Advanced Copper-Catalyzed Synthesis of 1,2,4-Thiadiazole Derivatives for Commercial Pharmaceutical Applications
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and sustainable synthetic routes for bioactive scaffolds. A significant advancement in this field is documented in Chinese patent CN111018807B, which details a robust method for synthesizing 1,2,4-thiadiazole derivatives. Unlike the more common 1,3,4-isomers, the 1,2,4-thiadiazole core offers unique pharmacological profiles, yet its synthesis has historically been plagued by complex multi-step procedures and expensive starting materials. This new methodology leverages a copper-catalyzed oxidative cyclization strategy that directly couples phenylacetic acid derivatives with benzamidine hydrochlorides using elemental sulfur. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a pivotal shift towards cost-effective and scalable manufacturing of high-value nitrogen-sulfur heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-thiadiazole ring has been a formidable challenge in organic synthesis, often requiring harsh conditions that limit industrial applicability. Early methods, such as those reported by Haruki Eiichi in 1968, relied on the condensation of N-chlorobenzamidine with phenylthioamide, a process that necessitates the pre-synthesis of unstable and difficult-to-handle thioamide substrates. More recent literature, including work by the Xu group, utilized thioamides and nitriles with iodine as an oxidant, which, while effective, involves complex operational protocols and moderate yields. Furthermore, alternative routes employing benzyl bromides and lithium tert-butoxide introduce significant safety hazards due to the pyrophoric nature of the base and the toxicity of halogenated solvents. These conventional pathways are characterized by high material costs, difficult post-treatment procedures involving toxic waste streams, and a lack of substrate generality, severely restricting their utility in the commercial scale-up of complex pharmaceutical additives.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN111018807B introduces a streamlined, one-pot synthesis that dramatically simplifies the production workflow. By utilizing readily available phenylacetic acids and benzamidine hydrochlorides as direct precursors, the need for pre-functionalized thioamide intermediates is completely eliminated. The reaction employs elemental sulfur powder as both the sulfur source and the oxidant, mediated by a catalytic amount of inexpensive copper salts such as CuI or CuBr. This approach not only reduces the raw material costs significantly but also enhances safety by avoiding hazardous reagents like lithium tert-butoxide. The reaction proceeds in dimethyl sulfoxide (DMSO) at temperatures between 130°C and 140°C, offering a balance between reaction kinetics and thermal safety. This novel pathway provides a versatile platform for generating diverse 1,2,4-thiadiazole libraries, addressing the critical need for cost reduction in heterocyclic compound manufacturing.
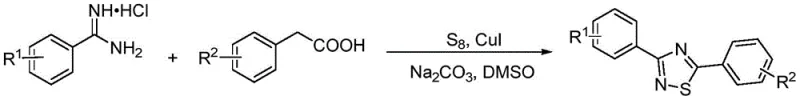
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The core of this synthetic breakthrough lies in the efficient copper-catalyzed oxidative coupling mechanism that facilitates the formation of the C-S and C-N bonds simultaneously. In this catalytic cycle, the copper species likely activates the benzylic C-H bond of the phenylacetic acid derivative, enabling a nucleophilic attack or radical coupling with the amidine nitrogen. Elemental sulfur plays a dual role; it serves as the source of the sulfur atom incorporated into the heterocyclic ring and acts as a mild oxidant to drive the aromatization of the intermediate dihydro-thiadiazole species. The use of sodium carbonate as a base is crucial, as it neutralizes the hydrochloride salt of the amidine and facilitates the deprotonation steps without inducing side reactions common with stronger bases. This mechanistic elegance ensures high product selectivity, minimizing the formation of polymeric byproducts or over-oxidized species that often plague sulfur chemistry. For technical teams, understanding this mechanism highlights the robustness of the process, as the catalyst loading is kept low (0.1 to 0.2 equivalents), reducing metal contamination risks in the final API intermediate.
Impurity control is another critical aspect where this method excels, particularly for applications requiring high-purity OLED material or pharmaceutical grades. The mild reaction conditions (130-140°C) prevent the thermal degradation of sensitive functional groups such as methoxy or trifluoromethyl substituents, which are often present in bioactive molecules. Traditional methods using strong oxidants or harsh bases often lead to decomposition or rearrangement of these groups, resulting in complex impurity profiles that are difficult to separate. In this copper-catalyzed system, the chemoselectivity is preserved, allowing for the successful synthesis of derivatives with electron-donating and electron-withdrawing groups alike. The workup procedure, involving simple extraction and column chromatography, effectively removes residual copper and sulfur byproducts. This level of purity control is essential for reducing lead time for high-purity intermediates, as it minimizes the need for extensive recrystallization or preparative HPLC purification steps downstream.
How to Synthesize 3,5-Diphenyl-1,2,4-Thiadiazole Efficiently
The practical implementation of this synthesis is straightforward, making it accessible for both laboratory discovery and pilot plant operations. The standard protocol involves dissolving the benzamidine hydrochloride and phenylacetic acid in DMSO, followed by the sequential addition of sulfur powder, sodium carbonate, and the copper catalyst. The mixture is then heated in a pressure tube or reactor to maintain the required temperature range of 130°C to 140°C for a duration of 18 to 24 hours. Reaction progress is typically monitored via TLC until the starting materials are consumed. Upon completion, the reaction mixture is cooled to room temperature, extracted with an organic solvent, dried, and concentrated. The crude product is then purified by column chromatography to afford the target 1,2,4-thiadiazole derivative as a solid or oil, depending on the substitution pattern. Detailed standardized synthesis steps see the guide below.
- Dissolve phenylacetic acid and benzamidine hydrochloride in DMSO with a molar ratio of 1-2: 1.
- Add sulfur powder, sodium carbonate, and a copper catalyst (CuI, CuBr, or Cu(OAc)2) to the mixture.
- Stir the reaction at 130-140°C for 18-24 hours, then cool, extract, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers compelling advantages that directly impact the bottom line and supply chain resilience. The shift from specialized, pre-synthesized thioamides to commodity chemicals like phenylacetic acid and benzamidine hydrochloride drastically simplifies the raw material supply chain. These starting materials are produced on a massive global scale for various industries, ensuring consistent availability and shielding manufacturers from the volatility associated with niche fine chemical suppliers. Furthermore, the elimination of hazardous reagents like lithium tert-butoxide reduces the regulatory burden and safety infrastructure costs required for storage and handling. This translates into substantial cost savings in facility maintenance and insurance, making the overall manufacturing process more economically viable for long-term production contracts.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive and complex substrates with low-cost commodity chemicals. Elemental sulfur is one of the cheapest reagents available in the chemical industry, and copper salts are significantly less expensive than precious metal catalysts like palladium or rhodium often used in cross-coupling reactions. Additionally, the low catalyst loading (10-20 mol%) further reduces the material cost per kilogram of product. By streamlining the synthesis into a single pot, the process eliminates multiple isolation and purification steps associated with multi-step routes, thereby reducing labor costs, solvent consumption, and energy usage. These factors combine to deliver a highly competitive cost structure for producing high-purity 1,2,4-thiadiazole derivatives.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for pharmaceutical and agrochemical manufacturers, and this method strengthens reliability by diversifying the source of key inputs. Since phenylacetic acids and benzamidines are widely available from multiple global vendors, the risk of supply disruption due to a single supplier failure is minimized. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the rate of batch failures. This stability allows for more accurate production planning and inventory management, ensuring that critical intermediates are available when needed for downstream API synthesis. Consequently, partners can rely on a more predictable and resilient supply network for their heterocyclic building blocks.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant often reveals hidden bottlenecks, but this methodology is inherently designed for scalability. The use of DMSO, a high-boiling polar aprotic solvent, facilitates heat transfer and mixing in large reactors, while the absence of gaseous reagents or highly exothermic steps simplifies process control. From an environmental standpoint, the avoidance of halogenated solvents and toxic heavy metals aligns with increasingly stringent global regulations on chemical manufacturing. The simplified workup generates less hazardous waste, lowering disposal costs and improving the overall environmental footprint of the production facility. This compliance advantage is critical for maintaining operating licenses and meeting the sustainability goals of major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers. Understanding these details helps in evaluating the feasibility of adopting this route for specific project requirements and ensures alignment with quality and regulatory standards.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method utilizes inexpensive elemental sulfur and readily available phenylacetic acids, avoiding the need for pre-synthesized thioamides or hazardous reagents like lithium tert-butoxide found in prior art.
Q: What is the typical yield range for these 1,2,4-thiadiazole derivatives?
A: According to the patent data, yields vary by substrate but generally range from moderate to high, with specific examples achieving up to 82% yield under optimized conditions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses simple operational steps, common solvents like DMSO, and avoids highly toxic reagents, making it highly amenable to commercial scale-up and regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Thiadiazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN111018807B for accelerating drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory chemistry can be seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle copper-catalyzed reactions and sulfur chemistry with the highest safety standards, while our rigorous QC labs enforce stringent purity specifications to meet the demanding requirements of the global pharmaceutical market. We are committed to delivering high-quality intermediates that empower our clients to bring life-saving therapies to patients faster.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and target specifications. Please contact us to request specific COA data for our existing thiadiazole library or to discuss route feasibility assessments for your custom molecules. Together, we can optimize your supply chain and achieve superior commercial outcomes through scientific excellence and operational efficiency.
