Advanced Synthesis of Axial and P-Center Chiral Compounds for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access high-value chiral building blocks, particularly those containing phosphorus centers which are critical for asymmetric catalysis. Patent CN115477672A introduces a groundbreaking methodology for the preparation of chiral compounds containing both axial chirality and a phosphine center, addressing long-standing challenges in ligand synthesis. This innovation utilizes a palladium-catalyzed system to achieve asymmetric carbon-phosphine bond formation, a transformation that has historically been difficult to control with high stereoselectivity. By leveraging quaternary phosphonium salts and aryl or alkenyl boronic acids as starting materials, this process bypasses the need for stoichiometric chiral auxiliaries or tedious resolution steps. The technical significance of this patent lies in its ability to generate P-centered chiral phosphine compounds under remarkably mild conditions, offering a robust platform for the production of next-generation catalysts used in the synthesis of active pharmaceutical ingredients. For R&D directors and procurement specialists, understanding the mechanistic depth and commercial viability of this route is essential for optimizing supply chains and reducing the cost of goods for complex chiral intermediates.
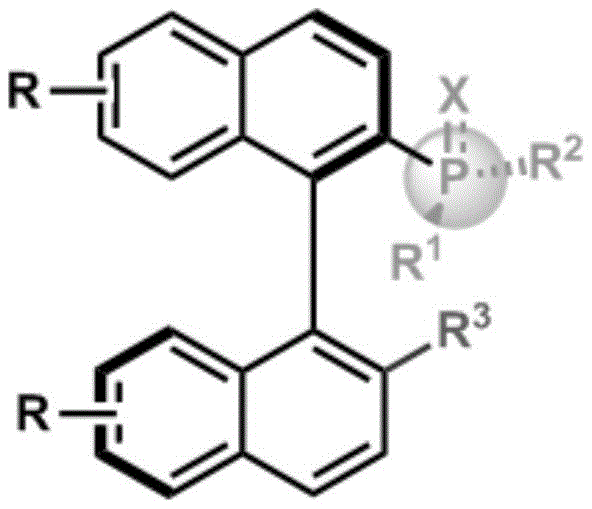
The development of P-chiral ligands has traditionally been hindered by the reliance on classical resolution techniques, which inherently cap the maximum theoretical yield at 50% and generate significant waste in the form of unwanted enantiomers. Conventional methods often require expensive chiral resolving agents and multiple crystallization steps, leading to prolonged production cycles and inflated manufacturing costs that are unsustainable for large-scale commercial operations. Furthermore, the structural complexity of binaphthyl-based or spiro-type phosphine ligands often necessitates harsh reaction conditions that can compromise functional group tolerance, limiting the scope of substrates that can be effectively utilized in downstream applications. In contrast, the novel approach detailed in this patent employs a catalytic asymmetric strategy that fundamentally shifts the paradigm from separation to direct synthesis. By utilizing a palladium catalyst in conjunction with a chiral phosphoramidite ligand, the method enables the direct construction of the stereogenic phosphorus center with high fidelity. This transition from stoichiometric resolution to catalytic asymmetry not only enhances the overall atom economy but also simplifies the purification workflow, as the reaction proceeds with high diastereoselectivity and enantiomeric excess, reducing the burden on downstream processing units.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of P-chiral phosphine ligands has been dominated by resolution strategies that are inherently inefficient and resource-intensive. These traditional pathways typically involve the synthesis of a racemic mixture followed by separation using chiral acids or bases, a process that discards half of the produced material and requires significant solvent volumes for recrystallization. The economic impact of this 50% yield ceiling is profound, effectively doubling the raw material costs and increasing the environmental footprint due to the disposal of the unwanted enantiomer. Additionally, the operational complexity of resolution processes often introduces variability in batch-to-batch consistency, posing risks for supply chain reliability and regulatory compliance in highly controlled pharmaceutical manufacturing environments. The reliance on specific chiral pool materials further restricts the structural diversity of accessible ligands, limiting the ability of chemists to fine-tune steric and electronic properties for specific catalytic applications. These limitations underscore the urgent need for a more direct, catalytic approach that can overcome the thermodynamic and kinetic barriers associated with creating phosphorus stereocenters without the inefficiencies of resolution.
The Novel Approach
The methodology disclosed in patent CN115477672A represents a significant leap forward by utilizing a palladium-catalyzed asymmetric cleavage of carbon-phosphine bonds to construct the desired chiral architecture. This innovative route employs readily available quaternary phosphonium salts and boronic acids, which are stable and commercially accessible feedstocks, thereby enhancing the robustness of the supply chain. The reaction proceeds under mild thermal conditions, typically around 40°C, which minimizes energy consumption and reduces the risk of thermal degradation of sensitive functional groups on the substrate. The use of a chiral phosphoramidite ligand ensures that the stereochemical information is effectively transferred to the phosphorus center, achieving enantiomeric excess values as high as 98% and diastereomeric ratios exceeding 25:1. This high level of stereocontrol eliminates the need for subsequent enrichment steps, streamlining the production process and significantly lowering the cost of manufacturing. Furthermore, the compatibility of this system with various substituents on the boronic acid and phosphonium salt allows for a broad scope of derivatives, providing R&D teams with the flexibility to optimize ligand performance for specific asymmetric transformations.
Mechanistic Insights into Pd-Catalyzed Asymmetric C-P Bond Formation
The core of this technological advancement lies in the sophisticated catalytic cycle that facilitates the asymmetric coupling of the phosphonium salt and the boronic acid. The mechanism initiates with the oxidative addition of the palladium catalyst into the carbon-phosphine bond of the quaternary phosphonium salt, a step that is critically enabled by the specific electronic properties of the palladium precursor and the supporting ligands. This oxidative addition generates a key organopalladium intermediate that retains the stereochemical integrity required for the subsequent transmetallation step. The chiral phosphoramidite ligand plays a pivotal role in this stage, creating a chiral environment around the metal center that discriminates between the pro-chiral faces of the reacting species. Following transmetallation with the boronic acid species, which is activated by the base such as cesium carbonate, the reductive elimination step occurs to form the new carbon-phosphine bond. This final step releases the chiral product and regenerates the active palladium catalyst, allowing the cycle to continue with high turnover numbers. The inclusion of additives like cuprous chloride further stabilizes the reaction intermediates and enhances the rate of transmetallation, ensuring that the reaction proceeds to completion with minimal side product formation.
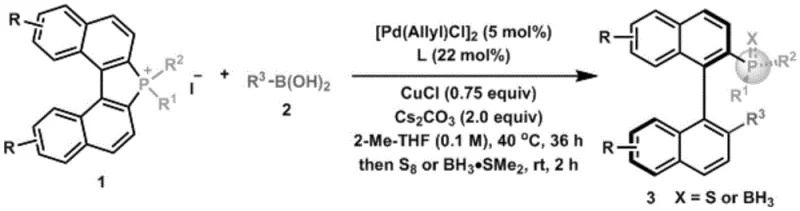
Controlling the impurity profile in the synthesis of chiral phosphine compounds is paramount for their application in pharmaceutical catalysis, where trace metals or isomeric impurities can poison downstream reactions or compromise drug safety. The described process incorporates a specific post-reaction treatment involving the addition of sulfur or borane dimethyl sulfide at room temperature, which serves to stabilize the sensitive phosphine product against oxidation. This step is crucial because free phosphines are prone to air oxidation, which would degrade the ligand quality and reduce its efficacy in subsequent catalytic applications. By converting the phosphine into a more stable sulfide or borane complex immediately after synthesis, the method ensures that the product can be isolated and stored without significant degradation. The purification process, involving filtration, concentration, and column chromatography, is designed to remove palladium residues and inorganic salts effectively. The high diastereoselectivity of the reaction (>25:1 dr) inherently limits the formation of diastereomeric impurities, simplifying the chromatographic separation and reducing the solvent load required for purification. This rigorous control over the impurity profile ensures that the final ligand meets the stringent quality specifications required for use in GMP manufacturing of active pharmaceutical ingredients.
How to Synthesize Chiral Phosphine Compounds Efficiently
The practical implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and stereoselectivity while ensuring operational safety. The process begins with the preparation of the reaction mixture under an inert atmosphere, typically nitrogen or argon, to prevent the oxidation of the palladium catalyst and the phosphine intermediates. The precise stoichiometry of the reagents, including the base, catalyst, ligand, and additives, must be maintained within the optimized ranges specified in the patent to achieve the reported performance metrics. The reaction temperature is controlled at 40°C for a duration of approximately 36 hours, allowing sufficient time for the slow but highly selective catalytic cycle to proceed to completion. Following the coupling reaction, the stabilization step with sulfur or borane dimethyl sulfide is conducted at room temperature to lock in the chirality and protect the phosphorus center. The detailed standardized synthesis steps, including specific workup procedures and purification protocols, are provided in the guide below to ensure reproducibility and scalability for industrial applications.
- Mix base, Pd catalyst, chiral phosphoramidite ligand, additive, quaternary phosphonium salt, boronic acid, and solvent under inert gas.
- Heat the mixture to 38-42°C for 35-37 hours to facilitate the asymmetric carbon-phosphine bond formation.
- Add sulfur or borane dimethyl sulfide at room temperature, then filter, concentrate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic asymmetric synthesis method offers substantial advantages over traditional resolution-based manufacturing, particularly in terms of cost structure and supply chain resilience. The elimination of the resolution step effectively doubles the theoretical yield from the same amount of starting material, which directly translates to a significant reduction in raw material costs per kilogram of final product. This improvement in atom economy also reduces the volume of waste generated, lowering the costs associated with waste disposal and environmental compliance, which are increasingly critical factors in chemical manufacturing. The use of mild reaction conditions reduces energy consumption and minimizes the need for specialized high-pressure or high-temperature equipment, allowing for production in standard glass-lined or stainless steel reactors that are widely available in the CDMO network. These factors combine to create a more cost-effective and sustainable manufacturing process that can be scaled up reliably to meet global demand.
- Cost Reduction in Manufacturing: The shift from stoichiometric resolution to catalytic asymmetric synthesis fundamentally alters the cost equation by removing the 50% yield penalty associated with discarding unwanted enantiomers. This efficiency gain means that less raw material is required to produce the same amount of active ligand, leading to substantial cost savings in the procurement of starting materials like quaternary phosphonium salts and boronic acids. Additionally, the simplified purification process reduces the consumption of solvents and chromatography media, further driving down the variable costs of production. The ability to achieve high enantiomeric excess directly from the reaction minimizes the need for costly recrystallization or chiral HPLC purification steps, streamlining the overall manufacturing workflow and reducing labor costs.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials such as aryl boronic acids and quaternary phosphonium salts enhances the robustness of the supply chain by reducing dependency on scarce or custom-synthesized chiral pool reagents. These feedstocks are produced by multiple suppliers globally, mitigating the risk of supply disruptions and providing procurement teams with greater flexibility in sourcing strategies. The mild reaction conditions also reduce the risk of batch failures due to thermal runaway or equipment limitations, ensuring consistent delivery schedules and reliable inventory levels. This stability is crucial for pharmaceutical customers who require uninterrupted supply of critical intermediates to maintain their own production schedules for active pharmaceutical ingredients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as mixing, heating, filtration, and chromatography that are easily transferred from laboratory to pilot and commercial scale. The reduced waste generation and lower energy requirements align with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals. The ability to operate at near-ambient temperatures reduces the carbon footprint of the manufacturing process, while the high selectivity minimizes the formation of by-products that require complex treatment. This environmental advantage not only reduces compliance costs but also enhances the brand value of the supply chain partners by demonstrating a commitment to sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is critical for assessing the feasibility of integrating this technology into existing manufacturing workflows and for optimizing the economic benefits of the new process. The information covers aspects ranging from reaction performance to product stability, offering a comprehensive overview of the technology's capabilities.
Q: What is the primary advantage of this Pd-catalyzed method over traditional resolution?
A: Unlike traditional resolution which limits yield to 50%, this catalytic asymmetric synthesis allows for theoretical yields up to 83% with high enantioselectivity (up to 98% ee), significantly improving atom economy.
Q: What are the key reaction conditions for scalability?
A: The process operates under mild conditions (40°C) using commercially available reagents like quaternary phosphonium salts and boronic acids, facilitating easier thermal management and scale-up.
Q: How is the P-chirality controlled in this mechanism?
A: Chirality is controlled by a chiral phosphoramidite ligand coordinated to the palladium center, which directs the asymmetric cleavage of the carbon-phosphine bond in the quaternary salt.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphine Ligand Supplier
The technical potential of this Pd-catalyzed asymmetric synthesis route represents a significant opportunity for pharmaceutical and fine chemical companies to optimize their supply chains for high-value chiral intermediates. NINGBO INNO PHARMCHEM, as a specialized CDMO partner, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative chemistry to the market. Our facility is equipped with state-of-the-art rigorous QC labs and analytical capabilities to ensure stringent purity specifications are met for every batch of chiral phosphine ligands produced. We understand the critical nature of these materials in asymmetric catalysis and are committed to delivering products that meet the highest standards of quality and consistency, enabling our clients to accelerate their drug development timelines with confidence.
We invite procurement leaders and technical directors to engage with our technical procurement team to discuss how this technology can be integrated into your specific manufacturing requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this catalytic route for your specific target molecules. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring that you have all the necessary information to make informed sourcing decisions. Our team is ready to support your transition to more efficient and sustainable manufacturing processes, providing the reliability and expertise needed to succeed in the competitive pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
