Advanced Manufacturing of Roflumilast Intermediates via Activated Ester Amidation
The pharmaceutical industry's relentless pursuit of efficient Chronic Obstructive Pulmonary Disease (COPD) therapeutics has placed significant scrutiny on the manufacturing processes of key active ingredients like Roflumilast. Patent CN102336705B introduces a transformative synthetic methodology that addresses long-standing inefficiencies in producing N-(3,5-dichloropyridyl-4-yl)-3-cyclopropylmethoxy-4-difluoromethoxybenzoyl amine. Unlike traditional approaches that rely on highly reactive and unstable acid chlorides, this innovation leverages the stability of activated aromatic esters to drive the amidation reaction. This strategic shift not only mitigates the violent exothermic profiles associated with prior art but also drastically improves the impurity profile of the final API. For R&D directors and procurement specialists alike, this patent represents a pivotal opportunity to optimize the supply chain for this high-value respiratory medication.
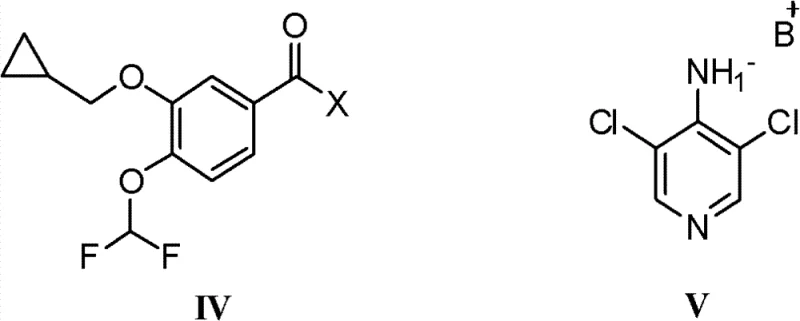
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Roflumilast has been dominated by methods described in international patents such as WO2004/080967, which utilize 3-cyclopropylmethoxy-4-difluoromethoxybenzoyl chloride as the electrophile. While chemically feasible, this acid chloride route suffers from severe practical drawbacks when translated to an industrial setting. The reaction between the acid chloride and the amine salt is inherently violent and difficult to control, often requiring cryogenic conditions to manage the exotherm. Furthermore, the high reactivity of the acid chloride makes it susceptible to hydrolysis, leading to the persistent formation of the troublesome byproduct N-(3,5-dichloropyridine-4-yl)-3-cyclopropylmethoxy-4-hydroxybenzamide. This specific impurity is structurally similar to the target molecule, making it exceptionally difficult to remove via standard recrystallization, thereby necessitating multiple purification cycles that erode overall yield. Additionally, the conventional process typically demands a significant excess of the amine starting material, often exceeding 2.2 equivalents, which creates substantial waste and inflates raw material costs.
The Novel Approach
The methodology disclosed in CN102336705B fundamentally re-engineers the electrophile by replacing the acid chloride with a 3-cyclopropylmethoxy-4-difluoromethoxybenzoic acid aromatic ester. This structural modification allows the amidation to proceed under remarkably mild conditions, typically between 10°C and 50°C, eliminating the need for hazardous cryogenic control. The reduced reactivity of the ester compared to the acid chloride ensures a smoother reaction profile that minimizes side reactions, particularly the hydrolysis that plagues the older methods. By utilizing a leaving group such as a nitrophenyl or substituted phenoxy group, the reaction achieves high conversion rates with a near-stoichiometric ratio of reactants, often requiring only 1.1 to 1.3 equivalents of the amine component. This precision not only enhances atom economy but also simplifies the downstream processing, as the crude product obtained possesses a purity greater than 99% before any recrystallization is even attempted.
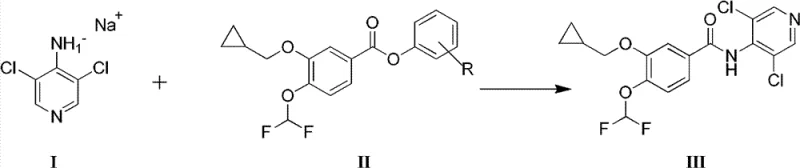
Mechanistic Insights into Activated Ester Amidation
The core mechanistic advantage of this process lies in the nuanced balance of nucleophilicity and leaving group ability. In the first stage, the 4-amino-3,5-dichloropyridine is deprotonated by a strong base such as sodium hydride or sodium tert-butoxide in an aprotic polar solvent like DMF to generate a stable anionic species. This anion then acts as a potent nucleophile, attacking the carbonyl carbon of the activated aromatic ester. Unlike the acid chloride, where the chloride leaving group departs almost instantaneously and can lead to uncontrolled kinetics, the phenoxy leaving group in the ester provides a more moderated reaction rate. This moderation is critical for preserving the integrity of the sensitive difluoromethoxy moiety on the benzene ring, which is prone to decomposition under harsh acidic or basic conditions found in less optimized routes. The transition state is stabilized by the solvent cage of the DMF, facilitating a clean substitution that releases the phenol byproduct without generating significant thermal spikes.
From an impurity control perspective, the mechanism effectively suppresses the formation of the hydrolyzed byproduct. In the conventional acid chloride route, trace moisture or the inherent instability of the intermediate can lead to the cleavage of the difluoromethoxy group or the amide bond itself. However, the activated ester route operates in a regime where the activation energy for the desired amidation is lower than that for the degradation pathways. The use of specific leaving groups, such as p-nitrophenyl, further drives the equilibrium towards the product due to the electron-withdrawing nature of the nitro group, which enhances the electrophilicity of the carbonyl carbon just enough to react with the pyridine anion but not enough to promote random hydrolysis. Consequently, the final crude solid contains minimal levels of the hydroxy-impurity, often below 0.1%, allowing for a single recrystallization step from ethanol to achieve the stringent pharmacopeial purity of >99.8%.
How to Synthesize Roflumilast Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing safety and reproducibility. The process begins with the preparation of the activated ester, followed by the generation of the pyridine anion, and concludes with the coupling reaction and purification. Operators must maintain strict inert atmosphere conditions using nitrogen to prevent moisture ingress, which could compromise the base or the intermediate. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and temperature ramps, are critical for replicating the high yields reported in the examples. For a comprehensive breakdown of the operational parameters required to achieve these results, please refer to the technical guide below.
- Preparation of the activated ester intermediate by reacting 3-cyclopropylmethoxy-4-difluoromethoxybenzoyl chloride with substituted phenols under basic conditions.
- Generation of the 4-amino-3,5-dichloropyridine anion using a strong base like sodium hydride in an inert aprotic solvent such as DMF.
- Controlled amidation reaction between the anion and the activated ester at mild temperatures (10-50°C), followed by aqueous workup and ethanol recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this activated ester technology translates directly into tangible operational efficiencies and risk mitigation. The shift away from the volatile acid chloride chemistry reduces the dependency on specialized cryogenic equipment and lowers the safety risks associated with handling highly corrosive reagents. This simplification of the process infrastructure allows for more flexible manufacturing scheduling and reduces the capital expenditure required for reactor setups. Furthermore, the ability to use near-stoichiometric amounts of the expensive amine starting material significantly lowers the bill of materials, as there is no longer a need to purchase and dispose of large excesses of raw materials. The streamlined purification process, which requires fewer solvent exchanges and recrystallization cycles, also leads to a substantial reduction in solvent consumption and waste disposal costs, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of excess reagents and the reduction in purification steps create a leaner manufacturing process that significantly lowers the cost of goods sold. By avoiding the need for multiple recrystallizations to remove stubborn impurities, manufacturers save on both solvent costs and the time value of money tied up in production cycles. The higher overall yield means that less raw material is required to produce the same amount of final API, providing a direct margin improvement that can be passed down the supply chain or retained as profit.
- Enhanced Supply Chain Reliability: The use of stable activated esters as intermediates offers greater flexibility in inventory management. Unlike acid chlorides, which often have short shelf lives and require cold storage, the aromatic ester intermediates are robust and can be stockpiled, ensuring continuity of supply even during raw material fluctuations. The mild reaction conditions also reduce the likelihood of batch failures due to thermal runaways, leading to more predictable delivery schedules and higher reliability for downstream pharmaceutical customers who depend on consistent API availability.
- Scalability and Environmental Compliance: The process is inherently scalable due to its mild thermal profile and the use of common industrial solvents like DMF and ethanol. The reduction in hazardous waste generation, specifically the avoidance of large volumes of saline wastewater from neutralization steps typical in acid chloride routes, simplifies environmental compliance. This makes the technology particularly attractive for manufacturing sites facing strict regulatory scrutiny regarding effluent discharge, facilitating easier permitting and long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of technology transfer and the potential impact on existing production lines.
Q: Why is the activated ester method superior to the acid chloride method for Roflumilast?
A: The activated ester method operates under much milder conditions, significantly reducing the formation of hydrolysis byproducts and eliminating the need for excessive reagent equivalents, which lowers costs and simplifies purification.
Q: What represents the critical quality attribute in this synthesis?
A: The critical quality attribute is the minimization of the N-(3,5-dichloropyridine-4-yl)-3-cyclopropylmethoxy-4-hydroxybenzamide impurity, which is notoriously difficult to remove in conventional acid chloride routes.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents like DMF and ethanol, operates at safe temperatures below 50°C, and achieves high yields with simple filtration and crystallization steps, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Roflumilast Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits observed in the laboratory are fully realized in the manufacturing plant. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Roflumilast intermediate meets the highest global standards. Our commitment to quality assurance ensures that the complex challenges of impurity control are managed proactively, delivering a product that is ready for the next stage of API synthesis.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages tailored to your volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and our demonstrated capability to deliver high-performance pharmaceutical intermediates reliably.
