Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. Patent CN115353511A introduces a groundbreaking multicomponent method for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This technological advancement addresses critical pain points in modern organic synthesis, particularly the challenges associated with traditional carbonylation reactions that often require hazardous carbon monoxide gas. By leveraging a transition metal palladium-catalyzed cascade reaction, this protocol enables the one-pot assembly of diversified substituted double heterocyclic compounds containing trifluoromethyl and carbonyl groups. The significance of this invention lies not only in its chemical elegance but also in its practical applicability for producing high-purity pharmaceutical intermediates. For R&D directors and process chemists, this represents a viable pathway to access bioactive molecular frameworks that are prevalent in natural products and drug candidates, all while operating under remarkably mild conditions of 30°C.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems has relied on three primary strategies, each fraught with significant limitations that hinder efficient manufacturing. The first approach involves the direct coupling of two pre-formed heterocycle substrates, which often suffers from low atom economy and requires harsh reaction conditions to overcome steric hindrance. The second method utilizes oxidative cyclization of substrates bearing dual nucleophiles with activated methyl-substituted heterocycles, a process that frequently generates substantial waste and demands stoichiometric oxidants. Perhaps most critically, the third conventional route involves transition metal-catalyzed tandem cyclization using external carbon monoxide gas. While effective, the reliance on toxic CO gas presents severe safety hazards, necessitating specialized high-pressure equipment and rigorous safety protocols that drastically increase capital expenditure and operational complexity. Furthermore, controlling the selectivity in these traditional carbonylation reactions remains a persistent challenge, often leading to complex impurity profiles that complicate downstream purification.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a safe and efficient multicomponent reaction strategy. By employing cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, the process achieves high reaction efficiency without the need for toxic carbon monoxide gas. The core innovation lies in the use of a formic acid and acetic anhydride mixture as a safe carbon monoxide substitute, which releases CO in situ under mild conditions. This eliminates the need for high-pressure gas cylinders and allows the reaction to proceed at a gentle 30°C in common organic solvents like tetrahydrofuran (THF). The method exhibits exceptional substrate compatibility, tolerating a wide range of functional groups including halogens, alkyls, and electron-withdrawing groups. This versatility allows for the rapid generation of diverse chemical libraries, making it an ideal tool for medicinal chemistry campaigns aiming to optimize lead compounds.
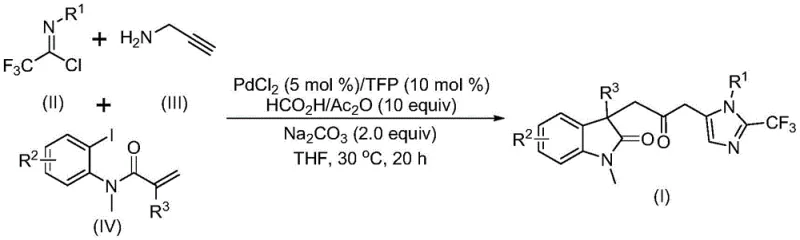
The general reaction scheme illustrates the convergence of three distinct building blocks into a single complex architecture. The use of trifluoroethylimidoyl chloride introduces the valuable trifluoromethyl group, which is known to enhance metabolic stability and lipophilicity in drug molecules. Simultaneously, the acrylamide component facilitates the formation of the indolinone core through an intramolecular cyclization event. This convergent synthesis strategy significantly reduces the number of synthetic steps compared to linear approaches, thereby improving overall yield and reducing waste generation. For procurement managers, the simplicity of this one-pot procedure translates directly into reduced processing time and lower utility costs, as there is no need for cryogenic cooling or high-temperature heating cycles.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams looking to adapt this chemistry for specific API intermediates. The reaction is believed to initiate with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This is followed by an intramolecular Heck reaction, which generates a divalent alkyl-palladium intermediate. Crucially, this intermediate then undergoes a carbonylation step driven by the carbon monoxide released from the decomposition of the formic acid and acetic anhydride mixture. This sequence forms an acyl-palladium intermediate, which serves as the electrophilic partner for the subsequent cyclization. In parallel, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine compound via intermolecular carbon-nitrogen bond formation. This amidine species subsequently undergoes isomerization to become nucleophilic enough to attack the acyl-palladium center.
The final stage of the catalytic cycle involves the activation of the trifluoroacetamidine compound by the acyl-palladium intermediate, triggering an intramolecular cyclization that constructs the imidazole ring and releases the final carbonyl-bridged biheterocyclic product. This intricate dance of bond formations—C-C, C-N, and C-O—occurring in a single pot highlights the power of modern cascade catalysis. The choice of ligand, specifically trifurylphosphine (TFP), plays a pivotal role in stabilizing the palladium center and facilitating the reductive elimination step. Furthermore, the use of sodium carbonate as a base ensures the neutralization of acidic byproducts while promoting the deprotonation steps necessary for amidine formation. This mechanistic clarity allows process chemists to fine-tune reaction parameters, such as catalyst loading (typically 5 mol% PdCl2) and ligand ratio, to maximize conversion and minimize the formation of side products like homocoupling dimers.
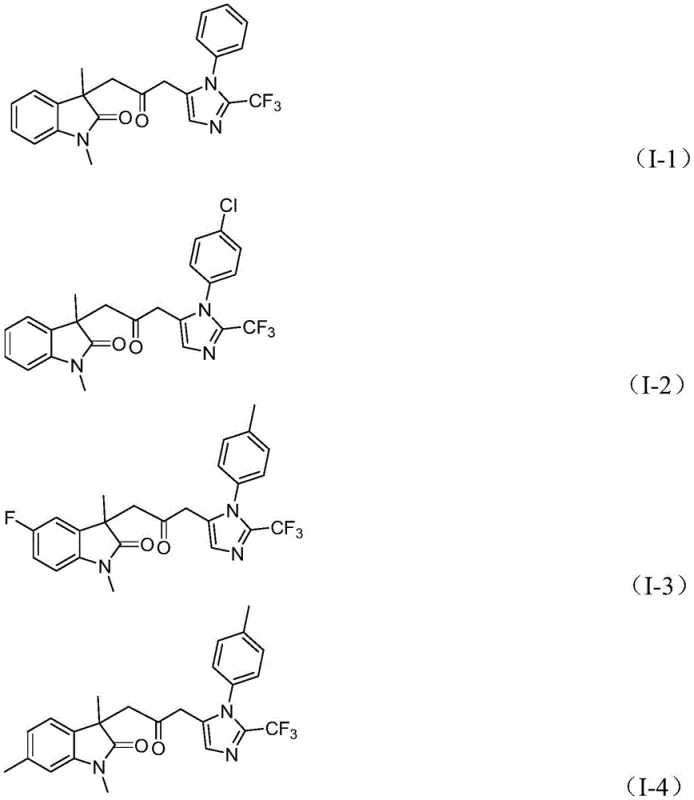
The structural diversity achievable through this mechanism is exemplified by the range of substituents tolerated on the aromatic rings. As shown in the specific examples, variations at the R1, R2, and R3 positions—including phenyl, chloro-phenyl, fluoro-phenyl, and benzyl groups—are accommodated with good to excellent yields. This broad scope confirms that the electronic nature of the substrates does not severely inhibit the catalytic cycle, providing flexibility for designing analogs with specific physicochemical properties. For supply chain heads, this robustness means that minor variations in raw material specifications are less likely to cause batch failures, ensuring consistent supply continuity.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and mixing protocols to ensure optimal performance. The patent outlines a standardized procedure where palladium chloride, trifurylphosphine, sodium carbonate, and the CO-source mixture are combined with the three organic substrates in an aprotic solvent like THF. The reaction is maintained at 30°C for a duration of 12 to 20 hours, allowing sufficient time for the cascade events to reach completion. Post-reaction workup involves simple filtration to remove inorganic salts, followed by silica gel treatment and column chromatography to isolate the pure product. Detailed standard operating procedures regarding specific molar equivalents and purification techniques are essential for reproducibility.
- Combine palladium chloride catalyst, trifurylphosphine ligand, sodium carbonate base, and a mixture of acetic anhydride and formic acid in an organic solvent such as THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the acrylamide derivative to the reaction mixture.
- Stir the reaction at 30°C for 12 to 20 hours, followed by filtration, silica gel treatment, and column chromatography purification to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that align with the strategic goals of cost reduction and supply chain resilience in pharmaceutical manufacturing. The shift away from hazardous reagents and high-pressure equipment fundamentally alters the cost structure of producing these complex intermediates. By utilizing commodity chemicals like formic acid and acetic anhydride instead of specialized gas feeds, manufacturers can significantly lower their raw material procurement costs and reduce the regulatory burden associated with handling toxic substances. Moreover, the mild reaction temperature of 30°C implies a drastic reduction in energy consumption compared to traditional processes that require reflux or cryogenic conditions. This energy efficiency contributes directly to a lower carbon footprint, supporting corporate sustainability initiatives without compromising on output quality.
- Cost Reduction in Manufacturing: The elimination of expensive high-pressure reactors and the associated safety infrastructure results in significant capital expenditure savings. Additionally, the use of inexpensive palladium chloride catalyst and commercially available ligands keeps the catalyst cost low relative to the value of the final product. The high atom economy of the multicomponent reaction means less waste disposal cost, further enhancing the overall economic viability of the process for large-scale production runs.
- Enhanced Supply Chain Reliability: The starting materials, including propargylamine and various acrylamides, are widely available from global chemical suppliers, reducing the risk of supply bottlenecks. The robustness of the reaction against functional group variations means that alternative suppliers for specific substituted anilines or acid chlorides can be qualified more easily. This flexibility ensures that production schedules remain uninterrupted even if a specific raw material vendor faces temporary shortages, thereby securing the continuity of supply for downstream API synthesis.
- Scalability and Environmental Compliance: The protocol has been successfully demonstrated on a gram scale with high efficiency, indicating a clear path toward kilogram and ton-scale manufacturing. The absence of toxic carbon monoxide gas simplifies environmental permitting and workplace safety compliance, accelerating the timeline for technology transfer from lab to plant. The simplified post-treatment process, involving standard filtration and chromatography, is easily adaptable to continuous flow chemistry or large batch reactors, facilitating rapid scale-up to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this process for adoption. Understanding these nuances is critical for making informed decisions about process integration and resource allocation.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: This method eliminates the need for toxic carbon monoxide gas cylinders by using a formic acid and acetic anhydride mixture to generate CO in situ, significantly reducing operational hazards and infrastructure costs.
Q: What is the substrate compatibility of this palladium-catalyzed reaction?
A: The reaction demonstrates excellent functional group tolerance, accommodating various substituents on the aryl rings including methyl, methoxy, halogens (chloro, bromo, fluoro), trifluoromethyl, and nitro groups without significant loss in yield.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly mentions that the method has been expanded to gram-scale reactions with high efficiency, indicating strong potential for commercial scale-up due to mild conditions (30°C) and readily available starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN115353511A for accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We are committed to delivering high-purity pharmaceutical intermediates that empower your research teams to focus on innovation rather than supply chain constraints.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data for related compounds and comprehensive route feasibility assessments. Let us be your trusted partner in turning complex chemical challenges into commercial successes.
