Advanced C-H Activation Strategy for Scalable Production of High-Purity Catechol Derivatives
Introduction to Next-Generation Catechol Synthesis
The landscape of organic synthesis is continuously evolving, driven by the demand for more efficient and sustainable pathways to access complex molecular architectures. Patent CN108164397B introduces a groundbreaking methodology for the preparation of catechol derivatives, a class of compounds pivotal in medicinal chemistry and crop protection. These ortho-functionalized phenols serve as key structural motifs in numerous biologically active agents, including kinase inhibitors. The disclosed technology leverages a transient directing group strategy that fundamentally alters the efficiency profile of C-H bond functionalization. By utilizing 2-chloro-5-nitropyrimidine as a robust yet removable guiding group, the invention overcomes historical limitations associated with regioselectivity and harsh deprotection conditions. This report analyzes the technical merits of this approach, highlighting its potential to redefine supply chain reliability for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the ortho-functionalization of phenols has been plagued by significant synthetic challenges that hinder industrial adoption. Traditional protocols often rely on the introduction of directing groups that are notoriously difficult to remove without compromising the integrity of the sensitive catechol core. Conventional deprotection strategies frequently necessitate the use of highly reactive alkali metals, such as sodium or potassium, under elevated temperature conditions. These harsh environments not only pose severe safety risks due to the pyrophoric nature of the reagents but also limit the functional group tolerance of the synthesis. Furthermore, the multi-step nature of introducing and subsequently removing these guiding groups typically results in lower overall yields and increased waste generation. For procurement managers, these inefficiencies translate into higher raw material costs and extended lead times, making the reliable sourcing of complex intermediates a persistent bottleneck in drug development pipelines.
The Novel Approach
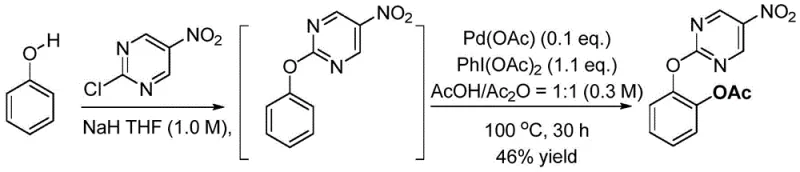
In stark contrast to legacy methods, the technology described in CN108164397B offers a streamlined, three-step sequence that prioritizes operational simplicity and safety. The core innovation lies in the use of a 2-chloro-5-nitropyrimidine moiety, which acts as an effective directing group for palladium-catalyzed C-H activation and can be cleaved under exceptionally mild conditions. The process allows for a telescoped one-pot synthesis for the initial coupling and functionalization steps, drastically reducing solvent consumption and processing time. Crucially, the final deprotection is achieved using hydrazine hydrate in tetrahydrofuran at ambient temperature (25°C), eliminating the need for dangerous alkali metals. This shift not only enhances laboratory safety but also facilitates easier scale-up for commercial production. The ability to tolerate diverse substituents, ranging from electron-donating alkyl groups to electron-withdrawing halogens, underscores the versatility of this route for generating a wide library of analogs.
Mechanistic Insights into Pd-Catalyzed C-H Acetoxylation
The mechanistic elegance of this synthesis rests on the coordination chemistry between the palladium catalyst and the pyrimidine directing group. Initially, the phenolic substrate is deprotonated by sodium hydride to form a nucleophilic phenoxide, which displaces the chloride on the 2-chloro-5-nitropyrimidine to install the directing group. In the subsequent C-H activation step, the nitrogen atoms of the pyrimidine ring coordinate with the palladium(II) species, positioning the metal center in close proximity to the ortho-C-H bond. This pre-organization lowers the activation energy for the C-H cleavage, likely proceeding through a concerted metalation-deprotonation (CMD) pathway or electrophilic palladation. The presence of iodobenzene diacetate (PhI(OAc)2) serves a dual role as both the oxidant and the source of the acetoxy group, facilitating the oxidation of the palladium center to a higher valence state (Pd(IV)) prior to reductive elimination. This sequence ensures high regioselectivity for the ortho-position, minimizing the formation of meta- or para-isomers that would complicate downstream purification efforts.
Impurity control is inherently built into the design of this catalytic cycle, particularly during the final deprotection phase. The use of hydrazine hydrate allows for the selective cleavage of the aryl-pyrimidine ether bond without affecting other sensitive functionalities such as esters or ketones that might be present on the substrate. The reaction proceeds rapidly at 25°C, completing within 30 minutes, which limits the exposure of the product to potentially degradative conditions. This mildness is critical for maintaining the stringent purity specifications required for pharmaceutical intermediates. By avoiding high-temperature acidic or basic hydrolysis, the process prevents common side reactions like polymerization or over-oxidation of the catechol ring. Consequently, the resulting crude product requires less intensive chromatographic purification, leading to improved mass balance and reduced solvent waste, which aligns with modern green chemistry principles.

How to Synthesize Catechol Derivatives Efficiently
Implementing this synthesis route requires careful attention to reaction conditions, particularly regarding moisture sensitivity and stoichiometry. The initial installation of the directing group must be conducted under an inert nitrogen atmosphere using distilled tetrahydrofuran to prevent quenching of the sodium hydride. Following the coupling, the C-H activation step utilizes a mixture of acetic acid and acetic anhydride, which must be degassed to ensure optimal catalyst performance. The final deprotection is straightforward but requires precise monitoring to ensure complete removal of the pyrimidine fragment. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical process controls necessary for reproducible results.
- Install the 2-chloro-5-nitropyrimidine directing group onto the phenol substrate using NaH in THF.
- Perform Pd-catalyzed C-H acetoxylation using PhI(OAc)2 oxidant in AcOH/Ac2O solvent at 100°C.
- Remove the directing group under mild conditions using hydrazine hydrate in THF at 25°C to yield the final catechol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers substantial strategic benefits for organizations managing the supply of fine chemical intermediates. The primary advantage lies in the drastic simplification of the process workflow, which directly correlates to reduced manufacturing costs and enhanced supply chain resilience. By enabling a one-pot procedure for the first two synthetic steps, the method eliminates the need for intermediate isolation and purification, thereby saving significant amounts of solvents and labor hours. This consolidation of steps reduces the physical footprint required for production and minimizes the risk of material loss during transfer operations. Furthermore, the replacement of hazardous alkali metals with benign hydrazine hydrate for deprotection lowers the barrier for regulatory compliance and waste disposal, contributing to a more sustainable manufacturing profile.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents, combined with the reduction in unit operations, leads to significant cost optimization. The use of air-stable palladium acetate and commercially available oxidants ensures that raw material costs remain predictable and manageable. Additionally, the high regioselectivity of the C-H activation minimizes the formation of difficult-to-separate isomers, reducing the burden on purification resources and increasing the overall yield of the desired API intermediate. This efficiency translates into a more competitive pricing structure for the final product without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against varying substrate electronics ensures a consistent supply of diverse catechol derivatives. Since the method tolerates a wide range of functional groups, manufacturers can produce multiple analogs using the same core infrastructure, enhancing flexibility in responding to market demands. The mild reaction conditions reduce the likelihood of batch failures due to thermal runaways or equipment corrosion, ensuring steady production schedules. This reliability is crucial for pharmaceutical clients who require uninterrupted access to high-quality intermediates to maintain their own drug development timelines.
- Scalability and Environmental Compliance: The demonstrated success at the gram level provides a solid foundation for scaling up to kilogram and tonne quantities. The process avoids the use of heavy metal scavengers often required for other transition metal catalyzed reactions, simplifying the downstream processing. Moreover, the reduced solvent usage and avoidance of pyrophoric materials align with increasingly strict environmental regulations, mitigating the risk of future compliance issues. This forward-looking approach ensures long-term viability of the supply chain in a regulatory environment that favors greener and safer chemical processes.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing workflows. The answers provide clarity on safety, scalability, and substrate compatibility, helping stakeholders make informed decisions about adopting this innovative C-H activation strategy for their specific project needs.
Q: What are the advantages of this C-H activation method over traditional ortho-functionalization?
A: Traditional methods often require harsh conditions involving active metals like sodium or potassium at high temperatures for directing group removal. This patent utilizes a mild hydrazine hydrate deprotection at 25°C, significantly improving safety and operational feasibility.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the patent demonstrates a one-pot synthesis capability for the first two steps and has been validated at the gram level. The use of stable catalysts and standard solvents supports potential commercial scale-up.
Q: What types of substrates are compatible with this protocol?
A: The method exhibits broad substrate scope, tolerating various functional groups including alkyl (methyl, t-butyl), halogens (fluoro, chloro, bromo), methoxy, and acetyl groups on the phenol ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Catechol Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to accelerate drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory innovations can be seamlessly translated into industrial reality. We are committed to delivering high-purity catechol derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging the efficiencies of the C-H activation methodology described in CN108164397B, we can offer our partners a distinct competitive advantage in terms of both cost and speed to market.
We invite you to collaborate with us to explore the full potential of this synthetic route for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical intermediates can support your supply chain goals. Together, we can drive innovation and efficiency in the production of next-generation pharmaceutical building blocks.
