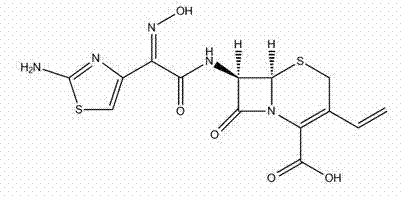
Optimizing Cefdinir Manufacturing: A Technical Breakthrough in Beta-Lactam Synthesis
The pharmaceutical landscape for third-generation cephalosporins continues to evolve, driven by the relentless demand for higher purity antibiotics and more efficient manufacturing processes. Patent CN102516261A introduces a significant advancement in the preparation of Cefdinir, a broad-spectrum oral cephalosporin known for its efficacy against Gram-positive and Gram-negative bacteria. This technical insight report analyzes the novel synthetic route which addresses critical bottlenecks in traditional production, specifically focusing on yield enhancement and crystal stability. By leveraging a specialized active ester intermediate and a refined hydrolysis protocol, this method offers a robust pathway for the commercial scale-up of complex beta-lactams. The structural integrity of the final product is paramount, as illustrated by the molecular architecture below.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefdinir has been plagued by harsh reaction conditions and suboptimal efficiency. Prior art, such as the method disclosed in Chinese patent CN101565427A, relies on low-temperature condensation reactions typically conducted at -10°C. These cryogenic requirements not only escalate energy consumption but also necessitate prolonged reaction times, often extending up to 24 hours. Furthermore, the resulting yields in these conventional processes are frequently capped at approximately 70%, with product content struggling to exceed 95%. The complexity of the reaction pathway, involving multiple protection and deprotection steps, introduces significant opportunities for impurity formation, thereby complicating downstream purification and increasing the overall cost of goods sold (COGS).
The Novel Approach
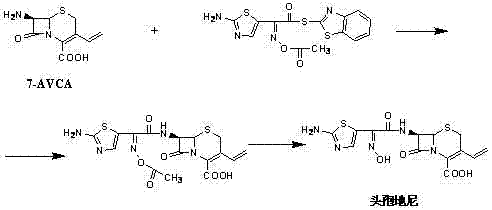
In stark contrast, the methodology outlined in CN102516261A presents a streamlined and economically superior alternative. This innovative approach utilizes (Z)-2-(2-aminothiazole-4-yl)-2-acetyloxyimino thioacetic acid (S-2-benzothiazole), referred to as the "Cefdinir active new ester," as the acylating agent. The reaction proceeds in a THF and aqueous system under the catalysis of triethylamine at much milder temperatures ranging from 10°C to 40°C. This shift away from extreme cold significantly reduces energy overheads and shortens the reaction cycle. The subsequent hydrolysis and crystallization steps are meticulously optimized to ensure that the final Cefdinir product achieves a molar yield of greater than 75% and a purity exceeding 99.5%, representing a substantial leap forward in cost reduction in antibiotic manufacturing.
Mechanistic Insights into Active Ester Acylation and Controlled Hydrolysis
The core of this technological breakthrough lies in the precise mechanism of the acylation and the subsequent controlled hydrolysis. The use of the specific active ester allows for a highly selective nucleophilic attack by the amino group of 7-AVCA (7-amino-3-vinyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid). The presence of triethylamine facilitates this coupling by neutralizing the acid byproduct, driving the equilibrium towards the formation of the cefdinir ester intermediate. Unlike traditional methods that may use less reactive acyl donors requiring forcing conditions, this active ester ensures rapid conversion even at ambient temperatures, minimizing the risk of beta-lactam ring opening or epimerization, which are common degradation pathways in cephalosporin synthesis.
Furthermore, the hydrolysis step is ingeniously managed through the addition of weak acid potassium salts, such as potassium acetate. In conventional processes, the removal of protecting groups often leads to messy reaction mixtures. Here, the addition of potassium acetate during the alkaline hydrolysis with potassium carbonate serves a dual purpose: it buffers the pH to a narrow range of 7.5 to 9.0 and promotes the orderly precipitation of the cefdinir potassium salt. This controlled crystallization is critical for excluding impurities and ensuring a stable crystal lattice. The final acidification step, carefully titrated to pH 2.4-2.5, allows for the isolation of the free acid form with exceptional purity, demonstrating a sophisticated understanding of solubility equilibria in high-purity cefdinir production.
How to Synthesize Cefdinir Efficiently
The synthesis protocol described in the patent provides a clear, three-stage roadmap for producing Cefdinir with industrial viability. The process begins with the acylation of 7-AVCA using the active ester in a biphasic solvent system, followed by a carefully monitored hydrolysis to form the potassium salt, and concludes with a pH-controlled crystallization. This sequence is designed to maximize throughput while maintaining stringent quality standards. For R&D teams looking to implement this route, the detailed operational parameters regarding solvent ratios, temperature gradients, and stoichiometric additions are critical for success. The standardized synthesis steps见下方的指南.
- React 7-AVCA with Cefdinir active new ester in THF/water using triethylamine to form the cefdinir ester liquid.
- Hydrolyze the ester liquid using ammonium chloride and potassium carbonate, adding a weak acid potassium salt to precipitate cefdinir potassium salt.
- Dissolve the potassium salt in water, adjust pH with acid, and crystallize to obtain high-purity Cefdinir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates directly into tangible operational benefits. The primary advantage is the significant improvement in process efficiency, which eliminates the need for energy-intensive cryogenic cooling systems required by older methods. By operating at near-ambient temperatures, the facility can reduce its carbon footprint and lower utility costs, contributing to a more sustainable manufacturing profile. Additionally, the shortened reaction time enhances equipment turnover rates, allowing for greater production capacity within the same timeframe. This efficiency is crucial for meeting the fluctuating demands of the global antibiotic market without compromising on delivery schedules.
- Cost Reduction in Manufacturing: The elimination of extreme low-temperature requirements drastically simplifies the engineering controls needed for the reactor setup. Traditional methods requiring -10°C demand specialized refrigeration units and extended reaction cycles, both of which inflate capital and operational expenditures. By shifting to a 10°C to 40°C window, the new method allows for the use of standard jacketed reactors with simple cooling water circulation. Furthermore, the improved yield means that less raw material is wasted per kilogram of finished API, directly lowering the variable cost of production. The high purity achieved (>99.5%) also reduces the burden on downstream purification steps, saving on solvent usage and waste disposal costs.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures a more consistent supply of reliable cefdinir intermediate supplier outputs. Older methods with long reaction times (24 hours) and sensitive conditions are prone to batch failures or delays, which can ripple through the supply chain causing shortages. The new method's shorter cycle time and tolerance for broader temperature ranges make the production schedule more predictable and resilient. This reliability is essential for pharmaceutical companies that must maintain strict inventory levels to support their own formulation lines and meet regulatory commitments.
- Scalability and Environmental Compliance: Scaling chemical processes from the lab to the plant floor often introduces unforeseen challenges, particularly with exothermic reactions or difficult filtrations. This patent describes a process where the crystallization of the potassium salt is facilitated by weak acid salts, resulting in a product with a stable crystal form and controlled particle size. This physical characteristic makes filtration and drying significantly easier on a large scale, reducing processing time and solvent retention. Moreover, the use of common solvents like THF, ethanol, and acetone, combined with the high efficiency of the reaction, minimizes the generation of hazardous waste, aligning with increasingly strict environmental regulations in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific Cefdinir preparation method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's potential impact on your manufacturing operations. Understanding these nuances is key for technical teams assessing the feasibility of technology transfer.
Q: How does the new preparation method improve upon conventional Cefdinir synthesis?
A: The novel method utilizes a specific active ester and mild hydrolysis conditions, significantly improving yield to over 75% and purity to above 99.5%, compared to older methods which often yielded less than 70% with lower content.
Q: What are the critical reaction conditions for the acylation step?
A: The acylation is conducted in a THF and water system (volume ratio 1:1 to 3:1) with triethylamine as the base. The temperature is maintained between 10°C and 40°C, ensuring gentle reaction conditions suitable for industrial scale-up.
Q: Why is the addition of weak acid potassium salts important in this process?
A: Adding weak acid potassium salts, such as potassium acetate, during the hydrolysis step helps control the pH and facilitates the precipitation of the cefdinir potassium salt, leading to a stable crystal form and easier filtration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefdinir Supplier
The technical advancements detailed in patent CN102516261A underscore the continuous innovation required to remain competitive in the pharmaceutical intermediates sector. At NINGBO INNO PHARMCHEM, we pride ourselves on our ability to translate such complex laboratory methodologies into robust, commercial-scale realities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high yield and purity are fully realized in the final product. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Cefdinir meets the highest international standards for safety and efficacy.
We invite potential partners to engage with us to explore how this optimized synthesis route can benefit your specific supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven chemical expertise. Together, we can drive efficiency and quality in the production of life-saving antibiotics.
