Advanced Palladium-Catalyzed Synthesis of N-Acyl Indoles for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing complex heterocyclic scaffolds, particularly the indole nucleus which serves as a privileged structure in numerous bioactive molecules. Patent CN112898192B introduces a significant advancement in this domain by disclosing a novel preparation method for N-acyl indole compounds that addresses many limitations associated with traditional synthetic routes. This technology leverages a palladium-catalyzed carbonylation cyclization strategy, utilizing 2-alkynylanilines and aryl iodides as key starting materials to achieve efficient molecular assembly. The significance of this innovation lies in its ability to generate high-value intermediates used in the production of anti-inflammatory, anti-viral, and anti-tumor agents, as illustrated by the diverse biological activities associated with the indole core structure. 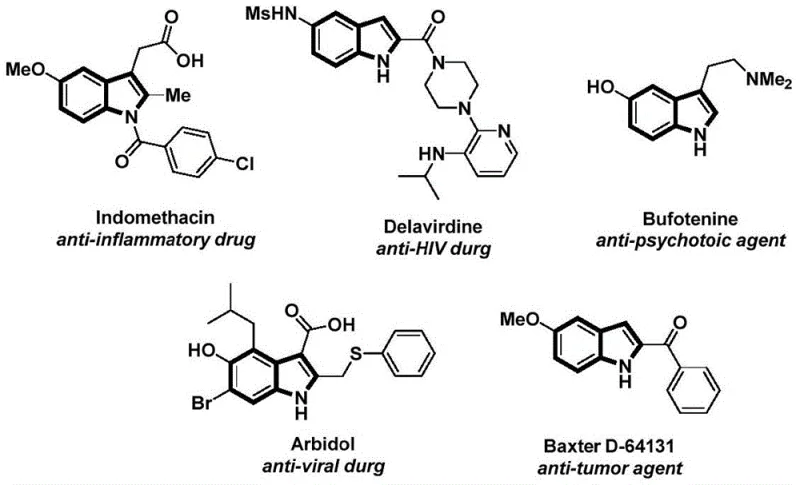 By integrating a solid carbon monoxide surrogate into the reaction system, this process mitigates the safety risks inherent to handling gaseous CO, thereby offering a safer and more manageable protocol for industrial chemists. As a reliable pharmaceutical intermediates supplier, understanding such technological shifts is crucial for maintaining competitive advantage in the global market.
By integrating a solid carbon monoxide surrogate into the reaction system, this process mitigates the safety risks inherent to handling gaseous CO, thereby offering a safer and more manageable protocol for industrial chemists. As a reliable pharmaceutical intermediates supplier, understanding such technological shifts is crucial for maintaining competitive advantage in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indole derivatives has often relied on carbonylation reactions that require the direct use of carbon monoxide gas, presenting substantial logistical and safety challenges for manufacturing facilities. Traditional protocols frequently necessitate high-pressure equipment and specialized containment systems to handle toxic CO gas, which drastically increases capital expenditure and operational complexity for chemical plants. Furthermore, conventional methods often suffer from limited substrate compatibility, where sensitive functional groups on the aromatic rings may degrade under harsh reaction conditions or fail to participate efficiently in the catalytic cycle. The reliance on gaseous reagents also introduces variability in mass transfer rates, leading to inconsistent reaction kinetics and potential difficulties in reproducing results across different batch sizes. These factors collectively contribute to higher production costs and extended lead times, creating bottlenecks in the supply chain for critical drug intermediates. Consequently, there is a pressing demand for alternative methodologies that can deliver high purity products without compromising on safety or operational simplicity.
The Novel Approach
The methodology described in patent CN112898192B represents a paradigm shift by employing 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide surrogate, effectively bypassing the need for hazardous gas cylinders. This innovative approach allows the reaction to proceed under atmospheric pressure conditions, significantly reducing the engineering controls required for safe operation and lowering the barrier to entry for scale-up. The process demonstrates exceptional functional group tolerance, accommodating various substituents such as halogens, alkyl groups, and alkoxy groups on both the aniline and aryl iodide components without significant loss in efficiency. By operating at a moderate temperature of 60°C, the method preserves the integrity of thermally sensitive moieties while ensuring high conversion rates through a optimized catalytic system. This transition from gas-phase to solid-phase carbonylation sources not only enhances safety but also streamlines the workflow, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing. The simplicity of the workup procedure further underscores the practical utility of this novel approach for commercial applications.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The underlying chemical mechanism of this transformation involves a sophisticated sequence of organometallic steps initiated by the oxidative addition of the palladium catalyst into the carbon-iodine bond of the aryl iodide substrate. Following this activation, the carbon monoxide released in situ from the decomposition of TFBen inserts into the aryl-palladium intermediate to generate a reactive acyl-palladium species. This acyl complex then undergoes nucleophilic attack by the amino group of the 2-alkynylaniline, followed by reductive elimination to form an amide intermediate. The presence of silver oxide in the second stage of the reaction plays a critical role in promoting the subsequent cyclization of the amide to form the final N-acyl indole ring system. 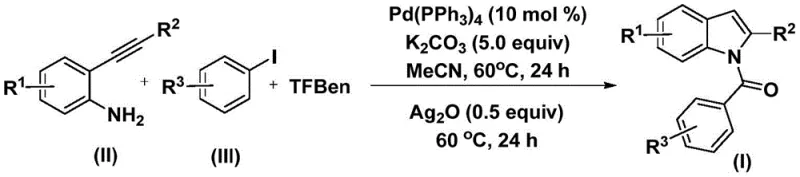 Understanding this mechanistic pathway is vital for R&D directors aiming to optimize reaction parameters for specific substrates, as it highlights the dual role of the catalyst system in both carbonylation and cyclization events. The careful balance of reagents ensures that the catalytic cycle turns over efficiently, minimizing the formation of side products and maximizing the yield of the desired heterocyclic scaffold.
Understanding this mechanistic pathway is vital for R&D directors aiming to optimize reaction parameters for specific substrates, as it highlights the dual role of the catalyst system in both carbonylation and cyclization events. The careful balance of reagents ensures that the catalytic cycle turns over efficiently, minimizing the formation of side products and maximizing the yield of the desired heterocyclic scaffold.
Impurity control is inherently managed through the high selectivity of the palladium catalyst and the mild reaction conditions which prevent degradation of the starting materials or the product. The use of acetonitrile as the solvent provides an optimal medium for dissolving both organic substrates and inorganic bases like potassium carbonate, ensuring homogeneous reaction conditions that favor the desired pathway. The stoichiometric ratio of the reagents, specifically the excess of base and the precise loading of the palladium catalyst, is tuned to suppress competing homocoupling reactions of the aryl iodide or alkyne polymerization. Post-reaction processing involves standard filtration and silica gel chromatography, techniques that are well-established in purification workflows and allow for the removal of residual metal catalysts to meet stringent purity specifications. This level of control over the chemical environment ensures that the final N-acyl indole compounds possess the high quality required for downstream pharmaceutical applications, reducing the burden on quality assurance teams.
How to Synthesize N-Acyl Indole Compounds Efficiently
The standardized protocol for executing this synthesis involves a sequential addition of reagents to ensure optimal mixing and reaction progression within a controlled thermal environment. Operators begin by charging a reaction vessel with the palladium catalyst, potassium carbonate, TFBen, 2-alkynylaniline, and aryl iodide dissolved in acetonitrile, followed by heating the mixture to 60°C for an initial period of 24 hours. Upon completion of the first stage, silver oxide is introduced to the reaction mixture to drive the cyclization step, with heating continued at the same temperature for an additional 24 hours to ensure full conversion. This detailed procedural framework ensures reproducibility and safety, aligning with good manufacturing practices required for the commercial scale-up of complex pharmaceutical intermediates.
- Combine palladium catalyst, potassium carbonate, TFBen, 2-alkynylaniline, and aryl iodide in acetonitrile.
- Heat the mixture at 60°C for 24 hours to facilitate the carbonylation and coupling reaction.
- Add silver oxide and continue heating at 60°C for another 24 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial strategic benefits by simplifying the raw material sourcing landscape and reducing dependency on specialized hazardous gas suppliers. The starting materials, including 2-alkynylanilines and aryl iodides, are commercially available commodities that can be sourced from multiple vendors, thereby enhancing supply chain resilience and mitigating the risk of single-source bottlenecks. The elimination of high-pressure carbon monoxide gas removes the need for expensive cylinder leasing, specialized transport logistics, and rigorous safety inspections associated with toxic gas handling, leading to significant overhead cost reductions. Furthermore, the mild reaction temperature of 60°C reduces energy consumption compared to high-temperature processes, contributing to lower utility costs and a smaller carbon footprint for the manufacturing facility. These operational efficiencies translate directly into improved margin potential and more competitive pricing structures for the final active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The replacement of gaseous carbon monoxide with a solid surrogate eliminates the capital investment required for high-pressure reactors and gas containment infrastructure, resulting in drastic simplification of the production setup. By avoiding the use of expensive transition metal removal resins often needed for harsher catalytic systems, the downstream purification costs are significantly lowered, enhancing the overall economic viability of the process. The high atom economy and efficient conversion rates minimize waste generation, reducing the expenses associated with waste disposal and environmental compliance management. Additionally, the robustness of the reaction allows for potential telescoping of steps, which can further compress the manufacturing timeline and reduce labor costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: Utilizing readily available solid reagents ensures that production schedules are not disrupted by the delivery delays often associated with regulated hazardous gases. The stability of the TFBen reagent allows for bulk purchasing and long-term storage without degradation, providing a buffer against market volatility and ensuring continuous operation during supply fluctuations. The compatibility of the method with standard glass-lined or stainless steel reactors means that existing manufacturing assets can be utilized without major retrofitting, accelerating the time-to-market for new products. This flexibility empowers supply chain heads to respond rapidly to demand surges without being constrained by specialized equipment availability or regulatory hurdles related to gas transport.
- Scalability and Environmental Compliance: The mild conditions and absence of toxic gas emissions make this process inherently safer and more environmentally friendly, aligning with increasingly strict global regulations on industrial emissions. Scaling from laboratory to pilot and commercial production is streamlined due to the homogeneous nature of the reaction and the lack of complex gas-liquid mass transfer limitations. The simplified workup procedure reduces the volume of organic solvents required for purification, supporting green chemistry initiatives and reducing the environmental impact of the manufacturing site. These factors collectively position this technology as a sustainable choice for long-term production strategies, ensuring compliance with environmental standards while maintaining high output capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method in an industrial setting. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for decision-makers.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen acts as a solid carbon monoxide surrogate, eliminating the need for hazardous high-pressure CO gas cylinders and significantly improving operational safety.
Q: What are the typical reaction conditions for this N-acyl indole preparation?
A: The reaction proceeds under mild thermal conditions at 60°C in acetonitrile solvent, utilizing a two-stage process with a total reaction time of approximately 48 hours.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the use of commercially available reagents, mild temperatures, and simple post-processing makes this method highly scalable for industrial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we leverage cutting-edge synthetic technologies like the one described in CN112898192B to deliver high-quality intermediates that meet the rigorous demands of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-acyl indole compounds complies with international regulatory standards. Our commitment to technical excellence allows us to offer customized solutions that optimize both cost and performance for your specific application requirements.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your supply chain and product portfolio. Request a Customized Cost-Saving Analysis today to understand the potential economic impact of switching to this safer and more efficient methodology. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your project needs, ensuring a partnership built on transparency and scientific rigor. Contact us now to secure a reliable supply of high-purity pharmaceutical intermediates for your next breakthrough therapy.
