Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Pharmaceutical Applications
Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Pharmaceutical Applications
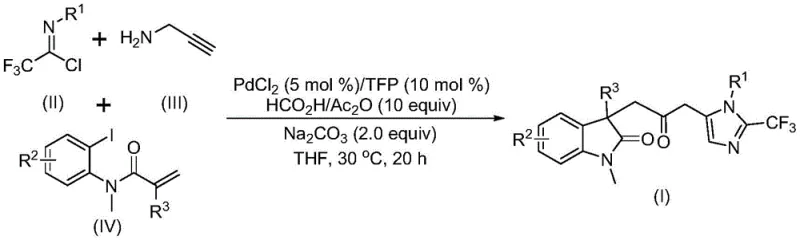
The pharmaceutical and fine chemical industries are constantly seeking more efficient, safer, and cost-effective pathways to construct complex molecular scaffolds, particularly those containing biheterocyclic motifs which are prevalent in bioactive molecules. A significant breakthrough in this domain is documented in Chinese Patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. This technology represents a paradigm shift from traditional step-wise syntheses to a streamlined, one-pot cascade reaction catalyzed by transition metals. By integrating indolinone and imidazole frameworks through a carbonyl bridge, this method provides access to diversified structures that are highly valuable for medicinal chemistry campaigns. The core innovation lies in the ability to assemble these complex architectures from simple, commercially available precursors—specifically trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives—under remarkably mild conditions. For R&D directors and procurement specialists alike, this patent offers a compelling solution to the challenges of building nitrogen-rich heterocyclic libraries with high atom economy and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems has relied on laborious multi-step sequences that often suffer from low overall yields and poor functional group tolerance. Traditional approaches typically involve the direct coupling of two pre-formed heterocyclic substrates, which requires the prior synthesis of both distinct ring systems, thereby increasing material costs and waste generation. Alternatively, oxidative cyclization reactions involving bis-nucleophiles and activated methyl-substituted heterocycles often necessitate harsh oxidants and elevated temperatures, leading to significant safety hazards and difficult purification processes due to the formation of complex byproduct mixtures. Furthermore, when carbonyl bridges are required, classical carbonylation protocols frequently mandate the use of toxic carbon monoxide gas stored in high-pressure cylinders, imposing stringent regulatory burdens and requiring specialized, expensive reactor infrastructure that many contract manufacturing organizations lack. These limitations create bottlenecks in the supply chain, extending lead times for high-purity pharmaceutical intermediates and inflating the cost of goods sold for downstream API manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN115353511A introduces a sophisticated yet operationally simple palladium-catalyzed carbonylation cascade that constructs the biheterocyclic core in a single pot. This innovative route eliminates the need for external carbon monoxide gas by utilizing an in situ generation system comprising formic acid and acetic anhydride, which safely releases CO under mild thermal conditions. The reaction proceeds efficiently at a low temperature of 30°C, drastically reducing energy consumption compared to high-temperature reflux protocols. By employing a multi-component strategy, the method simultaneously forms multiple chemical bonds—including C-C, C-N, and C-O linkages—thereby maximizing step economy. The use of cheap and readily available starting materials such as propargylamine and acrylamide derivatives ensures that the raw material costs remain low, while the broad substrate scope allows for the rapid generation of diverse analogues for structure-activity relationship (SAR) studies without the need for extensive process re-optimization.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, involving a intricate sequence of oxidative addition, migratory insertion, and cyclization events orchestrated by a palladium center. The cycle likely initiates with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive aryl-palladium(II) intermediate. This species subsequently undergoes an intramolecular Heck-type reaction with the pendant alkene moiety to form a cyclic alkyl-palladium intermediate, establishing the indolinone core structure. Crucially, the presence of the formic acid and acetic anhydride mixture facilitates the release of carbon monoxide, which then inserts into the palladium-carbon bond to yield an acyl-palladium species. Concurrently, the trifluoroethylimidoyl chloride and propargylamine undergo a base-promoted intermolecular reaction to form a trifluoroacetamidine intermediate, which subsequently isomerizes. The final stage involves the activation of this amidine by the acyl-palladium complex, triggering an intramolecular cyclization that closes the imidazole ring and releases the final carbonyl-bridged biheterocyclic product while regenerating the active palladium catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over step-wise syntheses. Because the reactive intermediates are generated and consumed within the same reaction vessel, the accumulation of unstable or hazardous intermediates is minimized, leading to a cleaner crude reaction profile. The use of tri-furylphosphine (TFP) as a ligand plays a critical role in stabilizing the palladium center and modulating its electronic properties to favor the desired carbonylation pathway over competing side reactions such as homocoupling or beta-hydride elimination. Furthermore, the mild basic conditions provided by sodium carbonate ensure that acid-sensitive functional groups on the substrate remain intact, thereby preserving the integrity of complex molecular architectures. This high level of chemoselectivity translates directly to simplified downstream processing, as fewer byproducts mean less rigorous chromatographic purification is required to achieve the stringent purity specifications demanded by the pharmaceutical industry.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, precise control over stoichiometry and reaction parameters is essential to maximize yield and reproducibility. The patent outlines a robust protocol where the molar ratio of trifluoroethylimidoyl chloride to propargylamine to acrylamide is optimized to ensure complete conversion while minimizing excess reagent waste. The detailed standardized synthesis steps below provide a clear roadmap for executing this transformation, ensuring that technical teams can replicate the high efficiencies reported in the patent data.
- Combine palladium chloride catalyst, TFP ligand, sodium carbonate base, and the formic acid/acetic anhydride CO source mixture in an organic solvent such as THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative into the reaction vessel under stirring.
- Maintain the reaction mixture at 30°C for 12 to 20 hours, followed by filtration, silica gel treatment, and column chromatography purification to isolate the final biheterocyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology presents a strategic opportunity to optimize the sourcing of complex heterocyclic intermediates. The shift from hazardous gas-based carbonylation to a liquid-phase CO source fundamentally alters the risk profile of the manufacturing process, allowing for production in facilities that may not be equipped for high-pressure gas handling. This flexibility expands the pool of potential contract manufacturing partners, thereby enhancing supply chain resilience and reducing the risk of single-source dependency. Moreover, the use of commodity chemicals like propargylamine and acrylamide derivatives ensures that raw material availability is stable, shielding the project from the volatility often associated with specialized custom building blocks.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive and dangerous carbon monoxide infrastructure, which significantly lowers capital expenditure requirements for production facilities. By avoiding the need for high-pressure autoclaves and specialized gas scrubbing systems, manufacturers can achieve substantial cost savings in both equipment investment and ongoing maintenance. Additionally, the one-pot nature of the reaction reduces the number of isolation and purification steps, leading to lower solvent consumption and reduced labor costs per kilogram of product. The high atom economy of the multi-component reaction further contributes to cost efficiency by maximizing the incorporation of starting materials into the final product, minimizing waste disposal fees.
- Enhanced Supply Chain Reliability: The reliance on widely available, off-the-shelf reagents such as palladium chloride, sodium carbonate, and common organic solvents like THF ensures that the supply chain is robust against disruptions. Unlike processes that depend on bespoke catalysts or unstable intermediates with short shelf lives, this method utilizes stable components that can be stocked in bulk, facilitating just-in-time manufacturing strategies. The mild reaction conditions also reduce the likelihood of thermal runaways or equipment failures, ensuring consistent batch-to-batch quality and reliable delivery schedules for downstream customers who depend on timely API production.
- Scalability and Environmental Compliance: The patent data confirms that the reaction has been successfully scaled to the gram level with maintained efficiency, suggesting a straightforward path to kilogram and tonne-scale production. The absence of toxic gas emissions aligns with increasingly stringent environmental regulations, simplifying the permitting process for new manufacturing lines. Furthermore, the simplified workup procedure involving filtration and standard column chromatography reduces the volume of hazardous waste generated, supporting corporate sustainability goals and reducing the environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this technology for their specific applications.
Q: What are the safety advantages of this carbonylation method compared to traditional techniques?
A: Unlike conventional carbonylation reactions that require toxic and hazardous carbon monoxide gas under high pressure, this patented method utilizes a safe formic acid and acetic anhydride mixture to generate carbon monoxide in situ, significantly enhancing operational safety and reducing infrastructure requirements.
Q: Does this synthesis method support a wide range of substrate functional groups?
A: Yes, the process demonstrates excellent substrate compatibility, tolerating various substituents such as halogens, alkyl groups, alkoxy groups, nitro groups, and trifluoromethyl groups on the aromatic rings, allowing for the diverse design of biheterocyclic libraries for drug discovery.
Q: Is this synthetic route suitable for large-scale industrial production?
A: The patent explicitly confirms that the method has been successfully expanded to gram-scale reactions with high efficiency, indicating strong potential for commercial scale-up due to the use of cheap, readily available starting materials and mild reaction conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to accelerate drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We understand that the successful commercialization of a new therapeutic candidate depends on a supply chain that is both agile and robust, and we are committed to delivering the reliability you need.
We invite you to engage with our technical procurement team to discuss how this innovative carbonylation technology can be integrated into your specific project pipeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this greener, more efficient synthetic route. We encourage you to contact us today to obtain specific COA data for related intermediates and to schedule a consultation regarding route feasibility assessments tailored to your unique molecular targets.
