Advanced Manufacturing Of High Purity Phloroglucinol For Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously demands higher purity standards for active ingredients and critical intermediates, particularly for spasmolytic agents like phloroglucinol which serve vital roles in treating smooth muscle spasms. Patent CN113429265A introduces a groundbreaking synthesis process that addresses long-standing challenges in producing medicinal high-purity phloroglucinol through a refined demethylation strategy. This innovative approach utilizes boron tribromide as a selective reagent to convert 1,3,5-trimethoxybenzene into the target trihydroxybenzene derivative with exceptional efficiency. By implementing strict temperature controls and a novel quenching protocol, the method achieves an HPLC purity of up to 99.95%, significantly surpassing conventional benchmarks. As a reliable phloroglucinol supplier, understanding these technical nuances is essential for ensuring the quality and safety of downstream drug formulations. The following analysis details how this patented technology transforms the manufacturing landscape for this critical pharmaceutical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of phloroglucinol has relied heavily on aluminum chloride-mediated demethylation or complex multi-step sequences starting from aniline derivatives. These traditional pathways often suffer from severe drawbacks, including the requirement for harsh reaction conditions that promote the formation of stubborn by-products such as gamboge salts. The presence of these impurities necessitates extensive and costly purification steps, often involving multiple extractions and recrystallizations that erode overall yield. Furthermore, the use of strong Lewis acids like aluminum chloride typically generates substantial amounts of waste acid, creating significant environmental disposal burdens and increasing operational costs. The violent heat release associated with these exothermic reactions also poses safety risks during commercial scale-up of complex pharmaceutical intermediates, limiting the ability of manufacturers to increase batch sizes safely. Consequently, many producers struggle to maintain consistent quality while managing the economic pressures of modern drug manufacturing.
The Novel Approach
In stark contrast, the novel process disclosed in the patent utilizes boron tribromide in a dichloromethane solvent system to facilitate a much cleaner and more controllable transformation. This method operates under significantly milder conditions, typically ranging from 0°C to 35°C, which effectively suppresses the formation of thermally induced impurities. The strategic choice of reagents allows for a direct conversion that minimizes side reactions, thereby simplifying the post-treatment workflow considerably. Instead of complex aqueous workups, the product can be isolated through straightforward filtration and pulping, drastically reducing solvent consumption and processing time. This streamlined approach not only enhances the chemical purity of the final product but also aligns with green chemistry principles by reducing waste generation. For procurement teams, this translates to a more robust supply chain capable of delivering high-purity phloroglucinol with greater consistency and reliability.
Mechanistic Insights into Boron Tribromide Catalyzed Demethylation
The core of this technological advancement lies in the precise mechanistic control of the ether cleavage reaction using boron tribromide as a potent Lewis acid. The reaction proceeds through the formation of a coordinate complex between the boron atom and the oxygen of the methoxy groups, which weakens the carbon-oxygen bond and facilitates nucleophilic attack. Maintaining the molar ratio of 1,3,5-trimethoxybenzene to boron tribromide between 1:3 and 1:6 is critical to ensuring complete conversion without excessive reagent waste. The use of dichloromethane as the solvent provides an ideal medium for solubilizing both the organic substrate and the Lewis acid complex while maintaining thermal stability. By keeping the reaction temperature low during the addition phase, the process prevents localized hot spots that could otherwise lead to polymerization or degradation of the sensitive phenolic product. This level of control is paramount for achieving the reported yields of 64.8% to 76.2% with minimal impurity profiles.
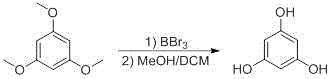
Equally important is the sophisticated quenching protocol designed to manage the exothermic nature of the hydrolysis step. The patent specifies adding the reaction mixture into a pre-cooled solution of methanol and dichloromethane, rather than the reverse, to ensure rapid heat dissipation. This specific order of addition prevents the formation of gelatinous precipitates that can trap impurities and hinder filtration efficiency. The methanol acts as a proton source to finalize the demethylation while the dichloromethane maintains the solubility of intermediate species. Following the quench, the product precipitates as a white solid which is then subjected to a water pulping process to remove residual salts and organic by-products. This meticulous attention to the workup phase ensures that the final crystalline material meets the stringent purity specifications required for pharmaceutical applications without the need for chromatographic purification.
How to Synthesize Phloroglucinol Efficiently
The synthesis of this high-value intermediate requires precise adherence to the patented parameters to replicate the high purity and yield outcomes described. Operators must ensure that all reagents are anhydrous and that temperature monitoring is continuous throughout the addition and reaction phases to prevent thermal runaway. The detailed standardized synthesis steps involve specific cooling rates and stirring speeds that are critical for crystal formation and impurity exclusion. For R&D teams looking to implement this route, understanding the nuances of the quenching solvent ratio is essential for optimizing the physical properties of the isolated solid. The following guide outlines the critical operational parameters derived from the patent examples to assist in technology transfer and process validation.
- Dissolve 1,3,5-trimethoxybenzene in dichloromethane and cool to -10°C before adding boron tribromide solution dropwise.
- Maintain reaction temperature between 0-10°C for 0.5 to 10 hours while absorbing generated gases with alkali water.
- Quench the reaction mixture into a pre-cooled methanol and dichloromethane solution, then filter and pulp to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this boron tribromide-mediated process offers substantial benefits for organizations focused on cost reduction in pharmaceutical intermediates manufacturing. The elimination of heavy metal catalysts like aluminum chloride removes the need for expensive and time-consuming metal scavenging steps, directly lowering the cost of goods sold. Additionally, the simplified workup procedure reduces the consumption of auxiliary solvents and utilities, contributing to a smaller environmental footprint and lower waste disposal fees. The mild reaction conditions also extend the lifespan of reactor equipment by reducing corrosion and thermal stress, leading to decreased maintenance downtime and capital expenditure over time. These operational efficiencies collectively enhance the economic viability of producing phloroglucinol at a commercial scale.
- Cost Reduction in Manufacturing: The process significantly lowers production costs by streamlining the purification workflow and eliminating the need for complex extraction units associated with traditional acid hydrolysis methods. By avoiding the formation of difficult-to-remove gamboge salt impurities, manufacturers save on the resources typically required for repeated recrystallization and chromatography. The higher selectivity of the boron tribromide reagent ensures that raw materials are converted more efficiently into the desired product, minimizing waste and maximizing atom economy. Furthermore, the reduced energy consumption resulting from milder temperature requirements contributes to overall operational savings. These factors combine to create a more competitive pricing structure for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Implementing this robust synthesis route improves supply chain stability by reducing the risk of batch failures due to thermal runaways or impurity spikes. The use of readily available starting materials and reagents ensures that production schedules are not disrupted by sourcing bottlenecks or specialized chemical shortages. The consistency of the product quality reduces the likelihood of regulatory rejection or customer returns, fostering stronger relationships with downstream pharmaceutical partners. Moreover, the scalability of the process allows suppliers to respond more flexibly to fluctuations in market demand without compromising on quality standards. This reliability is crucial for maintaining continuous production lines for essential spasmolytic medications.
- Scalability and Environmental Compliance: The technology is inherently designed for commercial scale-up of complex pharmaceutical intermediates, featuring safety mechanisms that manage exothermic heat release effectively. The reduction in waste acid generation simplifies effluent treatment processes, making it easier for facilities to comply with increasingly strict environmental regulations. The solid-state isolation of the product minimizes solvent losses and facilitates safer handling and packaging operations. By adopting this greener manufacturing approach, companies can enhance their sustainability profiles while meeting the growing demand for eco-friendly pharmaceutical production methods. This alignment with environmental goals adds long-term value to the supply chain beyond immediate financial metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These answers are derived directly from the patent data to provide accurate guidance for stakeholders evaluating this manufacturing route. Understanding these details is key to assessing the feasibility of integrating this process into existing production frameworks. The insights provided here aim to clarify the operational benefits and technical requirements for successful adoption.
Q: How does the BBr3 method improve purity compared to traditional AlCl3 processes?
A: The boron tribromide method operates under milder conditions that prevent the formation of gamboge salt impurities commonly associated with high-temperature aluminum chloride reactions, achieving HPLC purity up to 99.95%.
Q: What are the scalability advantages of this synthesis route?
A: The process utilizes a controlled quenching system that manages exothermic heat release effectively, allowing for safe commercial scale-up of complex pharmaceutical intermediates without violent thermal runaways.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method simplifies post-treatment by eliminating complex extraction units and reducing waste acid generation, making it highly suitable for cost reduction in pharmaceutical intermediates manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phloroglucinol Supplier
At NINGBO INNO PHARMCHEM, we leverage cutting-edge synthetic methodologies like the boron tribromide demethylation process to deliver superior quality intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity phloroglucinol meets the highest industry standards. Our commitment to technical excellence allows us to offer reducing lead time for high-purity pharmaceutical intermediates, enabling our partners to accelerate their own drug development timelines. We understand the critical nature of your supply chain and are dedicated to being a stable and responsive partner in your success.
We invite you to collaborate with us to optimize your sourcing strategy and achieve significant operational efficiencies. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality expectations. Please contact us to request specific COA data and route feasibility assessments that demonstrate how our advanced manufacturing capabilities can support your business goals. By partnering with us, you gain access to a wealth of chemical expertise and a reliable supply network dedicated to your growth.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
