Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Commercial Pharmaceutical Manufacturing
Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Commercial Pharmaceutical Manufacturing
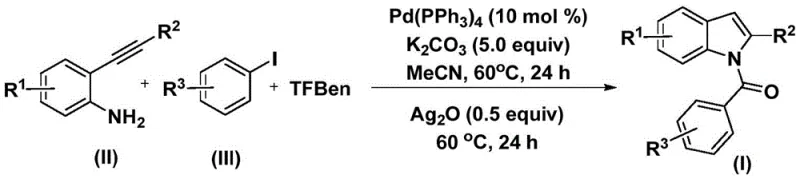
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex heterocyclic scaffolds, particularly indole derivatives which serve as critical backbones in numerous bioactive molecules. Patent CN112898192B introduces a groundbreaking preparation method for N-acyl indole compounds that addresses long-standing challenges in synthetic efficiency and operational safety. This technology leverages a sophisticated palladium-catalyzed carbonylation cyclization strategy, utilizing readily available 2-alkynyl anilines and aryl iodides as primary building blocks. By replacing hazardous carbon monoxide gas with a solid surrogate, the process not only mitigates safety risks but also streamlines the reaction workflow, making it an ideal candidate for the reliable pharmaceutical intermediate supplier seeking to optimize their production lines. The method's ability to operate under relatively mild thermal conditions while maintaining high conversion rates represents a significant leap forward in fine chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for N-acyl indoles often rely on direct carbonylation using carbon monoxide gas, which presents severe logistical and safety hurdles in a commercial manufacturing setting. Handling high-pressure CO gas requires specialized equipment and rigorous safety protocols, increasing both capital expenditure and operational complexity. Furthermore, conventional methods frequently suffer from poor atom economy, harsh reaction conditions that degrade sensitive functional groups, and multi-step sequences that erode overall yield. These inefficiencies create bottlenecks in cost reduction in API manufacturing, as the need for extensive purification and waste management drives up the final price of the active pharmaceutical ingredient. Additionally, the limited substrate scope of older methodologies often restricts the chemical diversity accessible to medicinal chemists, slowing down the drug discovery pipeline.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a solid carbon monoxide substitute, specifically phenol 1,3,5-tricarboxylate (TFBen), to generate CO in situ under controlled conditions. This innovation eliminates the need for gas cylinders and high-pressure reactors, allowing the reaction to proceed in standard laboratory glassware or simplified industrial vessels. The process integrates the carbonylation and cyclization steps into a streamlined sequence, first forming an amide intermediate and subsequently closing the ring to form the indole core. This one-pot style methodology drastically reduces solvent usage and processing time. As illustrated in the general reaction scheme below, the transformation is highly efficient and compatible with a wide array of substituents, ensuring that the structural integrity of complex drug candidates is preserved throughout the synthesis.
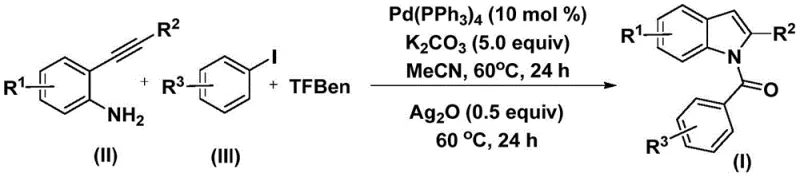
The versatility of this new route is further evidenced by its tolerance to electron-donating and electron-withdrawing groups, enabling the synthesis of diverse libraries of N-acyl indoles essential for structure-activity relationship studies. By shifting from gas-phase reagents to solid-state surrogates, the process aligns perfectly with modern green chemistry principles, reducing the environmental footprint of chemical production while enhancing the reliability of the supply chain for high-purity pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation is a testament to the elegance of transition metal catalysis in organic synthesis. The reaction initiates with the oxidative addition of the palladium catalyst, specifically tetrakis(triphenylphosphine)palladium, into the carbon-iodine bond of the aryl iodide substrate. This generates a reactive aryl-palladium intermediate which then undergoes migratory insertion of carbon monoxide. Crucially, the CO is released thermally from the TFBen precursor, ensuring a steady and controlled concentration of the gas within the reaction medium, which prevents the formation of unwanted bis-carbonylation byproducts. The resulting acyl-palladium species is then intercepted by the nitrogen atom of the 2-alkynyl aniline, undergoing reductive elimination to yield an N-aryl amide intermediate. This step is critical as it sets the stage for the subsequent ring closure.
Following the formation of the amide, the addition of silver oxide triggers the second phase of the mechanism. Silver oxide acts as a mild oxidant and promoter, facilitating the intramolecular cyclization of the alkyne moiety with the adjacent amide nitrogen. This oxidative cyclization step constructs the five-membered pyrrole ring of the indole system with high regioselectivity. The use of silver oxide is particularly advantageous because it operates effectively at moderate temperatures (60°C), preventing the thermal decomposition of sensitive functional groups that might occur under more vigorous conditions. This dual-catalyst system (Pd/Ag) ensures that impurity profiles remain clean, as side reactions such as polymerization of the alkyne or hydrolysis of the amide are minimized. For R&D directors, understanding this mechanism highlights the robustness of the process, ensuring that scaling up from gram to kilogram quantities will not introduce unpredictable impurities that complicate downstream purification.
How to Synthesize N-Acyl Indole Compounds Efficiently
Implementing this synthesis in a practical setting requires careful attention to reagent stoichiometry and thermal management to maximize yield and purity. The protocol is designed to be user-friendly, utilizing common organic solvents like acetonitrile which offer excellent solubility for both polar and non-polar substrates. The reaction proceeds in two distinct thermal stages: an initial carbonylation phase followed by a cyclization phase, both maintained at a consistent temperature of 60°C. This isothermal operation simplifies reactor control and energy consumption. The specific reaction conditions for representative examples are depicted below, showing the precise molar ratios and temporal parameters required for success.
- Combine palladium catalyst (Pd(PPh3)4), potassium carbonate, carbon monoxide substitute (TFBen), 2-alkynyl aniline, and aryl iodide in an organic solvent such as acetonitrile.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation steps.
- Add silver oxide (Ag2O) to the mixture and continue heating at 60°C for an additional 24 hours to induce cyclization, followed by filtration and purification.
To execute this synthesis, one must first combine the palladium catalyst, base, CO source, and substrates in the solvent, ensuring thorough mixing before applying heat. After the first 24-hour period, the careful addition of silver oxide is required without cooling the reaction excessively, maintaining the thermal momentum needed for the cyclization. Following the completion of the second heating cycle, the workup involves simple filtration to remove metal salts and silica gel treatment, followed by standard column chromatography to isolate the high-purity product. This straightforward isolation procedure is a key factor in the commercial viability of the method.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits beyond mere chemical yield. The shift away from gaseous carbon monoxide to a solid surrogate fundamentally alters the risk profile of the manufacturing process, eliminating the need for specialized gas handling infrastructure and reducing insurance and compliance costs associated with toxic gases. This simplification translates directly into cost reduction in pharmaceutical intermediate manufacturing, as the overhead for safety monitoring and equipment maintenance is significantly lowered. Furthermore, the use of commercially available starting materials such as aryl iodides and 2-alkynyl anilines ensures a stable and diversified supply base, reducing the risk of raw material shortages that can disrupt production schedules.
- Cost Reduction in Manufacturing: The elimination of high-pressure gas equipment and the use of a solid CO source drastically simplify the reactor setup, leading to lower capital expenditure and operational costs. The mild reaction conditions also reduce energy consumption compared to high-temperature alternatives, contributing to a leaner manufacturing budget. Additionally, the high atom economy of the carbonylation step minimizes raw material waste, ensuring that every gram of expensive palladium catalyst and substrate is utilized effectively to generate value.
- Enhanced Supply Chain Reliability: By relying on stable, shelf-stable solid reagents like TFBen and silver oxide, the process removes the logistical complexities of managing pressurized gas cylinders. This stability ensures continuous production capability even during supply chain disruptions, as solid chemicals are easier to store and transport globally. The robustness of the reaction against various functional groups means that a single standardized protocol can be used for multiple product variants, streamlining inventory management and reducing the need for specialized custom synthesis runs.
- Scalability and Environmental Compliance: The isothermal nature of the reaction at 60°C makes it highly amenable to scale-up in large-scale stirred tank reactors without the heat transfer issues associated with exothermic gas reactions. The simplified workup, involving filtration and chromatography, generates less hazardous waste compared to traditional methods that might require aggressive quenching or extraction procedures. This alignment with green chemistry principles facilitates easier regulatory approval and environmental compliance, securing the long-term sustainability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this N-acyl indole synthesis protocol. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, aimed at clarifying the operational nuances for technical teams. Understanding these details is crucial for successful technology transfer and process validation. Addressing these points early in the project lifecycle helps mitigate risks and ensures a smoother path from laboratory development to commercial production.
Q: What is the advantage of using phenol 1,3,5-tricarboxylate (TFBen) over carbon monoxide gas?
A: Using TFBen as a solid carbon monoxide substitute significantly enhances operational safety and simplifies handling compared to toxic CO gas, while maintaining high reaction efficiency under mild thermal conditions.
Q: What is the specific role of silver oxide in this synthesis pathway?
A: Silver oxide acts as a crucial promoter in the second stage of the reaction, facilitating the oxidative cyclization of the intermediate amide to form the final N-acyl indole core structure efficiently.
Q: Does this method support diverse functional groups on the substrate?
A: Yes, the protocol demonstrates excellent substrate compatibility, tolerating various substituents such as halogens, alkyl groups, and alkoxy groups on both the aniline and aryl iodide components without significant yield loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and reliable. We are committed to delivering high-purity N-acyl indole intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in palladium-catalyzed transformations allows us to optimize this specific patent technology for maximum yield and minimal impurity formation, providing our partners with a competitive edge in the marketplace.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can be integrated into your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial security.
