Advanced Palladium-Catalyzed Synthesis of Arylacetamides for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct the amide bond, a ubiquitous motif in bioactive molecules. A significant breakthrough in this domain is detailed in Chinese patent CN111978194A, which discloses a novel preparation method for arylacetamide compounds. This technology represents a paradigm shift from traditional, hazardous carbonylation techniques to a safer, more efficient catalytic system. By utilizing benzyl formate as a dual-purpose reagent—serving as both the carbon monoxide source and the alkyl reactant—this process circumvents the logistical and safety nightmares associated with handling toxic CO gas. Furthermore, the method demonstrates exceptional versatility in activating tertiary amines, a class of substrates historically resistant to direct carbonylation without harsh oxidants. For R&D directors and procurement strategists alike, this patent offers a compelling pathway to high-purity arylacetamide intermediates with a drastically simplified supply chain profile.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of arylacetamides has relied heavily on the direct amidation of phenylacetic acid derivatives or transition metal-catalyzed carbonylation using gaseous carbon monoxide. The former often necessitates aggressive activation agents and generates substantial salt waste, complicating downstream purification and environmental compliance. The latter, while atom-economical in theory, poses severe safety risks due to the requirement for high-pressure CO cylinders and frequently employs explosive CO/O2 mixtures to regenerate the active catalyst. Moreover, a persistent challenge in amide chemistry has been the functionalization of tertiary amines. The strength of the C-N bond in trialkylamines typically renders them inert to standard carbonylation conditions, forcing chemists to resort to multi-step sequences involving dealkylation or the use of primary/secondary amines followed by alkylation. These conventional routes are not only costly but also introduce significant variability in impurity profiles, posing risks to the consistency required for pharmaceutical intermediates.
The Novel Approach
The methodology outlined in patent CN111978194A elegantly resolves these bottlenecks through a palladium-catalyzed system that operates under remarkably practical conditions. By employing benzyl formate, the process utilizes a stable, liquid surrogate that releases carbon monoxide in situ, thereby eliminating the need for specialized high-pressure gas infrastructure. The reaction proceeds efficiently at 130°C in acetonitrile, utilizing a catalyst system composed of palladium acetate and the bidentate ligand DPEphos. Most critically, this approach achieves the cleavage of the C-N bond in tertiary amines without the addition of external oxidants, a feat that significantly streamlines the reaction matrix. This innovation allows for the direct conversion of readily available tertiary amines and benzyl formates into complex arylacetamides with high atom economy and operational simplicity.
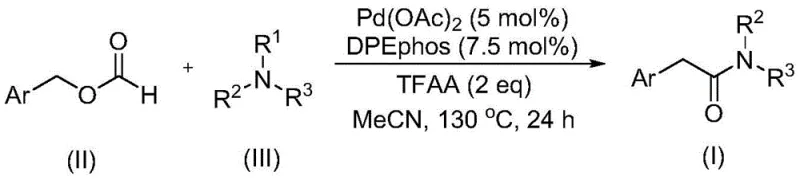
Mechanistic Insights into Pd-Catalyzed Carbonylative C-N Activation
From a mechanistic perspective, this transformation is a sophisticated example of transition metal catalysis designed to overcome thermodynamic barriers. The catalytic cycle likely initiates with the oxidative addition of the palladium species into the benzyl formate, or potentially the decarbonylation of the formate to generate a reactive Pd-CO species and a benzyl fragment. The presence of trifluoroacetic anhydride (TFAA) plays a crucial role, likely acting as a dehydrating agent or activator that facilitates the formation of the active acyl-palladium intermediate. The unique ability of this system to activate the tertiary amine suggests a mechanism where the palladium center coordinates with the amine nitrogen, weakening the adjacent C-N bond and allowing for selective cleavage and subsequent insertion of the carbonyl moiety. This avoids the radical pathways often associated with oxidative C-N cleavage, resulting in a cleaner reaction profile with fewer byproducts.
For the R&D team, understanding the impurity control mechanism is vital. The specificity of the DPEphos ligand ensures that the catalytic cycle remains tightly controlled, minimizing homocoupling of the benzyl fragments or over-carbonylation. The absence of external oxidants means that sensitive functional groups on the aromatic ring—such as halogens, nitriles, or trifluoromethyl groups—are preserved without side reactions like oxidation or hydrolysis. This functional group tolerance is essential for synthesizing diverse libraries of agrochemical intermediates and drug candidates where specific substitution patterns are required for biological activity. The result is a crude reaction mixture that is significantly cleaner than those produced by traditional methods, reducing the burden on purification resources.
How to Synthesize Arylacetamide Efficiently
The operational protocol for this synthesis is designed for scalability and ease of execution in a standard laboratory or pilot plant setting. The process begins by charging a reaction vessel with the palladium catalyst, ligand, benzyl formate derivative, tertiary amine, and TFAA in an aprotic solvent like acetonitrile. The detailed standardized synthesis steps see the guide below.
- Combine palladium catalyst (Pd(OAc)2), ligand (DPEphos), benzyl formate, tertiary amine, and trifluoroacetic anhydride (TFAA) in an organic solvent such as acetonitrile.
- Heat the reaction mixture to approximately 130°C in a sealed vessel and maintain stirring for 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the high-purity arylacetamide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic advantages beyond mere chemical yield. The shift from gaseous CO to liquid benzyl formate fundamentally alters the risk profile of the manufacturing site, removing the need for expensive gas handling systems and reducing insurance premiums associated with toxic gas storage. Furthermore, the starting materials—benzyl formates and common tertiary amines like triethylamine—are commodity chemicals available from multiple global suppliers, ensuring a resilient supply chain that is not dependent on single-source specialty gases.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of hazardous reagents and the simplification of the workflow. By removing the need for high-pressure reactors and external oxidants, the capital expenditure (CAPEX) for equipment is significantly lowered. Additionally, the use of cheap, bulk-available benzyl formate as a CO source replaces the logistical costs associated with purchasing and transporting compressed carbon monoxide. The high efficiency of the catalyst system means that precious metal loading is minimized, further driving down the variable cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of robust, shelf-stable reagents. Unlike CO gas, which requires specialized logistics and has limited supplier availability in certain regions, benzyl formate and tertiary amines are standard inventory items for most chemical distributors. This ubiquity reduces lead times and mitigates the risk of production stoppages due to raw material shortages. The ability to source these inputs locally in major chemical hubs ensures that production schedules can be maintained even during global logistics disruptions.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage is straightforward due to the homogeneous nature of the catalytic system and the absence of gas-liquid mass transfer limitations typical of CO carbonylation. From an environmental standpoint, the process generates less hazardous waste compared to traditional activation methods that produce stoichiometric amounts of coupling byproducts. The simplified workup, often requiring only filtration and chromatography, reduces solvent consumption and energy usage, aligning with modern green chemistry principles and easing the regulatory burden for waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential partners.
Q: What is the primary advantage of using benzyl formate over carbon monoxide gas?
A: Benzyl formate serves as a safe, liquid carbon monoxide surrogate, eliminating the need for high-pressure CO cylinders and explosive CO/O2 mixtures, thereby significantly enhancing operational safety and reducing regulatory compliance costs.
Q: Can this method activate stable tertiary amines?
A: Yes, the patented process uniquely enables the cleavage of the C-N bond in tertiary amines without requiring additional external oxidants, overcoming a major limitation in traditional amide synthesis.
Q: Is this process suitable for large-scale manufacturing?
A: Absolutely. The use of commercially available starting materials, mild reaction conditions, and simplified post-processing makes this method highly scalable for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylacetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN111978194A. As a leading CDMO partner, we possess the technical expertise to translate such innovative laboratory protocols into robust, GMP-compliant manufacturing processes. Our facilities are equipped to handle complex organometallic chemistry, ensuring that the transition from benchtop discovery to commercial production is seamless. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, maintaining stringent purity specifications through our rigorous QC labs to meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the market for high-value amide intermediates.
