Advanced Synthesis of Trifluoromethyl-Substituted Dihydrofuran Amines for Pharma
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to incorporate trifluoromethyl groups into heterocyclic scaffolds, driven by the unique metabolic stability and lipophilicity these motifs impart to drug candidates. Patent CN110922369B introduces a groundbreaking preparation method for trifluoromethyl-substituted dihydrofuran amine compounds, addressing the longstanding challenges associated with constructing CF3-bearing quaternary carbon centers. Dihydrofuran rings are pivotal structural elements found in numerous bioactive natural products such as Azadirachtin and Austocystin A, yet their synthesis often involves complex multi-step sequences. This innovation provides a direct, catalytic route that bypasses the limitations of earlier stoichiometric approaches, offering a streamlined pathway for generating high-value pharmaceutical intermediates. The ability to access these structures efficiently opens new avenues for medicinal chemists exploring novel chemical space.
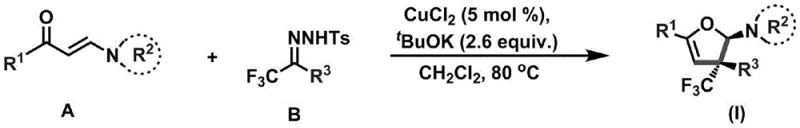
The significance of this technology extends beyond mere academic interest; it represents a tangible opportunity for cost reduction in fine chemical manufacturing. By utilizing readily available acyclic substrates like enaminones and trifluoromethylhydrazones, the process eliminates the need for pre-functionalized cyclic starting materials which are often expensive and difficult to source. Furthermore, the reaction demonstrates exceptional substrate applicability, tolerating a wide range of electronic and steric environments on the aromatic rings. This versatility ensures that a diverse library of analogs can be generated from a single platform, accelerating the lead optimization phase in drug discovery while maintaining a lean and efficient supply chain for critical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of dihydrofuran rings has relied heavily on classical cycloaddition strategies such as [4+1] or [3+2] reactions, which frequently suffer from significant operational drawbacks. Many of these traditional protocols necessitate the use of stoichiometric amounts of transition metals, leading to excessive metal waste and complicating the downstream purification processes required to meet stringent pharmaceutical purity standards. Additionally, these methods often demand harsh reaction conditions, including extreme temperatures or highly reactive reagents, which can compromise the integrity of sensitive functional groups present on the substrate. Such limitations restrict the scope of accessible chemical space and often result in poor yields when applied to complex molecular architectures, thereby increasing the overall cost and time required for process development.
The Novel Approach
In stark contrast, the methodology disclosed in CN110922369B employs a catalytic system that operates under remarkably mild conditions, typically requiring heating to only 80-100°C in a common solvent like dichloromethane. This shift from stoichiometric to catalytic usage of copper species not only reduces the raw material costs but also minimizes the environmental footprint associated with heavy metal disposal. The reaction exhibits superior functional group tolerance, allowing for the successful incorporation of diverse substituents including halogens, nitro groups, and ethers without the need for extensive protecting group strategies. This robustness translates directly into higher process efficiency and reliability, making it an ideal candidate for the commercial scale-up of complex heterocycles where consistency and yield are paramount.
Mechanistic Insights into CuCl2-Catalyzed Cyclization
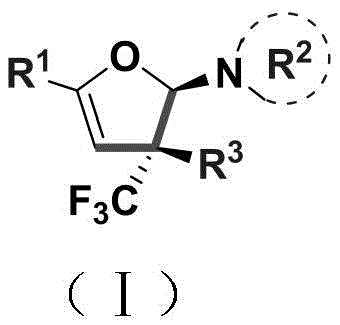
The core of this synthetic breakthrough lies in the copper-catalyzed coupling between an enaminone and a trifluoromethylhydrazone, which proceeds through a sophisticated cyclization mechanism to form the dihydrofuran core. The copper chloride catalyst, used in a mere 5 mol% loading, activates the hydrazone species to facilitate nucleophilic attack and subsequent ring closure, effectively constructing the challenging quaternary carbon stereocenter in a single operation. This catalytic cycle is highly efficient, driving the reaction to completion over a period of 48-72 hours while maintaining high stereoselectivity. The presence of potassium tert-butoxide as a base is crucial for deprotonation steps that enable the formation of the reactive intermediates necessary for the cyclization to proceed smoothly.
From an impurity control perspective, the mild nature of this reaction conditions plays a vital role in ensuring the quality of the final product. Harsh conditions often lead to decomposition pathways or side reactions that generate difficult-to-remove impurities, but this protocol minimizes such risks by operating within a温和 thermal window. The resulting dihydrofuran amine carries a defined stereochemistry that is preserved throughout the transformation, reducing the need for costly chiral resolution steps later in the synthesis. For R&D teams, understanding this mechanism allows for better prediction of substrate compatibility and optimization of reaction parameters, ensuring that the high-purity pharmaceutical intermediates produced meet the rigorous specifications required for clinical trial materials and eventual commercial production.
How to Synthesize Trifluoromethyl-Substituted Dihydrofuran Amine Efficiently
To implement this synthesis effectively, one must adhere to the specific molar ratios and conditions outlined in the patent to maximize yield and reproducibility. The process begins with the precise mixing of the enaminone and trifluoromethylhydrazone precursors under an inert atmosphere to prevent oxidation of the sensitive intermediates. Detailed standard operating procedures regarding temperature ramping, stirring rates, and workup techniques are essential for translating this laboratory-scale success to pilot plant operations. The following guide outlines the critical steps necessary to achieve the reported efficiency and quality.
- Mix enaminone compound A, trifluoromethylhydrazone compound B, copper chloride catalyst, and potassium tert-butoxide under inert gas protection.
- Add dichloromethane solvent and heat the reaction solution to 80-100°C, stirring continuously for 48-72 hours to ensure complete cyclization.
- Filter off insoluble solids, concentrate the solution under reduced pressure, and purify the crude product via column chromatography to obtain the target amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers substantial strategic benefits that extend well beyond the laboratory bench. The transition from stoichiometric to catalytic metal usage fundamentally alters the cost structure of the manufacturing process, eliminating the need for expensive metal scavengers and reducing the burden on waste treatment facilities. Moreover, the use of common solvents and mild temperatures lowers energy consumption and equipment stress, contributing to a more sustainable and economically viable production model. These factors collectively enhance the resilience of the supply chain against raw material price volatility and regulatory changes regarding environmental compliance.
- Cost Reduction in Manufacturing: The utilization of a catalytic amount of copper chloride significantly lowers the direct material costs compared to traditional methods that require stoichiometric transition metals. By avoiding the use of excess reagents and simplifying the purification workflow through filtration and standard chromatography, the overall processing time is reduced, leading to substantial cost savings. This efficiency allows for more competitive pricing structures without compromising on the quality or purity of the final intermediate, providing a clear economic advantage in a margin-sensitive market.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, specifically enaminones and trifluoromethylhydrazones, are generally more accessible and stable than the specialized cyclic precursors required by older methods. This availability reduces the risk of supply disruptions and shortens the lead time for sourcing raw materials. Furthermore, the robustness of the reaction conditions means that production is less susceptible to minor variations in utility supplies or environmental factors, ensuring a consistent and reliable flow of goods to downstream customers who depend on timely deliveries for their own manufacturing schedules.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the simplicity of the workup procedure, which involves basic filtration and solvent removal rather than complex extraction or crystallization steps. The reduced generation of heavy metal waste aligns with increasingly strict global environmental regulations, minimizing the compliance burden and potential liabilities associated with hazardous waste disposal. This environmental friendliness not only protects the company's reputation but also future-proofs the manufacturing process against tightening ecological standards in key markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their supply chains. Understanding these details is crucial for making informed decisions about process adoption and partnership opportunities.
Q: What are the key advantages of this copper-catalyzed method over traditional cycloadditions?
A: Unlike conventional [4+1] or [3+2] cycloadditions that often require stoichiometric transition metals and harsh conditions, this novel approach utilizes a catalytic amount of copper chloride under mild temperatures (80-100°C). This significantly enhances functional group tolerance and simplifies the purification process, making it more suitable for sensitive pharmaceutical intermediates.
Q: How does this synthesis method impact the stereochemistry of the final product?
A: The reaction is designed to effectively obtain stereoselective trifluoromethyl-substituted dihydrofuran amine derivatives. Crucially, the synthesized molecules carry a quaternary carbon stereocenter, which is a challenging structural motif to construct but highly valuable for creating rigid, biologically active scaffolds in drug discovery programs.
Q: Can this intermediate be further derivatized for other applications?
A: Yes, the resulting dihydrofuran amine serves as a versatile synthetic intermediate. It can be quantitatively converted into trifluoromethyl-containing 1,4-dicarbonyl compounds under mild acidic conditions, which are precursors for constructing various other trifluoromethyl-substituted heterocycles widely used in agrochemicals and materials science.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl-Substituted Dihydrofuran Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative academic research into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN110922369B can be realized on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of trifluoromethyl-substituted dihydrofuran amine meets the exacting standards required by top-tier pharmaceutical companies. Our commitment to quality assurance ensures that your drug development programs proceed without interruption due to material inconsistencies.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce your overall time to market. Let us be your partner in bringing these high-value intermediates from the lab to the global marketplace efficiently and sustainably.
