Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Manufacturing with Safe, Metal-Free Catalysis
Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Manufacturing with Safe, Metal-Free Catalysis
The landscape of heterocyclic chemistry is undergoing a significant transformation, driven by the urgent need for safer, more sustainable, and cost-effective synthetic routes. A pivotal advancement in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift away from hazardous traditional methodologies, offering a robust platform for the production of high-value pharmaceutical and agrochemical intermediates. By leveraging a base-promoted cyclization strategy that completely bypasses the use of toxic azides and transition metal catalysts, this invention addresses critical pain points in modern process chemistry. For R&D directors and procurement managers alike, understanding the implications of this patent is essential for optimizing supply chains and ensuring the continuity of complex molecule manufacturing.
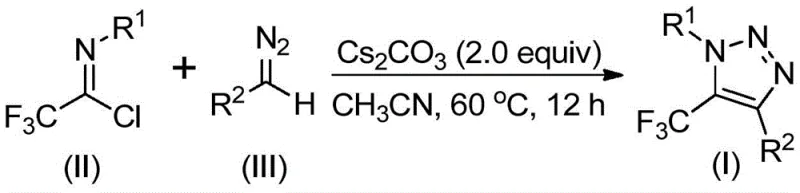
The core innovation lies in the strategic selection of starting materials: trifluoroethylimidoyl chloride and diazo compounds. Unlike conventional approaches that often struggle with the instability and safety hazards associated with organic azides, this method utilizes readily available and stable precursors. The reaction proceeds under mild thermal conditions, typically between 50°C and 70°C, utilizing cesium carbonate as a promoter in an aprotic solvent system. This not only simplifies the operational procedure but also enhances the overall safety profile of the manufacturing process. As the demand for fluorinated heterocycles continues to surge in drug discovery—driven by the trifluoromethyl group's ability to enhance metabolic stability and lipophilicity—this patent provides a commercially viable pathway to meet that demand efficiently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
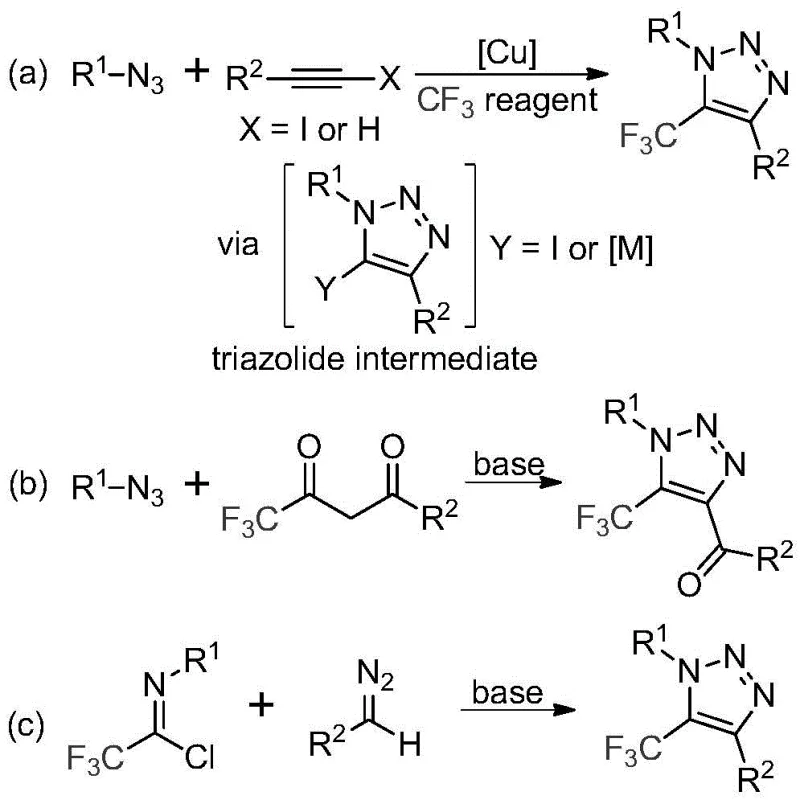
Historically, the synthesis of trifluoromethyl-substituted 1,2,3-triazoles has been dominated by two primary strategies, both of which carry significant drawbacks for large-scale industrial application. The first common method involves the copper-catalyzed [3+2] cycloaddition of alkynes with organic azides to form a triazole-metal complex, which subsequently reacts with various trifluoromethyl reagents. While effective in small-scale laboratory settings, this route is plagued by the inherent dangers of handling organic azides, which are known to be toxic and potentially explosive. Furthermore, the reliance on copper catalysts introduces the risk of heavy metal contamination, necessitating expensive and time-consuming purification steps to meet stringent pharmaceutical purity standards.
The second traditional approach utilizes organocatalytic 1,3-dipolar cycloaddition reactions between azides and trifluoromethyl ketones. Similar to the first method, this pathway is fundamentally dependent on azide chemistry, inheriting all the associated safety liabilities and regulatory hurdles. The use of explosive precursors significantly increases insurance costs, requires specialized containment infrastructure, and complicates logistics and transportation. For supply chain heads, these factors translate into higher operational expenditures and increased vulnerability to disruptions. Additionally, the atom economy and step efficiency of these legacy methods often leave room for improvement, resulting in higher waste generation and reduced overall process sustainability.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in patent CN113121462B offers a streamlined, metal-free alternative that fundamentally redesigns the synthetic logic. By employing trifluoroethylimidoyl chloride and diazo compounds as the foundational building blocks, the inventors have successfully eliminated the need for hazardous azides entirely. This substitution is not merely a change of reagents; it is a strategic overhaul that enhances process safety and operational simplicity. The reaction is promoted by cesium carbonate, a relatively inexpensive and manageable base, rather than expensive transition metals. This shift allows for a cleaner reaction profile, minimizing the formation of metal-associated impurities and simplifying the downstream purification process.
Moreover, the new approach demonstrates exceptional functional group tolerance, accommodating a wide variety of substituents on both the nitrogen and carbon positions of the triazole ring. The patent data indicates that substrates bearing electron-donating or electron-withdrawing groups, such as methyl, methoxy, halogens, and even bulky aryl groups, can be successfully converted into the desired products with good to excellent yields. This versatility is crucial for medicinal chemists who require diverse libraries of analogs for structure-activity relationship (SAR) studies. The ability to access these complex scaffolds through a single, unified protocol significantly accelerates the drug discovery timeline and reduces the cost per compound produced.
Mechanistic Insights into Base-Promoted Cyclization
To fully appreciate the technical sophistication of this invention, one must delve into the proposed reaction mechanism, which offers a fascinating glimpse into the reactivity of diazo compounds under basic conditions. The process is believed to initiate with a base-promoted intermolecular nucleophilic addition-elimination sequence. Specifically, the diazo compound, activated by the basic environment provided by cesium carbonate, acts as a nucleophile attacking the electrophilic carbon of the trifluoroethylimidoyl chloride. This step facilitates the formation of a critical carbon-carbon bond while eliminating a chloride ion, setting the stage for the subsequent ring closure.
Following the initial bond formation, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This specific mode of cyclization is kinetically favorable in this system and leads directly to the formation of the five-membered 1,2,3-triazole ring. The presence of the trifluoromethyl group plays a pivotal electronic role here, stabilizing the transition states and influencing the regioselectivity of the reaction. The result is the efficient construction of the 5-trifluoromethyl substituted 1,2,3-triazole core without the need for external oxidants or metal catalysts. This mechanistic pathway not only explains the high efficiency observed in the experimental data but also highlights the elegance of designing reactions that leverage the intrinsic reactivity of simple starting materials to build complex molecular architectures.
From an impurity control perspective, this mechanism is advantageous because it avoids the formation of metal-coordinated side products that are common in copper-catalyzed variants. The primary byproducts are likely inorganic salts derived from the base and the leaving group, which are easily removed during the aqueous workup or filtration steps. This clean reaction profile ensures that the final API intermediate meets rigorous quality specifications with minimal effort. For quality assurance teams, this translates to more consistent batch-to-batch reproducibility and a lower risk of failing release tests due to trace metal contaminants. The robustness of the mechanism against varying electronic environments on the aromatic rings further underscores its reliability for diverse synthetic applications.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
Implementing this synthesis in a practical setting requires careful attention to reaction parameters, although the protocol is designed to be user-friendly. The general procedure involves combining the trifluoroethylimidoyl chloride, the specific diazo compound, and cesium carbonate in a suitable aprotic solvent such as acetonitrile. The mixture is then heated to a moderate temperature, typically around 60°C, and maintained for a period of 8 to 16 hours to ensure full conversion. The use of molecular sieves, specifically 4 Å molecular sieves, is recommended in some embodiments to scavenge moisture and drive the equilibrium towards product formation. Upon completion, the reaction mixture is filtered to remove insoluble inorganic salts, and the filtrate is concentrated. The crude residue is then purified, usually via silica gel column chromatography, to afford the pure triazole derivative.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this technology offers tangible benefits that extend far beyond the laboratory bench. The most immediate advantage is the drastic simplification of the raw material portfolio. By replacing hazardous azides with stable diazo compounds and imidoyl chlorides, companies can significantly reduce the costs associated with special storage, handling, and disposal of dangerous goods. This shift lowers the barrier to entry for manufacturing these valuable intermediates and mitigates the risk of supply interruptions caused by regulatory crackdowns on explosive precursors. Furthermore, the elimination of transition metal catalysts removes a major cost center related to catalyst procurement and, more importantly, the extensive purification processes required to remove trace metals from the final product.
- Cost Reduction in Manufacturing: The economic impact of this metal-free protocol is substantial. Traditional copper-catalyzed routes often require ligands and scavengers to manage metal residues, adding layers of cost and complexity. In contrast, this method utilizes cesium carbonate, a commodity chemical that is both effective and affordable. The simplified workup procedure, which often involves simple filtration and standard chromatography rather than specialized metal scavenging resins, reduces solvent consumption and processing time. Consequently, the overall cost of goods sold (COGS) for the final triazole intermediate is lowered, providing a competitive edge in pricing negotiations with downstream pharmaceutical clients.
- Enhanced Supply Chain Reliability: Supply chain resilience is paramount in the post-pandemic era. The reliance on azides in conventional methods creates a fragile link in the supply chain, as their production and transport are heavily regulated. By pivoting to a chemistry based on imidoyl chlorides and diazo compounds, manufacturers can source materials from a broader range of suppliers with fewer regulatory constraints. This diversification reduces the risk of bottlenecks and ensures a steady flow of materials necessary for continuous production. Additionally, the mild reaction conditions (60°C) mean that the process can be run in standard glass-lined reactors without the need for high-pressure or cryogenic equipment, further enhancing operational flexibility and uptime.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry, the environmental footprint of a process is a key differentiator. This synthesis generates less hazardous waste compared to azide-based methods, aligning with global sustainability goals. The absence of heavy metals simplifies wastewater treatment and reduces the environmental liability associated with effluent discharge. From a scalability standpoint, the reaction has been demonstrated to proceed efficiently on a gram scale with high yields, indicating strong potential for kilogram and ton-scale production. The robustness of the reaction conditions suggests that scale-up risks are minimal, allowing for a smoother transition from pilot plant to commercial manufacturing without extensive re-optimization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for your specific manufacturing needs. Understanding these details is crucial for making informed decisions about process integration and vendor selection.
Q: Why is this new synthesis method safer than traditional azide-based routes?
A: Traditional methods rely on organic azides, which are toxic and potentially explosive. This patent utilizes stable diazo compounds and imidoyl chlorides, significantly reducing safety risks and handling costs.
Q: What are the optimal reaction conditions for scaling this process?
A: The patent specifies using cesium carbonate as a base in acetonitrile at 60°C for 12 hours. These mild conditions facilitate easy scale-up from gram to kilogram levels without specialized high-pressure equipment.
Q: Does this method require expensive transition metal catalysts?
A: No, this is a metal-free protocol. It eliminates the need for copper catalysts, thereby avoiding costly metal removal steps and ensuring higher purity suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in patent CN113121462B for the production of high-purity pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into a robust commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 5-trifluoromethyl-1,2,3-triazole meets the highest industry standards.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next project. Whether you require custom synthesis services or bulk supply of these critical intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of this metal-free technology can optimize your supply chain and reduce your overall manufacturing costs.
