Advanced Copper-Catalyzed Synthesis of Niraparib Intermediates for Commercial Scale-Up
Advanced Copper-Catalyzed Synthesis of Niraparib Intermediates for Commercial Scale-Up
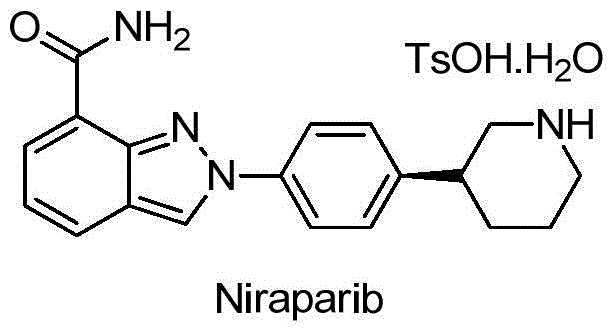
The pharmaceutical landscape for PARP inhibitors continues to evolve rapidly, with Niraparib standing out as a critical targeted therapy for BRCA-mutated ovarian and breast cancers. As demand for this potent oncology agent surges, the efficiency and safety of its supply chain become paramount concerns for global stakeholders. Patent CN111333544A introduces a groundbreaking methodology for synthesizing the key chiral intermediate, (S)-3-(4-bromophenyl)piperidine, which serves as the foundational building block for the final active pharmaceutical ingredient. This novel approach leverages a sophisticated copper-catalyzed asymmetric cyanation strategy to construct the chiral center directly, bypassing the need for hazardous azide chemistry or complex enzymatic resolutions that have historically plagued this synthetic pathway. By establishing a robust, metal-catalyzed route that operates under mild conditions, this technology offers a compelling solution for manufacturers seeking to optimize their production of high-purity pharmaceutical intermediates while adhering to stringent safety and environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the critical piperidine fragment for Niraparib has been fraught with significant operational hazards and scalability bottlenecks. Traditional routes often relied heavily on the use of sodium azide for introducing nitrogen functionality via nucleophilic substitution, a practice that poses severe safety risks due to the explosive nature of azides and the generation of toxic hydrazoic acid byproducts. Furthermore, alternative strategies attempted to circumvent these dangers through biocatalytic resolutions or enzymatic kinetic resolutions, which, while effective in achieving high enantiomeric excess, introduced substantial complexity regarding enzyme stability, specialized fermentation requirements, and high processing costs. These legacy methods frequently resulted in lengthy synthetic sequences with multiple purification steps, leading to poor overall atom economy and inflated manufacturing expenses that are unsustainable for large-scale commercial production. The reliance on such precarious chemistries not only jeopardizes worker safety but also creates fragile supply chains vulnerable to regulatory scrutiny and raw material shortages.
The Novel Approach
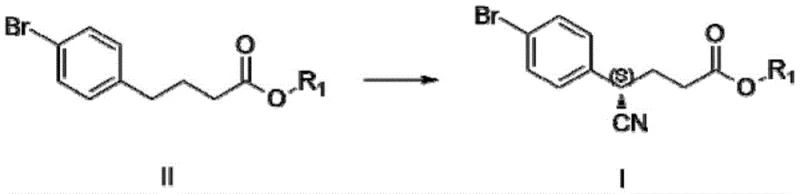
In stark contrast to these cumbersome legacy processes, the disclosed invention presents a streamlined, three-step chemical synthesis that fundamentally reimagines the construction of the chiral piperidine core. The cornerstone of this innovation is the direct asymmetric cyanation of a readily available 4-bromophenyl ester derivative using a chiral copper catalytic system. This transformation installs the crucial nitrile group and establishes the stereocenter in a single operation with high fidelity, effectively collapsing multiple synthetic steps into one efficient transformation. By utilizing robust transition metal catalysis instead of biological agents, the process achieves remarkable consistency and reproducibility, independent of the sensitivities associated with enzyme activity. The subsequent steps involve a straightforward reductive cyclization to form the lactam ring followed by a selective reduction to the amine, culminating in a final resolution step that guarantees exceptional optical purity. This cohesive strategy not only drastically shortens the production timeline but also aligns perfectly with green chemistry principles by eliminating high-risk reagents and minimizing waste generation.
Mechanistic Insights into Copper-Catalyzed Asymmetric Cyanation
The heart of this technological advancement lies in the precise orchestration of a copper(I) catalytic cycle mediated by chiral bisoxazoline ligands, which dictates the stereochemical outcome of the cyanation reaction. In this mechanism, the copper salt, typically cuprous acetate, coordinates with the chiral ligand—such as the cyclopentane-bridged bisoxazoline derivatives described in the patent—to form a highly organized chiral environment around the metal center. When exposed to the oxidant N-fluoro-bis-benzenesulfonamide and the cyanating reagent trimethylsilylcyanide, this complex facilitates the activation of the benzylic C-H bond or the addition across a transient intermediate, ensuring that the cyano group is delivered exclusively to one face of the substrate. The steric bulk and electronic properties of the ligand are meticulously tuned to maximize enantioselectivity, routinely delivering the chiral nitrile intermediate with an enantiomeric excess ranging from 86% to 87% in the crude reaction mixture. This level of control at the early stage of synthesis is critical, as it reduces the burden on downstream purification processes and ensures that the final API meets rigorous regulatory specifications for chiral impurities.
Beyond stereocontrol, the mechanistic design of this route inherently suppresses the formation of difficult-to-remove side products that often plague radical or ionic cyanation methods. The use of mild oxidants and specific solvent systems like benzene or chlorobenzene creates a reaction milieu that favors the desired catalytic turnover while minimizing non-selective background reactions. The resulting intermediate, a chiral gamma-cyano ester, possesses a stable structural motif that withstands the subsequent harsh conditions of hydrogenation and reduction without racemization. This robustness is a testament to the thoughtful engineering of the synthetic pathway, where each functional group transformation is designed to preserve the integrity of the newly formed chiral center. By avoiding the formation of unstable intermediates or highly reactive species that could lead to polymerization or decomposition, the process maintains a clean impurity profile throughout the synthesis, simplifying the isolation of the final product and reducing the need for extensive chromatographic purification.
How to Synthesize (S)-3-(4-bromophenyl)piperidine Efficiently
The practical execution of this synthesis is designed for industrial feasibility, utilizing standard reactor equipment and commercially sourced reagents to ensure seamless technology transfer from laboratory to plant. The process begins with the preparation of the chiral nitrile intermediate under inert atmosphere, followed by a catalytic hydrogenation to close the piperidine ring, and concludes with a hydride reduction to yield the free amine. Each step has been optimized to balance reaction rate with selectivity, ensuring that operators can achieve consistent results batch after batch. The detailed standardized synthesis steps for implementing this route are outlined in the guide below.
- Perform asymmetric cyanation of 4-bromophenyl ester using CuOAc, chiral bisoxazoline ligand, and TMSCN in benzene to form the chiral nitrile intermediate.
- Conduct reductive cyclization of the nitrile intermediate using Raney Nickel under hydrogen pressure (5-10 Kg) at 60-70°C to form the lactam ring.
- Reduce the lactam using sodium borohydride and boron trifluoride tetrahydrofuran, followed by resolution with L-tartaric acid to obtain the final amine with >99% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. By shifting away from hazardous azide chemistry and expensive enzymatic processes, manufacturers can significantly lower their operational overheads related to safety compliance, waste disposal, and specialized raw material sourcing. The elimination of sodium azide removes a major bottleneck in many facilities that lack the specific infrastructure required to handle explosive precursors, thereby unlocking production capacity and reducing the risk of shutdowns due to safety audits. Furthermore, the reliance on stable, shelf-stable chemical catalysts rather than perishable biological enzymes enhances supply chain resilience, ensuring that production schedules are not disrupted by the logistical challenges of cold-chain storage or limited enzyme availability. This robustness allows for more accurate forecasting and inventory management, critical factors in maintaining a steady supply of life-saving oncology medications to the global market.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the synthetic sequence and the use of cost-effective base metal catalysis. By replacing precious metal catalysts or proprietary enzymes with inexpensive copper salts and ligands, the direct material costs are drastically reduced, allowing for better margin management in a competitive generic landscape. Additionally, the shortened step count means fewer unit operations, less solvent consumption, and reduced labor hours per kilogram of output, all of which contribute to a leaner and more profitable manufacturing model. The high atom economy of the cyanation step ensures that raw materials are utilized efficiently, minimizing the volume of waste streams that require costly treatment before discharge. These cumulative efficiencies result in a substantially lower cost of goods sold, providing a competitive edge in pricing negotiations with downstream pharmaceutical partners.
- Enhanced Supply Chain Reliability: From a logistics perspective, the use of common industrial solvents and reagents ensures that the supply chain remains agile and responsive to market fluctuations. Unlike specialized biocatalysts that may have long lead times or single-source dependencies, the copper catalysts and ligands employed in this route are widely available from multiple global suppliers, mitigating the risk of supply disruptions. The process conditions, which operate at moderate temperatures and pressures, are compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, facilitating rapid scale-up without the need for capital-intensive equipment upgrades. This flexibility allows manufacturers to quickly ramp up production volumes in response to surging demand for Niraparib, ensuring continuity of supply for patients who depend on this critical therapy.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly smaller than that of traditional routes, aligning with the increasing regulatory pressure for sustainable pharmaceutical manufacturing. The avoidance of azides eliminates the generation of toxic nitrogenous waste, while the use of catalytic amounts of copper reduces the heavy metal load in the final effluent. The process generates fewer byproducts and requires less energy for heating and cooling compared to multi-step enzymatic cascades, contributing to a lower overall carbon footprint. These green chemistry attributes not only simplify regulatory filings and environmental permitting but also enhance the corporate sustainability profile of the manufacturer, a factor that is becoming increasingly important in vendor selection criteria for major pharmaceutical companies committed to ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric cyanation technology for Niraparib intermediate production. These answers are derived directly from the experimental data and process descriptions provided in the patent literature, offering clarity on the feasibility and advantages of this approach for potential licensees and manufacturing partners.
Q: How does this new route improve safety compared to traditional methods?
A: This method completely eliminates the use of sodium azide, a high-risk explosive reagent traditionally used in azide substitution reactions, thereby significantly enhancing operational safety and reducing hazardous waste disposal costs.
Q: What is the optical purity achievable with this copper-catalyzed process?
A: The process utilizes chiral bisoxazoline ligands to induce asymmetry directly at the benzylic position, achieving an initial ee of 86-87%, which is further upgraded to >99% ee after final crystallization with L-tartaric acid.
Q: Does this synthesis require expensive enzymatic catalysts?
A: No, unlike previous biocatalytic routes that require specialized enzyme cultivation and strict conditions, this chemical synthesis uses robust copper salts and commercially available ligands, simplifying supply chain logistics and reducing raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-(4-bromophenyl)piperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology drugs like Niraparib depends on a partnership rooted in technical excellence and unwavering reliability. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We are equipped with state-of-the-art rigorous QC labs and analytical capabilities to meet stringent purity specifications, guaranteeing that every batch of intermediate we deliver adheres to the highest international quality standards. Our commitment to continuous improvement means we are constantly optimizing our processes to enhance yield and reduce environmental impact, making us an ideal partner for long-term supply agreements.
We invite you to engage with our technical procurement team to discuss how this advanced copper-catalyzed route can be integrated into your supply chain to drive value and efficiency. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits this technology offers for your specific production volume and location. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring that your development timeline remains on track and your commercial goals are fully realized.
