Advanced Phloroglucinol Manufacturing: Overcoming Thermal Risks in Large-Scale Demethylation
Introduction to Patent CN106866378B
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical active pharmaceutical ingredients and their precursors, particularly for antispasmodic agents like phloroglucinol. A significant technological advancement in this domain is detailed in patent CN106866378B, which outlines a refined synthetic process for phloroglucinol starting from 1,3,5-trimethoxybenzene. This patent addresses long-standing challenges in the demethylation of aromatic ethers, specifically focusing on thermal management and impurity profiles during the hydrolysis stage. By utilizing aluminum trichloride as a complexing reagent in chlorobenzene, the disclosed method achieves a delicate balance between reaction kinetics and safety, offering a viable alternative to traditional bromination-heavy routes. For a reliable phloroglucinol supplier, understanding these mechanistic nuances is essential for ensuring consistent supply chain continuity and meeting the stringent purity requirements of global regulatory bodies.
The core innovation lies in the specific sequence of operations during the workup phase, where the reaction mixture is added to a cooled inorganic acid solution rather than the reverse. This counter-intuitive approach mitigates the violent heat release typically associated with Lewis acid complex dissociation, thereby preventing the formation of intractable solid masses that hinder stirring and heat transfer. Furthermore, the patent emphasizes the utility of a mixed solvent system for recrystallization, leveraging the principles of solubility to strip away both polar and non-polar contaminants. This dual focus on process safety and purification efficiency positions the technology as a cornerstone for cost reduction in pharmaceutical intermediates manufacturing, as it minimizes batch failures and reduces the need for extensive downstream processing.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phloroglucinol has been plagued by hazardous reagents and inefficient multi-step sequences that complicate scale-up. One prominent legacy method, illustrated in the reaction scheme below, relies on the bromination of aniline to form 2,4,6-tribromoaniline, followed by diazotization and subsequent substitution to generate 1,3,5-tribromobenzene. This intermediate is then converted to 1,3,5-trimethoxybenzene using expensive cuprous iodide catalysts in mixed solvents like methanol and DMF. The reliance on stoichiometric amounts of bromine introduces significant safety risks and corrosion issues, while the use of cuprous iodide necessitates rigorous metal scavenging steps to meet residual metal specifications. Moreover, the post-treatment of the substitution reaction is notoriously complex, often requiring extensive washing and separation protocols that drive up operational expenditures and extend production lead times.
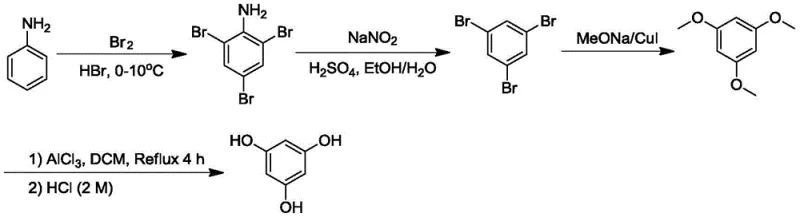
Another conventional approach involves the direct demethylation of 1,3,5-trimethoxybenzene using Lewis acids in low-boiling solvents such as dichloromethane or carbon tetrachloride. While conceptually simpler, this method suffers from sluggish reaction kinetics due to the limited temperature range imposed by the solvent's boiling point. As depicted in prior art diagrams, the hydrolysis step in these processes often leads to uncontrolled exotherms when inorganic acid is added directly to the reaction mass. This rapid heat evolution causes the immediate precipitation of large amounts of solid material, creating a slurry that is difficult to stir and cool effectively. The resulting local hot spots promote the formation of degradation byproducts and tars, severely compromising the yield and purity of the final high-purity phloroglucinol.
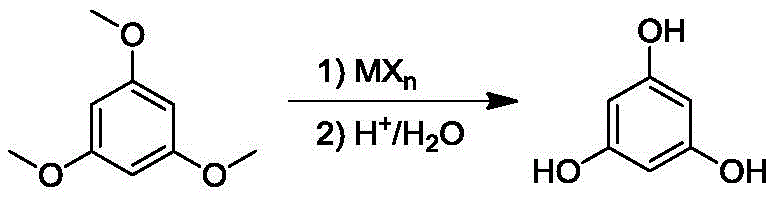
The Novel Approach
The methodology described in CN106866378B represents a paradigm shift by optimizing both the reaction medium and the quenching protocol. By employing chlorobenzene as the solvent, the process operates at elevated temperatures between 80°C and 100°C, which dramatically accelerates the complexation between 1,3,5-trimethoxybenzene and aluminum trichloride. This high-temperature regime ensures near-complete conversion of the starting material, reducing the burden on purification systems. Crucially, the patent dictates a reversed addition order for the hydrolysis step: the cooled reaction system is slowly introduced into a pre-chilled inorganic acid aqueous solution maintained at 0-5°C. This strategy ensures that the heat of dissociation is absorbed by the large thermal mass of the aqueous acid, maintaining a uniform temperature profile throughout the vessel and facilitating the formation of free-flowing granular solids rather than agglomerated chunks.
Mechanistic Insights into AlCl3-Catalyzed Demethylation
The chemical transformation underpinning this process is a classic Lewis acid-mediated ether cleavage, yet the execution details reveal sophisticated process engineering. Aluminum trichloride acts as a potent electrophile, coordinating with the oxygen atoms of the methoxy groups on the 1,3,5-trimethoxybenzene ring. This coordination weakens the carbon-oxygen bond, rendering the methyl group susceptible to nucleophilic attack or elimination upon exposure to protons during the hydrolysis phase. The use of a molar ratio of 1,3,5-trimethoxybenzene to aluminum trichloride ranging from 1:1 to 1:4 ensures that sufficient Lewis acid is available to complex all three methoxy groups simultaneously, driving the equilibrium towards the fully demethylated intermediate. The evolution of gas observed during the heating phase indicates the release of methyl chloride or related volatile byproducts, which are safely scrubbed using alkaline water, preventing pressure buildup and environmental release.
Impurity control is meticulously managed through the thermodynamics of the workup and crystallization stages. The patent highlights that single-solvent recrystallization, typically using water, is insufficient because it fails to remove fat-soluble organic impurities that co-precipitate with the product. By introducing an alcohol component, such as ethanol or methanol, into the recrystallization solvent system, the polarity of the medium is tuned to solubilize these hydrophobic contaminants while keeping the highly polar phloroglucinol in the crystal lattice upon cooling. The activation energy for nucleation is carefully managed by heating the mixture to reflux with activated carbon, which adsorbs colored impurities and polymeric tars, followed by hot filtration to remove the carbon and any insoluble particulates. This results in a crystalline product with a purity exceeding 99.5%, as confirmed by NMR analysis showing characteristic singlets at 5.67 ppm and 9.00 ppm.
How to Synthesize Phloroglucinol Efficiently
The synthesis of phloroglucinol via this optimized route requires precise adherence to temperature gradients and addition rates to maximize yield and safety. The process begins with the dissolution of the starting material in chlorobenzene, followed by the controlled addition of aluminum trichloride and heating to initiate the complexation. Once the reaction is complete, the system must be cooled significantly before the hydrolysis step to prevent thermal runaway. The subsequent recrystallization from an alcohol-water mixture is the final critical control point for achieving pharmaceutical-grade quality. For a detailed breakdown of the specific operating parameters, including exact molar ratios and time intervals, please refer to the standardized synthesis guide below.
- Dissolve 1,3,5-trimethoxybenzene in chlorobenzene and react with aluminum trichloride at 80-100°C for 0.5-6 hours.
- Cool the reaction mixture to 0-5°C and slowly add it into a cooled inorganic acid aqueous solution to precipitate the crude product.
- Recrystallize the crude solid using an ethanol-water mixed solvent with activated carbon treatment to obtain phloroglucinol dihydrate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits that extend beyond mere chemical yield. The elimination of bromine and cuprous iodide from the process flow removes two major cost drivers and supply chain bottlenecks. Bromine is a hazardous material requiring specialized storage and handling infrastructure, while cuprous iodide is a precious metal catalyst subject to market volatility and stringent recovery mandates. By replacing these with commodity chemicals like aluminum trichloride and chlorobenzene, manufacturers can achieve significant cost savings in raw material procurement and waste disposal. Furthermore, the simplified workflow reduces the number of unit operations, directly translating to lower labor costs and reduced equipment occupancy time, which enhances overall plant throughput.
- Cost Reduction in Manufacturing: The transition to a high-boiling solvent system eliminates the need for cryogenic conditions often required in low-temperature bromination or diazotization reactions, thereby reducing energy consumption for refrigeration. Additionally, the improved conversion rates mean that less starting material is wasted, and the solvent, chlorobenzene, can be efficiently recovered and recycled due to its immiscibility with the aqueous acid phase. This closed-loop solvent management significantly lowers the variable cost per kilogram of production, making the process economically attractive for commercial scale-up of complex phenolic compounds.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to higher batch success rates, minimizing the risk of production delays caused by off-spec material. The use of widely available industrial solvents and reagents ensures that the supply chain is not vulnerable to the shortages that often affect specialized catalysts or halogenating agents. This reliability is crucial for maintaining continuous supply to downstream customers who depend on just-in-time delivery models for their own API manufacturing schedules, effectively reducing lead time for high-purity phloroglucinol.
- Scalability and Environmental Compliance: The controlled exotherm management inherent in the reversed addition protocol makes this process inherently safer for large-scale reactors, where heat dissipation is a primary engineering constraint. The ability to handle the reaction without violent temperature spikes reduces the risk of reactor over-pressurization and containment breaches. Moreover, the process generates less hazardous waste compared to bromine-based routes, simplifying effluent treatment and ensuring compliance with increasingly strict environmental regulations regarding halogenated organic waste and heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this phloroglucinol synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational boundaries and quality outcomes. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into existing manufacturing facilities.
Q: Why is chlorobenzene preferred over dichloromethane in this synthesis?
A: Chlorobenzene has a higher boiling point, allowing the reaction to proceed at elevated temperatures (80-100°C), which significantly increases the reaction rate and conversion compared to low-boiling solvents like dichloromethane.
Q: How does the new process control the exothermic hydrolysis step?
A: Instead of adding acid to the reaction mix, the process involves adding the cooled reaction system into a pre-cooled inorganic acid solution (0-5°C). This reverses the heat generation dynamic, preventing local overheating and solid clumping.
Q: What is the advantage of using an alcohol-water mixed solvent for recrystallization?
A: A mixed solvent system effectively removes both water-soluble and fat-soluble impurities simultaneously, resulting in a final product purity exceeding 99.5%, which is superior to using water alone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phloroglucinol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the AlCl3-catalyzed demethylation process are fully realized in a GMP-compliant environment. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of phloroglucinol meets the exacting standards required for pharmaceutical applications, including residual solvent analysis and heavy metal testing.
We invite potential partners to engage with our technical team to explore how this optimized synthesis route can enhance your supply chain resilience and cost structure. By requesting a Customized Cost-Saving Analysis, you can gain a detailed projection of the economic impact of switching to this methodology. We encourage you to contact our technical procurement team today to索取 specific COA data and discuss route feasibility assessments tailored to your specific volume requirements and quality targets.
