Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazole Intermediates for Global Pharma
Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazole Intermediates for Global Pharma
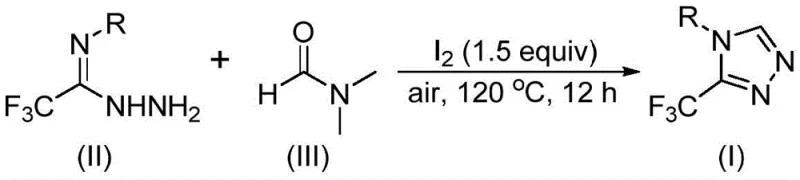
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which are pivotal for enhancing metabolic stability and bioavailability in modern drug candidates. A groundbreaking development in this domain is detailed in patent CN114920707B, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a paradigm shift by utilizing the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source in an iodine-promoted tandem cyclization. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediate supplier options, this patent offers a compelling value proposition by merging synthetic elegance with practical manufacturability. The ability to synthesize these core scaffolds under aerobic conditions without stringent anhydrous requirements addresses critical pain points in process chemistry, paving the way for more sustainable and cost-effective production of high-purity OLED material precursors and agrochemical intermediate variants alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of trifluoromethyl-substituted 1,2,4-triazole rings has relied upon multistep sequences that often involve hazardous reagents, expensive transition metal catalysts, and strictly controlled inert atmospheres. Conventional routes frequently necessitate the use of specialized C1 synthons that are costly to procure and handle, thereby inflating the overall cost reduction in electronic chemical manufacturing and pharmaceutical production. Furthermore, many existing protocols require rigorous exclusion of moisture and oxygen, demanding specialized equipment such as Schlenk lines or gloveboxes which are difficult to justify in large-scale commercial operations. The reliance on heavy metal catalysts also introduces significant downstream purification challenges, as residual metal levels must be reduced to parts-per-million specifications to meet regulatory standards for API intermediates. These factors collectively contribute to extended lead times and increased operational expenditures, creating bottlenecks for supply chain heads aiming for consistent commercial scale-up of complex polymer additives or fine chemical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN114920707B introduces a streamlined one-pot strategy that leverages the dual functionality of DMF to drive the cyclization process efficiently. By employing molecular iodine as a mild promoter, the reaction proceeds smoothly under an air atmosphere at moderate temperatures ranging from 110°C to 130°C, completely eliminating the need for expensive inert gas protection or anhydrous conditions. This approach not only simplifies the operational workflow but also significantly broadens the substrate scope, allowing for the introduction of various functional groups such as halogens, alkyls, and alkoxy groups without compromising yield. The use of DMF as both solvent and reactant means that fewer reagents are required overall, which inherently reduces waste generation and simplifies the post-reaction workup procedure. For procurement managers, this translates to a more resilient supply chain where raw materials are commodity chemicals rather than specialized reagents, ensuring reducing lead time for high-purity pharmaceutical intermediates and enhancing overall project viability.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation is a fascinating example of solvent participation in organic synthesis, where DMF acts as a versatile C1 building block through two potential pathways involving either its formyl or methyl group. In the formyl pathway, the carbonyl carbon of DMF undergoes condensation with the trifluoroethyliminohydrazide to form a hydrazone intermediate, which subsequently cyclizes intramolecularly with the elimination of dimethylamine to yield the target triazole. Alternatively, the N-methyl group can be activated by iodine to form an amine salt, which then participates in nucleophilic addition followed by oxidative aromatization. This dual reactivity underscores the flexibility of the system and highlights the sophisticated chemical design that enables such high efficiency under benign conditions. Understanding these mechanistic nuances is crucial for process chemists aiming to optimize reaction parameters for commercial scale-up of complex heterocycles, as it allows for precise tuning of stoichiometry and temperature to maximize conversion while minimizing byproduct formation.
Control of impurity profiles is another critical aspect where this mechanism offers distinct advantages over traditional metal-catalyzed cross-couplings. Since the reaction does not rely on transition metals like palladium or copper, the risk of metal contamination in the final product is virtually eliminated, simplifying the purification process to standard silica gel chromatography or crystallization. The oxidative aromatization step driven by air oxygen ensures that the final triazole ring is fully aromatic and stable, reducing the likelihood of unstable intermediates persisting into the final isolation stage. For quality control teams, this means a cleaner crude reaction profile and higher confidence in meeting stringent purity specifications required for regulatory filings. The robustness of the iodine promotion system also ensures consistent performance across different batches, providing the reliability needed for long-term supply agreements in the competitive landscape of fine chemical intermediates.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this synthesis effectively, operators should begin by charging a reaction vessel with trifluoroethyliminohydrazide and molecular iodine, followed by the addition of DMF which serves as the bulk solvent. The mixture is then heated to approximately 120°C and stirred for a period of 10 to 15 hours under ambient air pressure, monitoring progress via TLC or HPLC to ensure complete consumption of the starting hydrazide. Upon completion, the reaction mixture is cooled and subjected to a straightforward workup involving filtration to remove inorganic salts, followed by concentration and purification via column chromatography to isolate the pure triazole derivative. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Combine trifluoroethyliminohydrazide and molecular iodine in DMF solvent within a reaction vessel open to air.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for 10 to 15 hours to ensure complete conversion.
- Perform post-treatment involving filtration, washing, and silica gel chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial benefits that directly impact the bottom line for manufacturing organizations seeking cost reduction in API manufacturing. The substitution of expensive, specialized reagents with commodity solvents like DMF drastically lowers the raw material cost per kilogram, while the elimination of inert atmosphere requirements reduces capital expenditure on specialized reactor hardware. Additionally, the simplified workup procedure minimizes solvent consumption and waste disposal costs, contributing to a more sustainable and economically viable production model that aligns with modern green chemistry principles. These factors combined create a compelling economic case for adopting this technology over legacy methods, particularly for high-volume production runs where marginal savings translate into significant total cost avoidance.
- Cost Reduction in Manufacturing: The utilization of DMF as both solvent and carbon source eliminates the need for purchasing separate C1 synthons, which are often priced at a premium due to their specialized nature and handling requirements. Furthermore, the use of molecular iodine as a promoter is significantly more economical than noble metal catalysts, and the absence of metal residues removes the need for costly scavenging resins or extensive purification steps. This holistic reduction in reagent and processing costs ensures that the overall manufacturing expense is significantly reduced without compromising on the quality or purity of the final pharmaceutical intermediate product.
- Enhanced Supply Chain Reliability: All starting materials involved in this process, including DMF, iodine, and the hydrazide precursors, are commercially available in bulk quantities from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate air and moisture, means that production schedules are less susceptible to delays caused by environmental control failures or equipment maintenance issues associated with glovebox operations. This reliability is crucial for supply chain heads who need to guarantee consistent delivery timelines to downstream customers in the competitive pharmaceutical and agrochemical markets.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures and atmospheric pressure, making it inherently safer and easier to scale from laboratory benchtop to multi-ton industrial reactors without significant engineering modifications. The absence of toxic heavy metals and the use of common solvents simplify waste stream management and regulatory compliance, reducing the environmental footprint of the manufacturing process. This ease of scale-up ensures that the commercial scale-up of complex heterocycles can be achieved rapidly to meet market demand, providing a strategic advantage in time-to-market for new drug candidates incorporating this trifluoromethyl triazole scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and strategic benefits for stakeholders evaluating this route for their own production pipelines. Understanding these details is essential for making informed decisions about technology transfer and process adoption.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source, eliminating the need for additional expensive C1 building blocks and simplifying the workup process significantly.
Q: Does this method require inert gas protection?
A: No, the reaction proceeds efficiently under an air atmosphere, which drastically reduces operational complexity and equipment costs compared to traditional anaerobic methods.
Q: What is the substrate scope for this reaction?
A: The method tolerates a wide range of substituents on the aromatic ring, including halogens, alkyl groups, and electron-withdrawing groups, allowing for diverse derivative synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this DMF-mediated synthesis route for producing high-value trifluoromethyl triazole intermediates used in next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from gram-scale optimization to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole compound meets the exacting standards required by global regulatory bodies for pharmaceutical applications.
We invite you to collaborate with our technical procurement team to explore how this innovative chemistry can optimize your supply chain and reduce overall project costs. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments for your target molecules. Our experts are ready to assist you in navigating the complexities of fine chemical synthesis to secure a reliable and competitive supply of critical intermediates for your drug development programs.
