Advanced Palladium-Catalyzed Synthesis of N-Acyl Indole Compounds for Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of N-Acyl Indole Compounds for Pharmaceutical Applications
The structural motif of the indole ring serves as a foundational backbone in modern medicinal chemistry, appearing in a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to potent anti-HIV drugs such as Delavirdine. As illustrated in the diverse pharmacological landscape, the ability to efficiently construct functionalized indole derivatives is paramount for drug discovery and development pipelines. 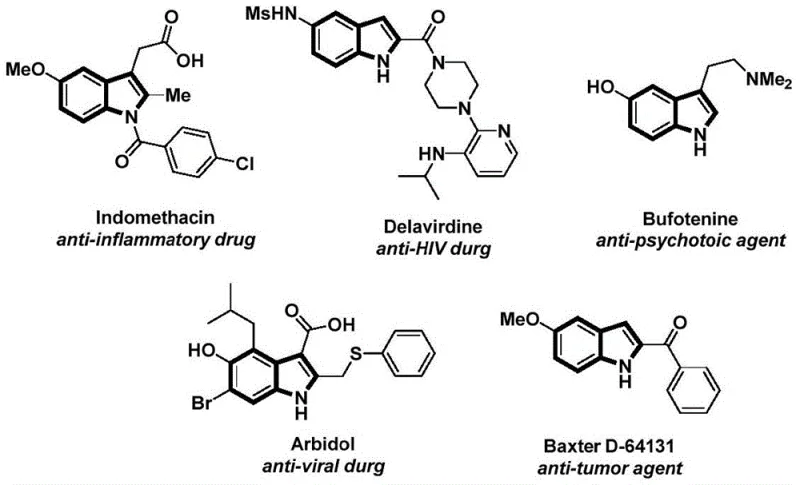 Recent advancements in synthetic methodology have sought to address the limitations of classical indole synthesis, culminating in the innovative approach detailed in Chinese Patent CN112898192B. This patent discloses a robust preparation method for N-acyl indole compounds that leverages a palladium-catalyzed carbonylation cyclization strategy. By utilizing 2-alkynyl aniline and aryl iodides as readily accessible starting materials, this technology offers a streamlined pathway to high-value pharmaceutical intermediates, bypassing the safety hazards and operational complexities associated with traditional high-pressure carbonylation processes.
Recent advancements in synthetic methodology have sought to address the limitations of classical indole synthesis, culminating in the innovative approach detailed in Chinese Patent CN112898192B. This patent discloses a robust preparation method for N-acyl indole compounds that leverages a palladium-catalyzed carbonylation cyclization strategy. By utilizing 2-alkynyl aniline and aryl iodides as readily accessible starting materials, this technology offers a streamlined pathway to high-value pharmaceutical intermediates, bypassing the safety hazards and operational complexities associated with traditional high-pressure carbonylation processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indoles has often relied on multi-step sequences involving harsh reaction conditions or the direct use of carbon monoxide gas, which poses significant safety risks in an industrial setting due to its toxicity and flammability. Conventional carbonylation reactions frequently require specialized high-pressure autoclaves and rigorous safety protocols, which drastically increase capital expenditure and operational overhead for chemical manufacturers. Furthermore, traditional methods often suffer from poor substrate compatibility, where sensitive functional groups on the aromatic rings may degrade under the aggressive thermal or acidic conditions required for cyclization. This lack of chemoselectivity frequently necessitates additional protection and deprotection steps, lengthening the overall synthesis timeline and reducing the overall atom economy of the process. Consequently, there has been a persistent demand in the fine chemical industry for a milder, safer, and more versatile synthetic route that can accommodate the complex structural requirements of modern drug candidates without compromising on yield or purity.
The Novel Approach
The methodology described in patent CN112898192B represents a paradigm shift by employing a solid carbon monoxide surrogate, specifically 1,3,5-tricarboxylic acid phenol ester (TFBen), thereby eliminating the need for handling gaseous CO. This novel approach enables a one-pot tandem reaction where the carbonylation and cyclization occur sequentially under relatively mild thermal conditions (60°C). The versatility of this system is evidenced by its broad substrate scope, successfully accommodating various substituents such as halogens, alkyl groups, and alkoxy groups on both the aniline and aryl iodide components. 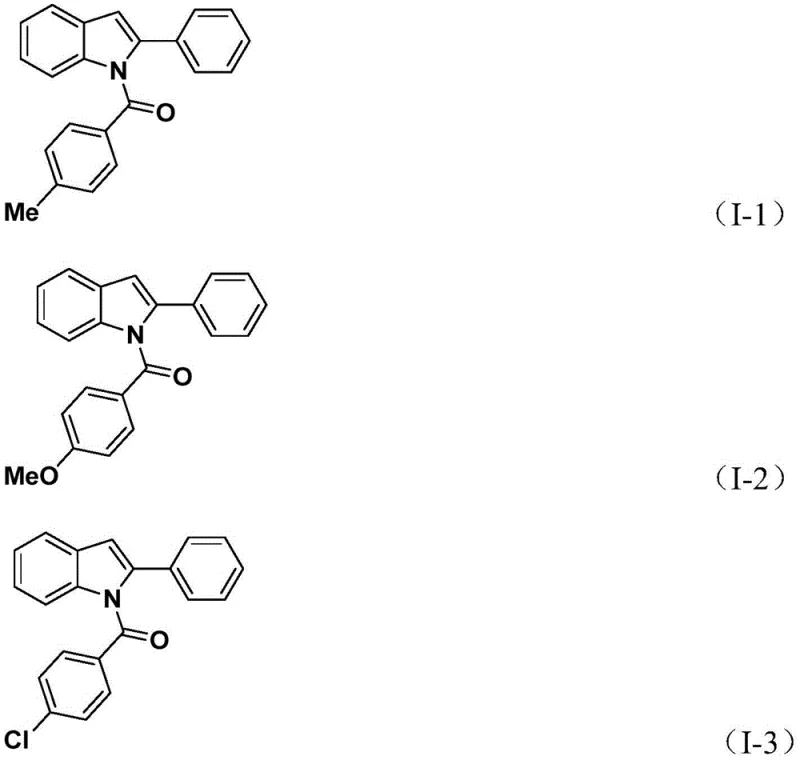 As shown in the specific examples provided, the method consistently delivers N-acyl indole products with high structural fidelity, such as compounds I-1 through I-5, proving its utility for generating diverse chemical libraries. This operational simplicity not only enhances safety but also significantly reduces the technical barriers for scaling up production, making it an ideal candidate for the cost reduction in pharmaceutical intermediate manufacturing.
As shown in the specific examples provided, the method consistently delivers N-acyl indole products with high structural fidelity, such as compounds I-1 through I-5, proving its utility for generating diverse chemical libraries. This operational simplicity not only enhances safety but also significantly reduces the technical barriers for scaling up production, making it an ideal candidate for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The core of this synthetic breakthrough lies in the intricate catalytic cycle driven by the palladium complex, which orchestrates the bond-forming events with high precision. The reaction initiates with the oxidative addition of the aryl iodide to the zero-valent palladium catalyst, forming a reactive aryl-palladium intermediate. Subsequently, carbon monoxide, which is slowly released in situ from the decomposition of the phenol 1,3,5-tricarboxylate surrogate, inserts into the palladium-carbon bond to generate an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, followed by reductive elimination to yield a linear amide precursor. 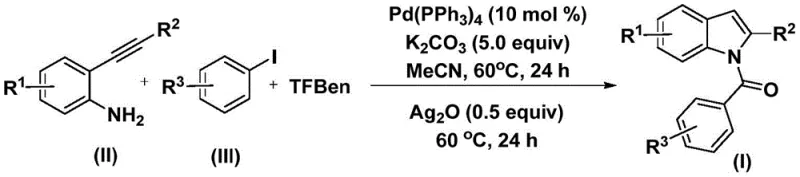 The final and crucial transformation involves the addition of silver oxide (Ag2O), which acts as a promoter to facilitate the intramolecular cyclization of the amide onto the alkyne moiety. This silver-mediated step ensures the efficient closure of the five-membered pyrrole ring, completing the formation of the N-acyl indole core. Understanding this mechanism is vital for R&D directors aiming to optimize reaction parameters, as the distinct two-stage temperature profile (both at 60°C) allows for the controlled evolution of the intermediates, minimizing side reactions and maximizing the yield of the desired heterocyclic product.
The final and crucial transformation involves the addition of silver oxide (Ag2O), which acts as a promoter to facilitate the intramolecular cyclization of the amide onto the alkyne moiety. This silver-mediated step ensures the efficient closure of the five-membered pyrrole ring, completing the formation of the N-acyl indole core. Understanding this mechanism is vital for R&D directors aiming to optimize reaction parameters, as the distinct two-stage temperature profile (both at 60°C) allows for the controlled evolution of the intermediates, minimizing side reactions and maximizing the yield of the desired heterocyclic product.
From an impurity control perspective, the use of a solid CO surrogate provides a steady, low-concentration supply of carbon monoxide, which helps prevent the formation of homocoupling byproducts that often plague gas-phase carbonylations. The choice of acetonitrile as the solvent further enhances the solubility of the polar intermediates and the inorganic bases, ensuring a homogeneous reaction environment that promotes consistent kinetics. The compatibility of the system with electron-withdrawing and electron-donating groups alike suggests that the electronic nature of the substrates does not severely inhibit the catalytic turnover, allowing for the synthesis of a wide array of analogues. This mechanistic robustness translates directly to a cleaner crude reaction profile, simplifying the downstream purification processes such as column chromatography or crystallization, which is a critical factor for maintaining high purity specifications in API production.
How to Synthesize N-Acyl Indole Compounds Efficiently
The practical implementation of this synthesis route is designed for ease of execution in standard laboratory and pilot plant settings, requiring no specialized high-pressure equipment. The protocol involves a straightforward charging of reagents followed by a controlled thermal sequence, making it highly accessible for process chemists looking to adopt this technology. For a detailed breakdown of the specific reagent ratios, solvent volumes, and workup procedures, please refer to the standardized synthesis guide below.
- Combine palladium catalyst, potassium carbonate, 1,3,5-tricarboxylic acid phenol ester, 2-alkynyl aniline, and aryl iodide in an organic solvent.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide to the mixture and continue heating at 60°C for another 24 hours to complete the cyclization into the N-acyl indole structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The shift from hazardous gaseous reagents to stable solid surrogates fundamentally alters the risk profile of the manufacturing process, leading to substantial cost savings in terms of safety infrastructure and regulatory compliance. By removing the dependency on high-pressure carbon monoxide cylinders or generators, facilities can operate with greater flexibility and reduced insurance liabilities, directly impacting the bottom line. Furthermore, the reliance on commercially available starting materials like 2-alkynyl anilines and aryl iodides ensures a resilient supply chain, as these precursors are widely produced and less susceptible to the bottlenecks often associated with specialty gases.
- Cost Reduction in Manufacturing: The elimination of expensive high-pressure reactors and the associated safety monitoring systems results in a drastic simplification of the production setup. Additionally, the use of inexpensive inorganic bases like potassium carbonate and the high atom efficiency of the one-pot process minimize waste generation, leading to lower disposal costs and improved overall process economics. The high reaction efficiency reported in the patent data suggests that less raw material is wasted on side products, further optimizing the cost per kilogram of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Sourcing stability is significantly improved because the key reagents, including the palladium catalyst and the CO surrogate, are stable solids with long shelf lives, unlike compressed gases which require complex logistics. This stability allows manufacturers to maintain leaner inventories without the risk of reagent degradation, ensuring continuous production capability even during global supply disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the rate of batch failures and ensuring consistent delivery schedules to downstream clients.
- Scalability and Environmental Compliance: The mild reaction temperature of 60°C and the use of acetonitrile, a common and recoverable solvent, make this process highly amenable to scale-up from gram to ton quantities. The absence of toxic gas emissions aligns perfectly with increasingly stringent environmental regulations, facilitating easier permitting for new production lines. The simplified post-treatment procedure, involving basic filtration and chromatography, reduces the consumption of silica gel and solvents during purification, contributing to a greener manufacturing footprint that appeals to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-acyl indole synthesis technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity for technical teams evaluating this route for potential integration into their manufacturing portfolios.
Q: What are the key advantages of this palladium-catalyzed method over traditional indole synthesis?
A: This method utilizes a mild one-pot carbonylation cyclization strategy that avoids the use of hazardous high-pressure carbon monoxide gas, replacing it with a solid CO surrogate (TFBen). It operates at moderate temperatures (60°C) and demonstrates excellent functional group tolerance, allowing for the synthesis of complex drug-like scaffolds without extensive protection-deprotection sequences.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process is highly scalable due to its operational simplicity and the use of commercially available reagents like tetrakis(triphenylphosphine)palladium and potassium carbonate. The elimination of high-pressure equipment requirements significantly lowers the barrier for commercial scale-up, making it an attractive route for industrial API production.
Q: What represents the critical role of Silver Oxide (Ag2O) in this reaction mechanism?
A: Silver oxide acts as a crucial promoter in the second stage of the reaction. After the initial formation of the amide intermediate, Ag2O facilitates the intramolecular cyclization step, effectively driving the conversion of the linear amide precursor into the final N-acyl indole heterocyclic core with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to stay competitive in the global pharmaceutical market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity N-acyl indole intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced catalytic technologies like the one described in CN112898192B, we can offer our partners a reliable supply of complex heterocyclic building blocks that are essential for the next generation of therapeutic agents.
We invite you to collaborate with us to explore how this efficient synthesis route can be tailored to your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis for your target molecules. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your drug development timelines while optimizing your manufacturing budget.
