Advanced Chiral Copper Complex Catalysts for High-Yield Pharmaceutical Intermediate Manufacturing
Advanced Chiral Copper Complex Catalysts for High-Yield Pharmaceutical Intermediate Manufacturing
The landscape of modern organic synthesis is continually evolving, driven by the relentless demand for higher efficiency, superior stereocontrol, and environmentally benign processes in the production of active pharmaceutical ingredients. A pivotal development in this arena is documented in patent CN102010443A, which introduces a novel chiral phosphonous bis-amino-oxazoline copper complex. This sophisticated metal-organic coordination compound represents a significant leap forward in asymmetric catalysis, specifically tailored for the Henry reaction, a fundamental carbon-carbon bond-forming transformation. For R&D directors and procurement specialists alike, understanding the structural nuances and catalytic prowess of this complex is essential for optimizing synthetic routes. The invention details a robust methodology for preparing this complex from di{N-2-[(4S)-4,5-dihydro-4-phenyl-2-oxazoline]-diphenyl-phenylphosphonic diamide and copper acetate monohydrate, resulting in a catalyst that delivers exceptional yields.
Furthermore, the commercial implications of adopting such advanced catalytic systems cannot be overstated for a reliable pharmaceutical intermediate supplier. The ability to achieve yields up to 98% in the Henry reaction of benzaldehyde signifies a drastic reduction in raw material waste and downstream purification costs. This patent not only elucidates the chemical structure but also provides a scalable synthesis method involving reflux in absolute alcohol, followed by crystallization to obtain reddish-brown crystals. By integrating this technology, manufacturers can address critical pain points related to cost reduction in API manufacturing and ensure a consistent supply of high-purity intermediates. The following analysis delves deep into the mechanistic advantages and commercial viability of this innovative copper complex.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-nitroalcohols via the Henry reaction has been plagued by several inherent challenges that hinder efficient large-scale production. Traditional methods often rely on stoichiometric amounts of chiral auxiliaries or less selective catalysts that require harsh reaction conditions, such as elevated temperatures or strong bases, which can lead to substrate decomposition and racemization. Moreover, many conventional transition metal complexes lack the necessary rigidity in their ligand architecture to effectively differentiate between enantiomeric transition states, resulting in poor enantiomeric excess (ee) values. This lack of stereocontrol necessitates expensive and time-consuming resolution steps, such as chiral chromatography or recrystallization, which significantly inflate the overall cost of goods. Additionally, the use of non-recyclable catalysts generates substantial heavy metal waste, posing severe environmental compliance issues and complicating waste management protocols for supply chain heads.
The Novel Approach
In stark contrast, the novel approach presented in patent CN102010443A utilizes a meticulously designed chiral phosphonous bis-amino-oxazoline ligand system coordinated with copper to overcome these longstanding limitations. This new catalyst features a unique structural motif where the oxazoline rings and the phosphonamide backbone create a highly defined chiral pocket around the copper center. This rigid architecture ensures precise spatial arrangement of the substrates during the catalytic cycle, thereby enforcing high stereoselectivity without the need for extreme conditions. The synthesis of the ligand itself is streamlined, involving the reaction of 2-cyano-aniline with L-phenylglycinol followed by phosphorylation, as illustrated in the reaction scheme below.
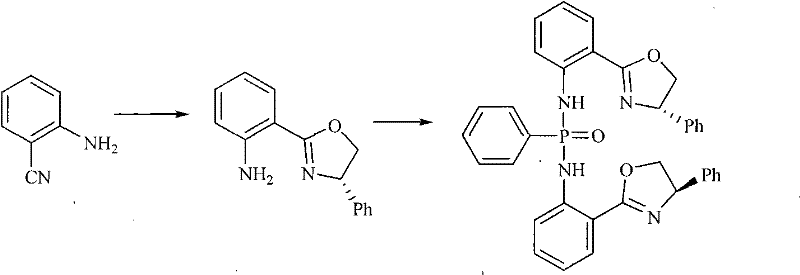
By employing this novel complex, the reaction proceeds smoothly in methanol at room temperature, eliminating the energy costs associated with heating and the safety risks of handling aggressive reagents. The result is a process that not only achieves a remarkable 98% yield but also simplifies the workup procedure, as the product can be isolated via standard column chromatography. This represents a paradigm shift towards greener, more economical synthesis strategies for complex pharmaceutical intermediates.
Mechanistic Insights into Cu-N Coordinated Asymmetric Catalysis
To fully appreciate the efficacy of this catalyst, one must examine the intricate coordination chemistry that governs its function. The core of the catalyst is the copper ion, which acts as a Lewis acid to activate the nitroalkane substrate. The ligand, di{N-2-[(4S)-4,5-dihydro-4-phenyl-2-oxazoline]-diphenyl-phenylphosphonic diamide, binds to the copper center through its nitrogen atoms, forming a stable chelate ring structure. The specific stereochemistry of the ligand, derived from the (4S)-configuration of the oxazoline rings, imparts a chiral environment that dictates the facial selectivity of the nucleophilic attack on the aldehyde. The phenyl groups on the oxazoline rings provide steric bulk that further shields one face of the reactive intermediate, ensuring that the reaction proceeds through a single, energetically favored transition state. This precise control is critical for producing single-enantiomer products required in modern drug discovery.
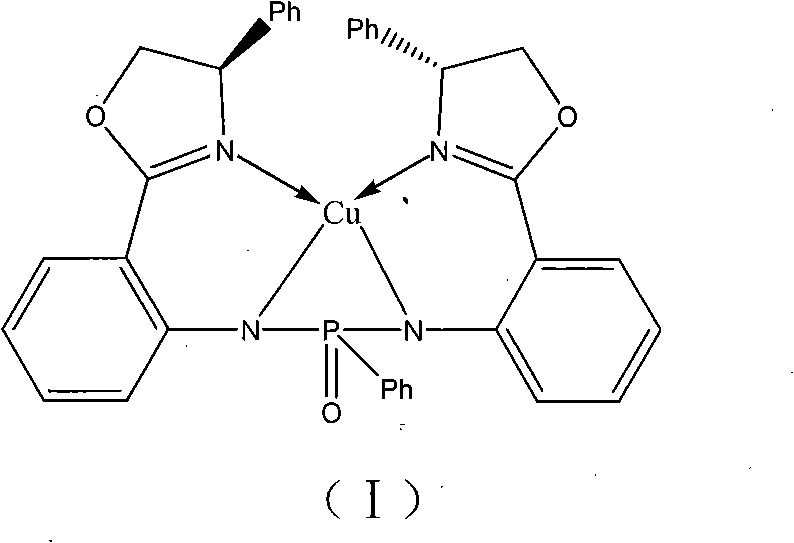
The mechanism likely involves the formation of a copper-nitronate species, which then attacks the carbonyl carbon of the benzaldehyde. The rigidity of the bis-amino-oxazoline framework prevents conformational flexibility that could lead to non-selective pathways. Furthermore, the phosphonamide linkage adds electronic tuning capabilities, potentially enhancing the Lewis acidity of the copper center compared to simple bis-oxazoline ligands. This enhanced activity allows the reaction to proceed efficiently even at low catalyst loadings (10 mol%), as demonstrated in the patent examples. The stability of the complex in alcoholic solvents like methanol is another key factor, preventing ligand dissociation and catalyst deactivation during the prolonged reaction times often required for high conversion. Understanding these mechanistic details allows chemists to fine-tune reaction parameters for optimal performance in diverse synthetic applications.
Regarding impurity control, the high selectivity of this catalyst inherently minimizes the formation of byproducts such as the dehydration product (nitroalkene) or the homo-coupling products often seen in less controlled Henry reactions. The clean reaction profile means that the crude product contains fewer impurities, reducing the burden on purification units. This is particularly advantageous for the production of high-purity OLED material precursors or sensitive agrochemical intermediates where trace impurities can be detrimental. The crystalline nature of the catalyst itself, described as reddish-brown crystals, also suggests a high degree of purity and homogeneity, which translates to batch-to-batch consistency in catalytic performance. Such reliability is paramount for maintaining stringent quality standards in regulated industries.
How to Synthesize Chiral Copper Complex Efficiently
The synthesis of this high-performance catalyst is designed to be accessible yet rigorous, ensuring that the final product meets the exacting standards required for asymmetric catalysis. The process begins with the preparation of the chiral ligand under strictly anhydrous and oxygen-free conditions to prevent oxidation of the phosphorus center or hydrolysis of the intermediate imines. The subsequent complexation with copper acetate monohydrate in absolute ethanol is a straightforward coordination reaction that leverages the solubility differences to isolate the pure complex. Detailed standard operating procedures for each step, including specific molar ratios, temperature controls, and purification techniques like column chromatography, are essential for reproducibility. For a comprehensive guide on executing this synthesis with precision, please refer to the standardized protocol outlined below.
- Prepare the chiral ligand by reacting 2-cyano-aniline with L-phenylglycinol under anhydrous conditions with ZnCl2 catalyst, followed by purification via column chromatography.
- Synthesize the bis-amino-oxazoline precursor by reacting the intermediate with phenylphosphonyl chloride in toluene and triethylamine under reflux.
- Form the final copper complex by reacting the ligand with copper acetate monohydrate in absolute ethanol, filtering, and crystallizing the reddish-brown product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this chiral copper complex technology offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary value proposition lies in the dramatic improvement in process efficiency, which directly correlates to cost reduction in pharmaceutical intermediate manufacturing. By achieving yields approaching quantitative levels (98%), the process minimizes the consumption of expensive starting materials like benzaldehyde and nitromethane, thereby lowering the variable cost per kilogram of the final product. Furthermore, the mild reaction conditions—specifically the use of methanol at ambient temperature—eliminate the need for specialized high-pressure reactors or extensive heating infrastructure, leading to significant capital expenditure savings and reduced energy consumption. This operational simplicity also enhances plant safety profiles, reducing insurance premiums and regulatory compliance burdens associated with hazardous high-energy processes.
- Cost Reduction in Manufacturing: The implementation of this catalyst fundamentally alters the economic model of producing chiral nitroalcohols by virtually eliminating the need for costly chiral resolution steps. In traditional workflows, obtaining a single enantiomer often requires discarding nearly half of the produced material or investing in expensive chiral separation columns; however, the high enantioselectivity of this copper complex ensures that the desired isomer is formed predominantly from the outset. This atom-economic approach drastically reduces waste disposal costs and maximizes the throughput of existing reactor volumes. Additionally, the catalyst loading of 10% is competitive, and the potential for ligand recovery or catalyst recycling in future iterations could further drive down the cost of goods, making the process highly attractive for margin-sensitive generic drug production.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this synthetic route ensures a more predictable and continuous flow of materials. The starting materials, such as 2-cyano-aniline and L-phenylglycinol, are commercially available commodity chemicals with stable supply chains, reducing the risk of raw material shortages that often plague specialized synthetic routes. The synthesis of the catalyst itself does not rely on exotic or geopolitically sensitive rare earth metals, relying instead on abundant copper, which mitigates supply risk. Moreover, the stability of the final complex allows for easier storage and transportation, enabling manufacturers to maintain strategic stockpiles without significant degradation, thus buffering against market volatility and ensuring on-time delivery to downstream customers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of common solvents like ethanol and toluene, which are well-understood in industrial settings and easy to recover and recycle. The absence of toxic heavy metals other than copper, which is relatively benign and easier to remove to ppm levels compared to palladium or platinum, simplifies the purification of the final API intermediate. This aligns perfectly with increasingly stringent global environmental regulations regarding heavy metal residues in pharmaceuticals. The simplified workup, involving filtration and crystallization rather than complex distillations or extractions, reduces the generation of solvent waste, supporting corporate sustainability goals and reducing the carbon footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the application and sourcing of this chiral copper complex. These insights are derived directly from the experimental data and structural analysis provided in the patent literature, aiming to clarify its utility for potential partners. Understanding the specific reaction scope, stability profiles, and handling requirements is crucial for successful technology transfer. We encourage technical teams to review these points carefully to assess the fit for their specific synthetic challenges.
Q: What is the primary advantage of this chiral copper complex in organic synthesis?
A: The primary advantage is its exceptional catalytic performance in the Henry reaction, achieving yields up to 98% while maintaining high enantioselectivity due to the rigid chiral environment provided by the bis-amino-oxazoline ligand.
Q: What are the reaction conditions required for using this catalyst?
A: The catalyst operates effectively under mild conditions, typically in methanol solvent at room temperature, which simplifies the process compared to traditional methods requiring extreme temperatures or pressures.
Q: Is this catalyst suitable for large-scale industrial production?
A: Yes, the synthesis involves standard organic operations like reflux and column chromatography, and the catalyst itself is stable enough for potential scale-up, making it viable for commercial pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Copper Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in the lab can be reliably replicated on an industrial scale. We understand the critical importance of stringent purity specifications and rigorous QC labs in the pharmaceutical sector, and our facilities are equipped to handle the precise synthesis and analysis required for high-value chiral catalysts. By leveraging our infrastructure, clients can accelerate their development timelines and secure a stable supply of this advanced catalytic technology.
We invite you to engage with our technical procurement team to discuss how this chiral phosphonous bis-amino-oxazoline copper complex can optimize your specific synthetic routes. We are prepared to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this high-yield methodology for your projects. Please contact us to request specific COA data and route feasibility assessments tailored to your production needs, and let us demonstrate how our commitment to innovation can drive value for your organization.
