Advanced Manufacturing of Ticagrelor Intermediates via Optimized Condensation Routes
Advanced Manufacturing of Ticagrelor Intermediates via Optimized Condensation Routes
The pharmaceutical landscape for antithrombotic agents continues to evolve, driven by the demand for safer, more efficient manufacturing processes for critical small molecule drugs. A pivotal development in this sector is detailed in patent CN102875537A, which discloses a novel preparation method for Ticagrelor (ADZ6140), a reversible P2Y12 adenosine diphosphate receptor antagonist. This technology addresses long-standing challenges in the synthesis of this potent anticoagulant, offering a pathway that significantly mitigates side reactions and simplifies purification protocols. For R&D directors and supply chain leaders, understanding this methodology is crucial, as it represents a shift from complex, low-yield legacy routes to a streamlined, industrially viable process. The core innovation lies in the strategic protection of reactive functional groups during the critical condensation phases, ensuring higher purity intermediates and a more robust supply chain for this life-saving medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ticagrelor has been plagued by inherent chemical instabilities in the starting materials used in prior art routes, such as those described in WO 00/34283 and US 2003/0148888. These conventional pathways typically rely on condensing raw materials that possess both active hydroxyl and amino groups simultaneously with highly electrophilic chloropyrimidine derivatives. The presence of strong electron-withdrawing nitro groups on the pyrimidine ring renders the chlorine atoms exceptionally reactive, leading to uncontrolled nucleophilic attacks by the hydroxyl groups alongside the desired amino coupling. This lack of chemoselectivity results in a complex mixture of byproducts that are structurally similar to the target intermediate, making purification extremely difficult and often necessitating resource-intensive column chromatography. Furthermore, these older routes frequently employ hazardous reagents like DIBAL-H for reduction steps, posing significant safety risks and generating complex waste streams that complicate environmental compliance and increase operational costs.
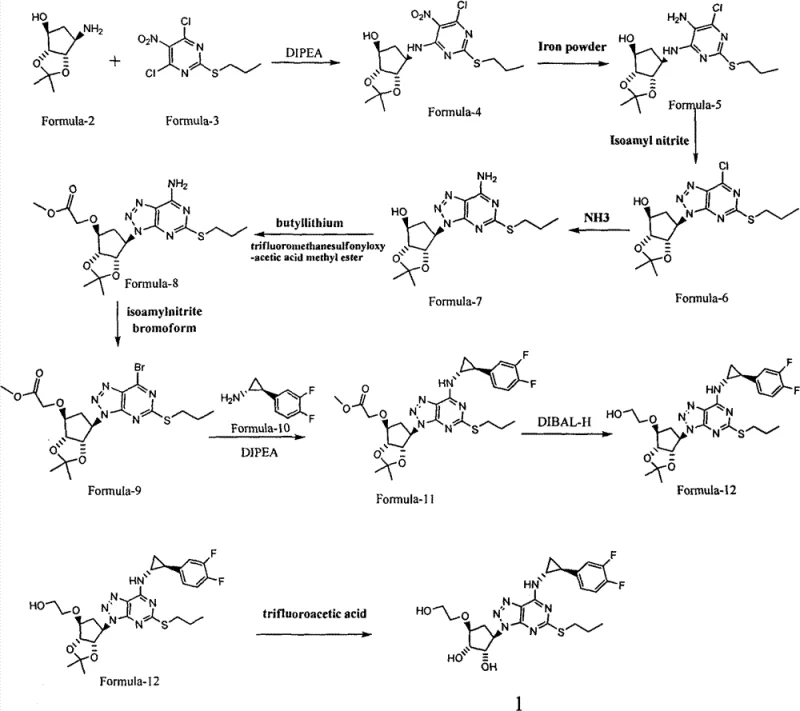
The Novel Approach
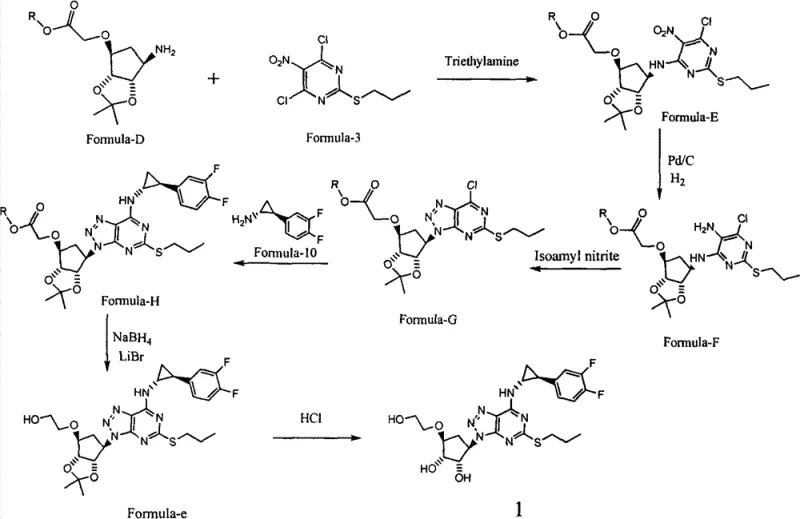
In stark contrast, the methodology presented in CN102875537A introduces a paradigm shift by utilizing a protected intermediate, specifically Formula-D, which features an ester group instead of a free hydroxyl. This strategic modification effectively masks the reactive hydroxyl functionality during the initial condensation with the chloropyrimidine derivative (Formula-3). By preventing the hydroxyl group from participating in nucleophilic substitution, the reaction becomes highly selective for the amino group, drastically reducing the formation of ether-linked byproducts. This enhanced selectivity translates directly into higher crude purity, allowing for purification via simple crystallization or filtration rather than chromatography. Additionally, the route replaces dangerous hydride reagents with a safer sodium borohydride and lithium bromide system for the final reduction, demonstrating a comprehensive optimization of both safety and efficiency that is essential for modern GMP manufacturing environments.
Mechanistic Insights into Chemoselective Condensation and Triazole Formation
The cornerstone of this improved synthesis is the initial condensation reaction between the protected cyclopentyl amine (Formula-D) and the nitro-chloropyrimidine (Formula-3). Mechanistically, the use of organic bases such as diisopropylethylamine (DIPEA) in an inert solvent like tetrahydrofuran (THF) facilitates the deprotonation of the amine, enhancing its nucleophilicity. Crucially, the ester moiety on the cyclopentyl ring remains inert under these mild alkaline conditions (0-10°C), whereas a free hydroxyl group would have competed aggressively for the electrophilic chlorine centers. Following this clean coupling, the nitro group is reduced to an amine using catalytic hydrogenation (Pd/C), a standard yet highly effective transformation that avoids the messy workups associated with metal powder reductions. The subsequent cyclization to form the triazole ring using isoamyl nitrite proceeds smoothly in acetonitrile at elevated temperatures (65-75°C), leveraging the newly formed ortho-diamine motif to close the heterocyclic ring with high fidelity.
Impurity control is further refined in the later stages of the synthesis, particularly during the coupling with the difluorophenyl cyclopropyl amine fragment (Formula-10) and the final deprotection. The patent highlights that the intermediate Formula-G, possessing the fully formed triazolopyrimidine core, reacts cleanly with Formula-10 under basic conditions to displace the remaining chlorine atom. The final conversion of the ester side chain to the requisite hydroxyethyl group is achieved using sodium borohydride mediated by lithium bromide. This specific reagent combination modulates the reducing power of the borohydride, allowing for selective ester reduction without affecting other sensitive functionalities in the complex molecule. This mechanistic precision ensures that the final API, Ticagrelor, is obtained with a purity exceeding 99% after a straightforward acid-catalyzed deprotection and crystallization sequence, effectively eliminating the trace impurities that often plague less optimized routes.
How to Synthesize Ticagrelor Efficiently
The synthesis of Ticagrelor via this novel route involves a sequence of highly controlled chemical transformations designed to maximize yield and minimize waste. The process begins with the preparation of the key protected intermediate, followed by stepwise construction of the heterocyclic core and final functional group manipulation. Each step has been optimized for temperature, stoichiometry, and solvent choice to ensure reproducibility on a large scale. While the general workflow is outlined below, precise adherence to the specific molar ratios and thermal profiles described in the patent is essential for achieving the reported high purity standards. Detailed standardized operating procedures for each reaction stage can be found in the technical documentation provided by our engineering team.
- Condense Formula-D with Formula-3 in THF using DIPEA at 0-10°C to form Formula-E, avoiding hydroxyl side reactions.
- Reduce the nitro group of Formula-E to an amino group using Pd/C hydrogenation to obtain Formula-F.
- React Formula-F with isoamyl nitrite in acetonitrile at 65-75°C to cyclize and form the triazole ring (Formula-G).
- Couple Formula-G with Formula-10, followed by ester reduction using NaBH4/LiBr and final acid deprotection to yield Ticagrelor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification workflow. By eliminating the need for column chromatography—a technique that is notoriously difficult to scale and expensive to operate in terms of silica gel and solvent consumption—the manufacturing process becomes significantly more cost-effective. The ability to purify intermediates through crystallization and filtration not only reduces direct material costs but also shortens the batch cycle time, allowing for faster throughput and improved responsiveness to market demand fluctuations. This efficiency gain is critical for maintaining a steady supply of high-purity pharmaceutical intermediates in a competitive global market.
- Cost Reduction in Manufacturing: The replacement of hazardous and expensive reagents like DIBAL-H with safer, commodity chemicals such as sodium borohydride and lithium bromide leads to substantial cost savings. Furthermore, the increased chemoselectivity of the protected intermediate route minimizes the loss of valuable starting materials to side reactions, thereby improving the overall mass balance and reducing the cost of goods sold (COGS). The elimination of chromatographic purification steps further drives down operational expenses by reducing solvent usage and waste disposal costs associated with silica gel treatment.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing the risk of batch failures due to purification bottlenecks. The use of stable, commercially available reagents and solvents ensures that production is not vulnerable to the supply volatility often associated with specialized or hazardous chemicals. Additionally, the simplified workup procedures allow for more predictable lead times, enabling manufacturers to commit to tighter delivery schedules and maintain optimal inventory levels for their downstream API production partners.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns perfectly with green chemistry principles. The avoidance of heavy metal waste from iron or zinc powder reductions, coupled with the safer handling profile of sodium borohydride compared to pyrophoric hydrides, significantly lowers the environmental footprint of the manufacturing process. This compliance ease facilitates smoother regulatory approvals and reduces the liability associated with handling dangerous goods, making the scale-up from pilot plant to commercial metric ton production a seamless and sustainable endeavor.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ticagrelor synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a transparent view of the process capabilities. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: How does this new synthesis route improve upon previous methods like WO 00/34283?
A: Previous methods utilized raw materials with active hydroxyl and amino groups that caused significant side reactions with the highly active chlorines on the pyrimidine ring. This new route uses an ester-protected intermediate (Formula-D), which stabilizes the reactive sites, drastically reducing byproduct formation and eliminating the need for difficult column chromatography purification.
Q: What safety advantages does the new reduction method offer?
A: Traditional routes often employed DIBAL-H for ester reduction, which is hazardous and generates complex waste streams. The patented method utilizes a sodium borohydride and lithium bromide system, which is significantly safer to handle, easier to quench, and produces less hazardous waste, facilitating smoother industrial scale-up.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is specifically designed for industrial feasibility. By replacing chromatographic purification with crystallization and filtration steps, and by utilizing robust reagents like Pd/C and NaBH4, the route ensures high throughput, consistent purity (>99%), and simplified downstream processing suitable for metric ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ticagrelor Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN102875537A can be executed with precision and consistency. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality assurance means that every gram of Ticagrelor intermediate we supply meets the exacting requirements necessary for final API synthesis.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can drive value and reliability for your organization.
