Advanced Base-Promoted Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Scale-Up
Advanced Base-Promoted Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Scale-Up
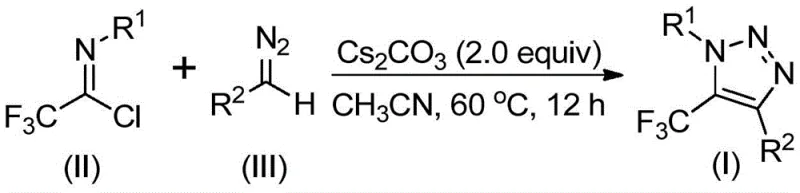
The rapid evolution of medicinal chemistry demands efficient access to fluorinated heterocycles, particularly those containing the 1,2,3-triazole motif which serves as a privileged scaffold in numerous bioactive molecules. Patent CN113121462B introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds that fundamentally shifts the paradigm from transition-metal catalysis to a simpler, base-promoted protocol. This technological breakthrough addresses critical pain points in the supply chain of high-purity pharmaceutical intermediates by eliminating the need for expensive copper catalysts and hazardous azide reagents. For R&D directors and procurement managers alike, this represents a significant opportunity to streamline synthesis routes while adhering to stricter environmental and safety regulations. The method leverages readily available starting materials to achieve high conversion rates under mild conditions, positioning it as a robust solution for the commercial scale-up of complex fluorinated intermediates required in modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on two primary strategies, both of which present substantial logistical and safety challenges for large-scale manufacturing. The first approach involves copper-catalyzed [3+2] cycloaddition of alkynes with organic azides to form triazole metal complexes, followed by reaction with trifluoromethyl reagents. The second strategy employs organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones. A critical commonality between these legacy methods is their dependence on organic azides, which are notoriously toxic, unstable, and prone to explosive decomposition, posing severe risks during storage and handling. Furthermore, the reliance on transition metals like copper necessitates rigorous purification steps to remove residual heavy metals to meet stringent pharmaceutical purity specifications, thereby increasing production costs and extending lead times for high-purity intermediates.
The Novel Approach
In stark contrast to these hazardous conventional pathways, the technology disclosed in CN113121462B utilizes a metal-free, base-promoted reaction between cheap and readily available diazo compounds and trifluoroethylimidoyl chloride. This innovative route completely bypasses the use of explosive azides and toxic trifluoromethylating agents, drastically simplifying the safety profile of the manufacturing process. By employing cesium carbonate as a promoter in an aprotic solvent, the reaction proceeds efficiently at moderate temperatures, offering a cleaner reaction profile with fewer byproducts. This shift not only enhances the safety of the operation but also reduces the complexity of the downstream processing, as there is no need for extensive metal scavenging. The ability to synthesize these valuable scaffolds without specialized equipment for handling high-energy azides makes this method exceptionally attractive for reliable agrochemical intermediate suppliers and pharmaceutical manufacturers seeking cost-effective solutions.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation offers fascinating insights into how simple reagents can be orchestrated to form complex heterocyclic systems with high regioselectivity. The reaction is hypothesized to initiate with a base-promoted intermolecular nucleophilic addition-elimination process where the diazo compound attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step facilitates the formation of a crucial carbon-carbon bond while eliminating a chloride ion, generating a reactive intermediate that sets the stage for ring closure. Subsequently, the system undergoes an intramolecular 5-endo-dig cyclization, a kinetically favorable process that constructs the five-membered triazole ring. This mechanism avoids the formation of metallacycles typical in copper-catalyzed variants, thereby preventing the entrapment of metal impurities within the crystal lattice of the final product. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for specific substrates, ensuring consistent quality and yield across different batches of production.
Furthermore, the tolerance of this mechanism towards various functional groups on both the imidoyl chloride and the diazo compound underscores its versatility in synthetic design. The electron-withdrawing nature of the trifluoromethyl group stabilizes the intermediate species, facilitating the cyclization step even under relatively mild thermal conditions ranging from 50°C to 70°C. This stability allows for the incorporation of diverse substituents such as halogens, alkoxy groups, and phospholipids without compromising the integrity of the triazole core. For R&D teams, this means the ability to rapidly generate libraries of analogs for structure-activity relationship (SAR) studies without being constrained by incompatible functional groups. The mechanistic robustness ensures that the process remains scalable, maintaining high efficiency whether performed on a milligram scale for screening or scaled up for commercial manufacturing of active pharmaceutical ingredients.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The operational simplicity of this synthesis protocol makes it highly accessible for laboratory and pilot plant settings, requiring standard glassware and common reagents. The process begins by combining cesium carbonate, 4 Å molecular sieves, trifluoroethylimidoyl chloride, and the chosen diazo compound in an organic solvent such as acetonitrile. The use of molecular sieves helps to maintain anhydrous conditions, which is critical for the stability of the diazo species and the efficiency of the base promotion. The reaction mixture is then heated to a controlled temperature, typically around 60°C, and stirred for a duration of 8 to 16 hours to ensure complete consumption of the starting materials. Following the reaction, the workup involves simple filtration to remove solids, followed by silica gel mixing and purification via column chromatography to isolate the pure triazole derivative. Detailed standardized synthesis steps are provided in the guide below.
- Combine cesium carbonate, molecular sieves, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature between 50°C and 70°C and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Perform post-treatment by filtration and silica gel mixing, followed by column chromatography purification to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthesis method offers compelling advantages that directly impact the bottom line and supply chain resilience. By eliminating the need for specialized copper catalysts and hazardous azide reagents, manufacturers can significantly reduce raw material costs and mitigate the risks associated with transporting and storing dangerous chemicals. The use of cesium carbonate, a relatively inexpensive and stable base, further contributes to cost reduction in API manufacturing by replacing costly transition metal systems. Additionally, the simplified workup procedure, which avoids complex metal removal steps, shortens the overall production cycle time, allowing for faster turnaround on custom synthesis orders. This efficiency translates into a more responsive supply chain capable of meeting the dynamic demands of the pharmaceutical and agrochemical industries without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the avoidance of specialized trifluoromethylating reagents result in substantial cost savings on raw materials. Furthermore, the simplified purification process reduces solvent consumption and waste disposal costs, leading to a more economically viable production model. The use of commercially available starting materials ensures a stable supply chain with predictable pricing, shielding manufacturers from the volatility often associated with specialty reagents.
- Enhanced Supply Chain Reliability: By removing explosive azides from the process, the logistical barriers to shipping and storing intermediates are drastically lowered, ensuring uninterrupted production schedules. The robustness of the reaction conditions allows for flexible manufacturing setups, reducing the dependency on highly specialized equipment and enabling production across multiple facilities. This decentralization capability enhances supply security, ensuring that critical pharmaceutical intermediates remain available even during regional disruptions or regulatory changes affecting hazardous material transport.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis aligns perfectly with green chemistry principles, minimizing heavy metal contamination in wastewater and reducing the environmental footprint of the manufacturing process. The method has been demonstrated to scale effectively to gram levels with high yields, indicating strong potential for ton-scale production without significant re-optimization. This scalability, combined with reduced hazardous waste generation, facilitates easier regulatory compliance and supports sustainability goals increasingly demanded by global corporate partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the method. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The responses highlight the safety, efficiency, and versatility that define this new approach to constructing fluorinated heterocycles.
Q: What are the safety advantages of this new triazole synthesis method compared to traditional routes?
A: Unlike conventional copper-catalyzed azide-alkyne cycloadditions that require toxic and explosive organic azides, this novel method utilizes stable diazo compounds and imidoyl chlorides, significantly enhancing operational safety and reducing hazardous waste handling costs.
Q: Can this synthesis protocol be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly demonstrates that the method can be easily extended to the gram level with high reaction efficiency. The use of cheap, commercially available reagents like cesium carbonate and simple workup procedures makes it highly suitable for commercial scale-up.
Q: What is the substrate scope for the R1 and R2 groups in this triazole formation?
A: The method exhibits broad functional group tolerance. R1 can be alkyl, substituted or unsubstituted aryl groups, while R2 accommodates aroyl, phospholipid, alkoxycarbonyl, or trifluoromethyl groups, allowing for the design of diverse molecular scaffolds for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for producing high-value fluorinated intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole delivered meets the highest industry standards. We are committed to leveraging our technical expertise to optimize this process for your specific needs, delivering cost-effective solutions that enhance your competitive edge in the global marketplace.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer, more efficient protocol. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver reliable, high-quality intermediates that drive your drug development programs forward with confidence and speed.
