Advanced One-Pot Synthesis of N-Acyl Indoles for High-Value Pharmaceutical Manufacturing
Advanced One-Pot Synthesis of N-Acyl Indoles for High-Value Pharmaceutical Manufacturing
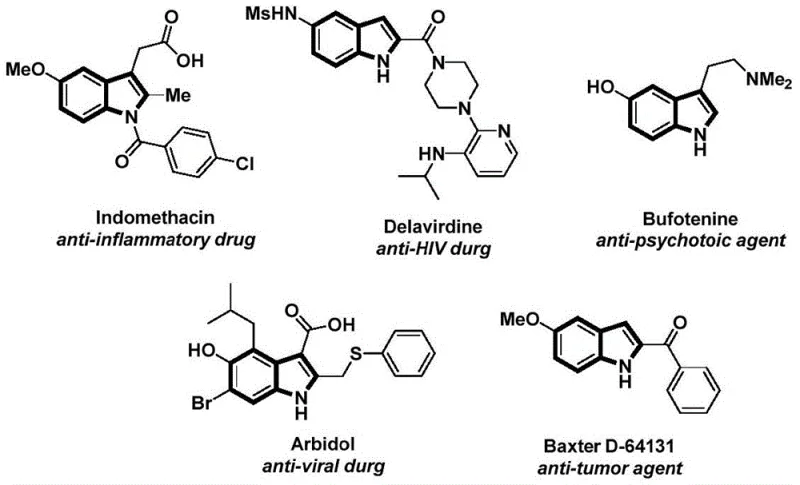
The structural motif of indole is ubiquitous in medicinal chemistry, serving as the core scaffold for a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to potent anti-tumor compounds. As depicted in the reference structures of known pharmaceuticals, the N-acyl indole framework is particularly prized for its stability and biological activity profile. However, constructing this specific architecture efficiently has historically posed significant challenges in process chemistry. Patent CN112898192B introduces a transformative approach to this problem, detailing a robust, palladium-catalyzed carbonylative cyclization strategy. This innovation allows for the direct assembly of N-acyl indoles from readily available 2-alkynyl anilines and aryl iodides in a single operational sequence. For R&D directors and procurement specialists alike, this methodology represents a paradigm shift, replacing hazardous gaseous carbon monoxide protocols with a safe, solid CO surrogate while maintaining high reaction efficiency and exceptional substrate compatibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of N-acyl indoles often relies on multi-step sequences that are inherently inefficient and costly for large-scale manufacturing. Classical routes typically involve the separate preparation of indole cores followed by N-acylation using acid chlorides or anhydrides, which generates stoichiometric amounts of corrosive waste and requires harsh reaction conditions. Alternatively, carbonylation reactions utilizing gaseous carbon monoxide (CO) present severe safety hazards and require specialized high-pressure equipment, making them unsuitable for many standard pharmaceutical production facilities. Furthermore, these conventional methods frequently suffer from poor regioselectivity and limited functional group tolerance, necessitating extensive protection and deprotection strategies that drastically increase the step count and reduce overall yield. The reliance on toxic gases and complex purification protocols creates significant bottlenecks in the supply chain, leading to extended lead times and inflated production costs for critical pharmaceutical intermediates.
The Novel Approach
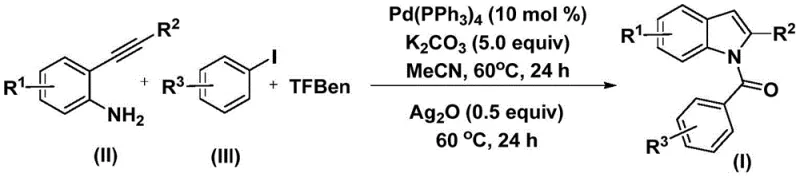
The methodology disclosed in patent CN112898192B elegantly circumvents these historical limitations through a cleverly designed tandem reaction sequence. By employing phenol 1,3,5-tricarboxylate (TFBen) as a solid carbon monoxide surrogate, the process eliminates the need for handling dangerous CO gas, thereby enhancing operational safety and simplifying reactor requirements. The reaction proceeds under remarkably mild conditions, utilizing a palladium catalyst system in acetonitrile at a moderate temperature of 60°C. This one-pot protocol seamlessly integrates the carbonylation, amidation, and cyclization steps, directly transforming simple starting materials into complex N-acyl indole scaffolds. The use of silver oxide as a secondary promoter in the latter stage ensures high conversion rates for the cyclization step, delivering products with impressive purity. This streamlined approach not only reduces the environmental footprint by minimizing waste but also significantly accelerates the timeline from raw material to finished intermediate, offering a compelling value proposition for cost-sensitive manufacturing environments.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the aryl iodide, generating a reactive aryl-palladium(II) species. Simultaneously, the thermal decomposition of TFBen releases carbon monoxide in situ, which subsequently inserts into the aryl-palladium bond to form an acyl-palladium intermediate. This key acyl species then undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, followed by reductive elimination to yield an intermediate amide. The elegance of this system lies in the second phase, where the addition of silver oxide facilitates the intramolecular cyclization. The silver species likely activates the alkyne moiety or assists in the deprotonation necessary for the nitrogen atom to attack the triple bond, closing the five-membered pyrrole ring to form the final indole core. This dual-catalytic influence ensures that the reaction proceeds with high fidelity, minimizing side reactions such as homocoupling or incomplete carbonylation.
From an impurity control perspective, the choice of reagents plays a pivotal role in ensuring a clean product profile. The use of potassium carbonate as a base provides sufficient alkalinity to drive the reaction without promoting excessive hydrolysis of the sensitive acyl intermediates. Furthermore, the specific molar ratios defined in the patent—such as 10 mol% of tetrakis(triphenylphosphine)palladium and 0.5 equivalents of silver oxide—are optimized to balance reaction rate with catalyst turnover. The broad substrate scope demonstrated in the patent data indicates that electronic effects on the aromatic rings (whether electron-donating methoxy groups or electron-withdrawing halogens) are well-tolerated, suggesting a robust catalytic cycle that is not easily poisoned by common functional groups. This mechanistic resilience translates directly to manufacturing reliability, as it reduces the risk of batch-to-batch variability caused by minor fluctuations in raw material quality.
How to Synthesize N-Acyl Indole Compounds Efficiently
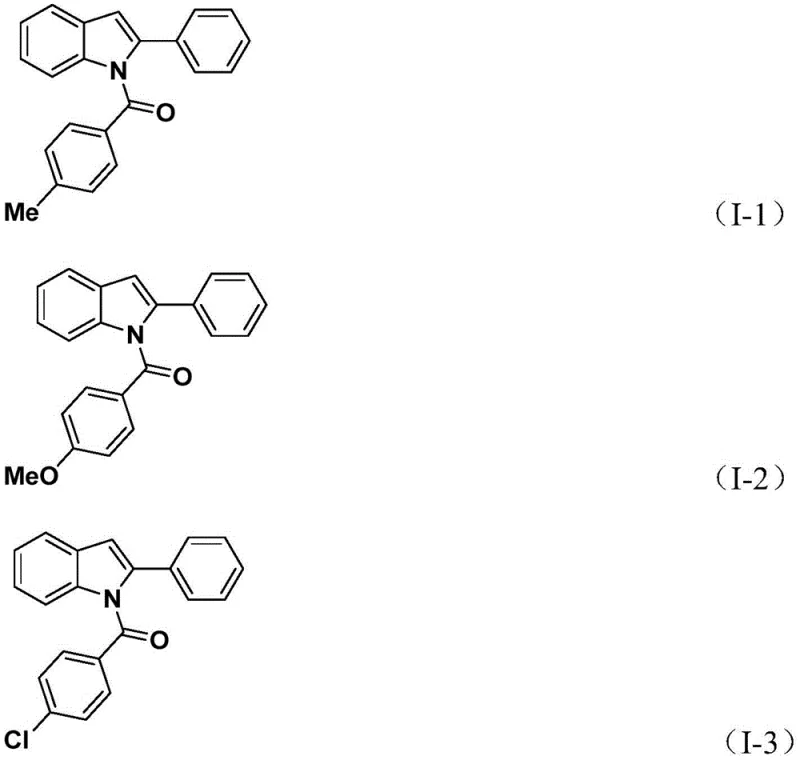
The practical execution of this synthesis is designed for scalability and ease of operation, making it highly attractive for process chemists looking to transition from benchtop discovery to pilot plant production. The protocol utilizes standard laboratory glassware and does not require exotic equipment, relying instead on precise control of temperature and reagent addition timing. The two-stage heating process is critical; the initial 24-hour period allows for the complete formation of the amide linkage, while the subsequent addition of silver oxide and further heating drives the cyclization to completion. Workup procedures are equally straightforward, involving simple filtration to remove inorganic salts followed by standard silica gel chromatography. This simplicity in downstream processing is a major advantage, as it minimizes solvent consumption and reduces the time operators spend on purification, thereby increasing overall throughput.
- Combine palladium catalyst (Pd(PPh3)4), potassium carbonate, TFBen (CO source), 2-alkynyl aniline, and aryl iodide in acetonitrile.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide (Ag2O) to the mixture and continue heating at 60°C for another 24 hours to induce cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the N-acyl indole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the significant reduction of operational complexity and associated costs. By replacing hazardous gaseous reagents with stable solids, the process lowers the barrier for entry for contract manufacturing organizations (CMOs) that may lack high-pressure gas infrastructure. This accessibility expands the pool of potential suppliers, fostering a more competitive bidding environment and enhancing supply chain resilience. Moreover, the high yields reported across a diverse range of substrates indicate a mature and reliable process that minimizes raw material waste, directly contributing to cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this methodology is driven by the elimination of expensive and dangerous reagents. Traditional carbonylation often requires specialized autoclaves and rigorous safety protocols for handling carbon monoxide, which incur substantial capital and operational expenditures. In contrast, this method utilizes TFBen, a benign solid that decomposes cleanly, removing the need for such specialized infrastructure. Additionally, the high atom economy of the reaction means that a greater proportion of the starting materials ends up in the final product, reducing the cost per kilogram of the active pharmaceutical ingredient (API) intermediate. The ability to run the reaction at a moderate 60°C also results in lower energy consumption compared to high-temperature alternatives, further driving down utility costs over long production runs.
- Enhanced Supply Chain Reliability: Supply continuity is paramount in the pharmaceutical industry, and this process bolsters reliability through the use of commercially available starting materials. Both 2-alkynyl anilines and aryl iodides are commodity chemicals that can be sourced from multiple global vendors, mitigating the risk of single-source dependency. The robustness of the reaction conditions ensures that production schedules are less likely to be disrupted by sensitivity to moisture or oxygen, which often plague other transition-metal catalyzed processes. This stability allows for more accurate forecasting and inventory management, ensuring that critical drug candidates do not face delays due to intermediate shortages. The simplified workup also means faster turnaround times from reaction completion to shipment, effectively reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: As regulatory pressures regarding waste disposal and environmental impact intensify, this green chemistry approach positions manufacturers favorably. The avoidance of toxic CO gas and the use of acetonitrile, a solvent with established recovery protocols, align with modern sustainability goals. The reaction generates minimal hazardous byproducts, primarily inorganic salts that are easy to dispose of or treat, simplifying compliance with environmental regulations. From a scale-up perspective, the exothermic profile of the reaction is manageable, and the absence of high-pressure steps removes significant engineering hurdles associated with scaling carbonylation reactions. This makes the commercial scale-up of complex N-acyl indoles feasible even for facilities with standard reactor configurations, ensuring a smooth transition from gram-scale optimization to ton-scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this patented synthesis route. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity for technical teams evaluating this technology for their own pipelines. Understanding these nuances is essential for successful technology transfer and process validation.
Q: What is the carbon monoxide source in this synthesis?
A: The process utilizes phenol 1,3,5-tricarboxylate (TFBen) as a solid, safe, and efficient carbon monoxide substitute, avoiding the handling of toxic CO gas.
Q: Why is silver oxide added in the second step?
A: Silver oxide acts as a crucial promoter for the intramolecular cyclization step, converting the intermediate amide into the final N-acyl indole structure under mild thermal conditions.
Q: What is the substrate compatibility of this method?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents such as halogens (F, Cl, Br), alkyl groups (Me, tBu), and alkoxy groups (OMe) on both the aniline and aryl iodide components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists has thoroughly analyzed the potential of the palladium-catalyzed carbonylative cyclization described in patent CN112898192B and is fully prepared to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including the safe handling of palladium catalysts and the rigorous purification steps needed to meet stringent purity specifications. With our dedicated QC labs and commitment to quality, we guarantee that every batch of N-acyl indole intermediate delivered meets the highest international standards.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your specific supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our existing library of indole derivatives or to discuss route feasibility assessments for your novel candidates. Let us help you secure a reliable, cost-effective, and scalable supply of these vital pharmaceutical building blocks.
