Advanced Capecitabine Manufacturing: Safer Lewis Acid Catalysis and Scalable Process Optimization
Introduction to Next-Generation Capecitabine Synthesis
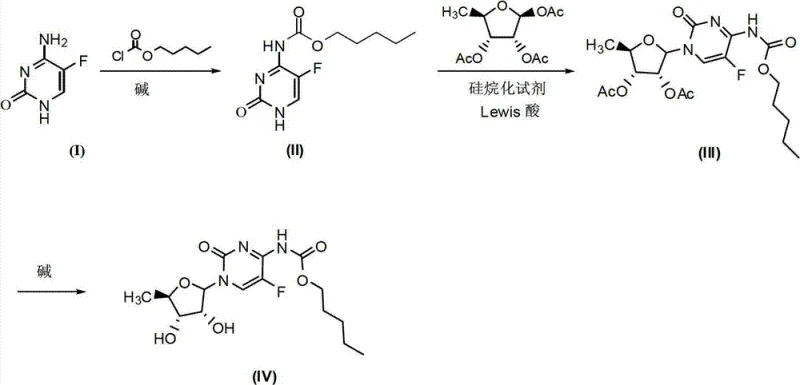
The global demand for high-quality oncology therapeutics continues to drive innovation in the manufacturing of key pharmaceutical intermediates. Patent CN102924548A introduces a transformative synthetic methodology for Capecitabine, a critical prodrug of 5-fluorouracil used extensively in the treatment of breast and colorectal cancers. This technology addresses long-standing industry pain points by replacing hazardous traditional catalysts with safer, more efficient Lewis acid systems. For R&D directors and supply chain leaders, this patent represents a significant leap forward in process safety and scalability. By eliminating the need for toxic tin tetrachloride and streamlining the purification workflow through a novel one-pot strategy, this method offers a robust pathway for the commercial scale-up of complex nucleoside analogs. The following analysis dissects the technical merits and commercial implications of this advanced synthesis route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Capecitabine has been plagued by significant safety and environmental challenges that complicate large-scale manufacturing. Traditional routes often rely on tin tetrachloride (SnCl4) as a catalyst for the glycosylation step, a reagent known for its high toxicity, tendency to absorb moisture, and difficulty in handling. Furthermore, legacy processes frequently necessitate the use of triphosgene, which carries the risk of generating lethal phosgene gas during operation, posing severe safety liabilities for production facilities. Another major bottleneck is the reliance on column chromatography for purification, a technique that is notoriously difficult to scale and results in substantial product loss. The accumulation of heavy metal residues from tin catalysts also imposes a heavy burden on downstream purification, requiring expensive removal steps to meet stringent regulatory limits for elemental impurities in active pharmaceutical ingredients.
The Novel Approach
The methodology disclosed in CN102924548A fundamentally reengineers the synthesis pathway to overcome these structural inefficiencies. Instead of toxic tin salts, the process utilizes a selection of milder and safer Lewis acids such as magnesium bromide, zinc chloride, or boron trifluoride etherate. This substitution not only mitigates occupational health risks but also simplifies the waste treatment protocol. A standout feature of this innovation is the implementation of a telescoped one-pot reaction for the coupling step. By reacting the protected cytosine intermediate directly with the silylated sugar donor in the presence of the Lewis acid, the need to isolate the unstable intermediate (Compound III) is completely eliminated. This continuous flow from coupling to hydrolysis drastically reduces solvent usage and processing time, establishing a new benchmark for cost reduction in API manufacturing while maintaining high stereochemical control.
Mechanistic Insights into Lewis Acid-Catalyzed Glycosylation
The core chemical innovation lies in the activation of the glycosyl donor through a coordinated Lewis acid mechanism that ensures high regioselectivity and yield. In the critical coupling step, the silylating agent first protects the exocyclic amine of the 5-fluorocytosine derivative, enhancing its nucleophilicity and solubility in organic media. Simultaneously, the Lewis acid coordinates with the acetyl groups of the ribofuranose donor, facilitating the formation of an oxocarbenium ion intermediate. This activated species is then attacked by the nitrogen atom of the pyrimidine ring to form the N-glycosidic bond. Unlike the harsh conditions required by tin catalysts, this system operates effectively at temperatures ranging from 25°C to 110°C, providing operators with a wide thermal window to optimize reaction kinetics without degrading the sensitive fluorinated scaffold. The choice of solvent, such as dichloromethane or acetonitrile, further modulates the polarity of the transition state, ensuring the preferential formation of the desired beta-anomer.
From an impurity control perspective, this mechanism offers distinct advantages over prior art by minimizing side reactions associated with heavy metal coordination. The absence of tin prevents the formation of stable organotin byproducts that are notoriously difficult to separate from the final product matrix. Additionally, the mild basic conditions used in the initial amidation step (using pyridine or DIPEA) prevent the degradation of the fluoropyrimidine ring, which can occur under strongly acidic or nucleophilic conditions. The final hydrolysis step employs mild alkalis like lithium hydroxide or ammonia water to cleave the acetyl protecting groups without affecting the carbamate linkage at the N4 position. This precise control over reaction orthogonality ensures a clean impurity profile, significantly reducing the burden on analytical quality control teams and accelerating batch release times for high-purity pharmaceutical intermediates.
How to Synthesize Capecitabine Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and moisture control, particularly during the silylation and coupling phases. The process begins with the protection of 5-fluorocytosine, followed by the direct addition of the sugar donor and catalyst in a single vessel. This streamlined approach minimizes unit operations and maximizes throughput. For detailed operational parameters, temperature profiles, and workup procedures, please refer to the standardized technical guide below which outlines the critical process parameters derived from the patent examples.
- React 5-fluorocytosine with n-pentyl chloroformate in the presence of a base like pyridine to form the protected intermediate (II).
- Perform a one-pot coupling reaction using Lewis acids (e.g., BF3·OEt2, ZnCl2) and silylating agents with acetylated ribose to form intermediate (III) without isolation.
- Hydrolyze the acetyl protecting groups using an alkaline solution (e.g., LiOH, ammonia water) to yield the final Capecitabine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology translates into tangible strategic benefits beyond mere chemical elegance. The elimination of hazardous reagents like tin tetrachloride and triphosgene simplifies the logistics of raw material sourcing, as these safer alternatives are more readily available and less regulated. This shift reduces the administrative overhead associated with handling dangerous goods and lowers the insurance premiums for manufacturing sites. Furthermore, the telescoped nature of the reaction sequence means fewer discrete processing steps, which directly correlates to reduced labor costs and lower energy consumption per kilogram of product. By removing the column chromatography requirement, the process becomes inherently more suitable for multi-ton production, ensuring a stable and continuous supply of material for downstream formulation.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the significant improvement in overall yield achieved through the one-pot methodology. By avoiding the isolation of intermediate Compound III, manufacturers eliminate the mechanical losses typically associated with filtration, drying, and transfer operations. Additionally, the removal of expensive heavy metal scavengers and the reduction in solvent volume required for chromatography lead to substantial cost savings in raw materials and waste disposal. The use of recyclable Lewis acids and common organic solvents further enhances the economic viability of the process, allowing for competitive pricing structures in the global market for oncology intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized or hazardous reagents that may face shipping restrictions or supply shortages. This new method utilizes commodity chemicals such as zinc salts, magnesium bromide, and standard silylating agents, which are produced by a wide network of global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks. Moreover, the robustness of the reaction conditions allows for manufacturing in a broader range of facilities, increasing the resilience of the supply chain against regional disruptions. The simplified purification process also shortens the production cycle time, enabling faster response to fluctuations in market demand for Capecitabine.
- Scalability and Environmental Compliance: As regulatory pressure on pharmaceutical manufacturing intensifies, the environmental footprint of a synthesis route becomes a critical factor in vendor selection. This process aligns perfectly with green chemistry principles by reducing the E-factor (mass of waste per mass of product) through solvent minimization and catalyst replacement. The absence of toxic tin waste simplifies effluent treatment and ensures compliance with increasingly strict environmental regulations. From a scalability standpoint, the exothermic profile of the reaction is manageable, and the lack of complex purification steps makes the technology easily transferable from pilot plant to commercial scale reactors, ensuring that capacity can be ramped up rapidly to meet clinical or commercial needs without compromising quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on process feasibility and quality outcomes. Understanding these details is essential for technical teams evaluating the integration of this route into existing manufacturing portfolios.
Q: Why is replacing Tin Tetrachloride critical for Capecitabine production?
A: Traditional methods use toxic SnCl4 which poses severe environmental hazards and risks heavy metal contamination in the final API. The new method uses safer Lewis acids like MgBr2 or BF3·OEt2, eliminating the need for expensive heavy metal removal steps and ensuring stricter compliance with ICH Q3D guidelines.
Q: How does the 'one-pot' method improve manufacturing efficiency?
A: By avoiding the isolation and purification of the key intermediate (Compound III) before hydrolysis, the process significantly reduces material loss, solvent consumption, and operational time. This direct telescoping of steps enhances overall yield and simplifies the workflow for large-scale production.
Q: What are the typical reaction conditions for the glycosylation step?
A: The glycosylation proceeds under mild conditions ranging from 25°C to 110°C depending on the solvent and Lewis acid chosen. Common solvents include dichloromethane, toluene, or acetonitrile, allowing flexibility in process optimization for different reactor setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Capecitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis methodologies requires a partner with deep technical expertise and proven execution capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific requirements of Lewis acid catalysis and moisture-sensitive reactions, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets global pharmacopoeia standards. We are committed to delivering high-purity Capecitabine and related intermediates that support the development of life-saving cancer therapies.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless path from development to commercial success.
