overall production budget. Additionally, the avoidance of expensive oxidants and the ability to use lower catalyst loadings further drive down the variable costs per kilogram of product. These efficiencies allow for a more competitive pricing structure without compromising on the quality of the final phosphoramide intermediates.
- Enhanced Supply Chain Reliability: The robustness of the copper-catalyzed system contributes directly to supply chain resilience. Because the reaction tolerates a wide range of functional groups and operates under relatively mild thermal conditions, the risk of batch failure due to thermal excursions or side reactions is minimized. The use of stable starting materials means that inventory can be held for longer periods without degradation, providing a buffer against supply disruptions. This reliability is essential for long-term contracts where consistent delivery of high-purity intermediates is mandatory. The simplified workup procedure, involving standard distillation and chromatography, ensures that the turnaround time from reaction completion to finished goods is optimized, facilitating faster response to market demands.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the absence of highly exothermic oxidative steps and the use of common organic solvents like toluene and dioxane. The safety profile of the reductive coupling allows for larger reactor volumes without the need for exotic containment systems required for phosphoryl chlorides. From an environmental perspective, the reduction in hazardous waste generation—specifically the elimination of chloride salts and oxidized byproducts—simplifies effluent treatment and reduces the carbon footprint of the manufacturing process. This alignment with sustainability goals not only meets regulatory requirements but also enhances the brand value of the supply chain partners by supporting greener pharmaceutical and agrochemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosphoramide synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: Why is this reductive coupling method safer than traditional phosphoryl chloride methods?
A: Traditional methods often utilize unstable and corrosive phosphoryl chlorides or hazardous oxidants. This novel approach uses stable aromatic nitro compounds and phosphine oxyhydrogen compounds under reducing conditions, significantly eliminating safety risks associated with volatile chlorides and strong oxidizers.
Q: How does this method improve cost efficiency for phosphoramide manufacturing?
A: By utilizing cheap and readily available aromatic nitro compounds as nitrogen sources instead of pre-synthesized amines, the process achieves better step economy. Furthermore, avoiding expensive oxidants and complex purification steps for metal removal leads to substantial operational cost reductions.
Q: Does the weak coordination of nitro compounds affect catalyst performance?
A: On the contrary, the weak coordination property of aromatic nitro compounds prevents the saturation of transition metal binding sites, a common issue with amine substrates. This ensures higher catalytic activity and efficiency throughout the reaction cycle.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable P,P-Diphenyl-N-(4-methylphenyl)phosphoramide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed reductive coupling method for producing high-value phosphoramide compounds. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of phosphoramide intermediate meets the exacting standards required for downstream drug synthesis. We understand that consistency and quality are non-negotiable for our global partners, and our technical team is dedicated to optimizing this novel route for maximum efficiency and yield.
We invite R&D directors and procurement leaders to collaborate with us to leverage this advanced synthetic technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this reductive coupling protocol for your specific portfolio. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your needs. Let us help you secure a sustainable, cost-effective, and reliable supply of critical phosphoramide intermediates for your commercial operations.
