Advanced One-Pot Synthesis of 1,4-Cyclohexanedimethanol Using Cobalt-Phosphine Catalysts
Introduction to Next-Generation CHDM Manufacturing
The global demand for high-performance polyesters, particularly those requiring enhanced thermal resistance and crystallinity, has driven intense research into advanced diol monomers. Patent CN107986943B introduces a transformative synthetic methodology for 1,4-cyclohexanedimethanol (CHDM), a critical building block for engineering plastics like PCT and PETG. Unlike conventional industrial routes that rely heavily on the hydrogenation of dimethyl terephthalate or terephthalic acid derivatives, this innovation leverages a sophisticated cobalt-catalyzed hydroformylation strategy starting from cyclohexene carboxaldehyde. This shift represents a significant paradigm change in polymer intermediate manufacturing, moving away from aromatic petrochemical feedstocks toward more versatile olefinic precursors. By integrating hydroformylation and hydrogenation into a streamlined process, the technology addresses long-standing inefficiencies in yield and separation, offering a compelling value proposition for reliable polymer intermediate suppliers seeking to optimize their production portfolios.
The technical breakthrough lies in the precise tuning of the catalytic system to achieve high regioselectivity. Traditional methods often struggle with isomeric mixtures that require energy-intensive distillation columns to separate the desired 1,4-isomer from the 1,3-isomer. The disclosed method utilizes a specialized organophosphine ligand system that sterically directs the addition of the formyl group, ensuring that the final product is predominantly the commercially valuable 1,4-cyclohexanedimethanol. This level of control not only enhances the purity profile required by discerning R&D Directors but also simplifies the overall mass balance of the plant. Furthermore, the avoidance of terephthalic acid mitigates exposure to the volatility of aromatic feedstock markets, providing a strategic hedge for procurement managers focused on long-term cost stability and supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 1,4-cyclohexanedimethanol has been dominated by routes originating from terephthalic acid (PTA) or dimethyl terephthalate (DMT). These traditional pathways involve the esterification of PTA followed by high-pressure catalytic hydrogenation. While established, this approach suffers from inherent thermodynamic and kinetic limitations. The oxidation of paraxylene to produce PTA is a harsh process that generates substantial amounts of carbon dioxide and other byproducts, creating significant environmental compliance burdens and waste treatment costs. Moreover, the hydrogenation of the aromatic ring in PTA derivatives requires extreme conditions and expensive noble metal catalysts, often leading to incomplete conversion or the formation of unwanted byproducts that complicate downstream purification. The reliance on PTA also ties the production of CHDM directly to the dynamics of the polyester fiber market, creating supply bottlenecks when demand for fibers spikes. For supply chain heads, this dependency introduces a single point of failure that can disrupt the availability of critical monomers for engineering plastic applications.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a hydroformylation-hydrogenation sequence starting from cyclohexene carboxaldehyde, which itself can be derived from the Diels-Alder reaction of acrolein and 1,3-butadiene. This route bypasses the aromatic ring entirely, constructing the six-membered ring through olefin chemistry which offers superior atom economy. The core innovation is the ability to couple the hydroformylation (adding a formyl group) and hydrogenation (reducing aldehyde to alcohol) steps, potentially in a one-pot operation. This integration drastically reduces the number of unit operations, eliminating the need for intermediate isolation and storage. By employing a cobalt-based catalyst system modified with bulky phosphine ligands, the process achieves high activity under relatively moderate temperatures (110-180°C) and pressures (5-10 MPa). This represents a substantial reduction in capital expenditure (CAPEX) for new plants and operational expenditure (OPEX) for existing facilities, directly addressing the need for cost reduction in polymer intermediate manufacturing.
Mechanistic Insights into Cobalt-Phosphine Catalyzed Hydroformylation
The heart of this synthetic advancement is the synergistic interaction between the cobalt metal center and the specialized organophosphine ligand. The catalyst system typically employs a hydrogenated polycarbonyl cobalt species generated in situ from precursors such as Co2(CO)8 or C3H7CCo3(CO)9. However, unmodified cobalt carbonyls often lack the necessary selectivity for complex substrates like cyclohexene carboxaldehyde. The introduction of a tri-substituted alkyl cyclohexylphosphine ligand fundamentally alters the electronic and steric properties of the active catalytic species. The phosphine ligand acts as a strong electron donor, increasing the electron density on the cobalt atom. This electronic modification enhances the hydridic character of the cobalt-hydrogen bond, thereby accelerating the hydrogenation step which is often the rate-limiting factor in converting aldehydes to alcohols. Simultaneously, the bulky nature of the ligand prevents the formation of inactive cobalt clusters, stabilizing the catalyst against decomposition under the rigorous conditions of syngas processing.
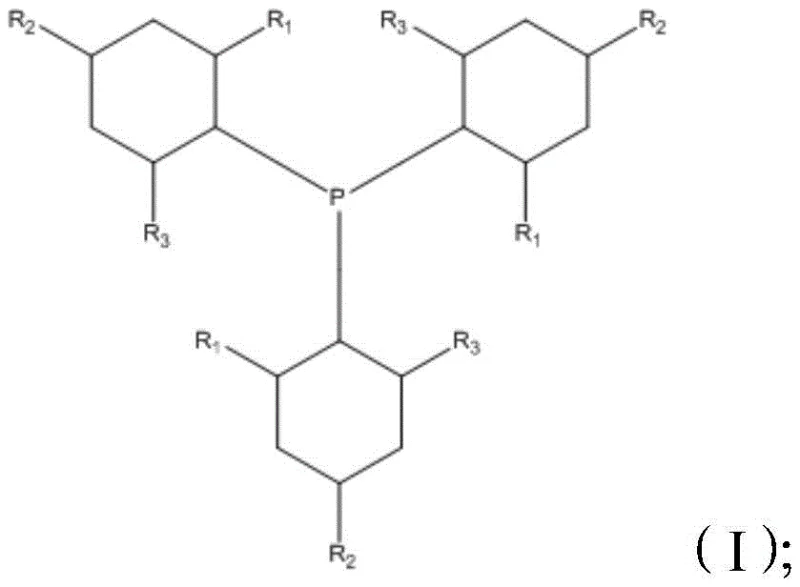
As illustrated in the structural formula, the ligand features significant steric bulk due to the presence of tert-butyl groups on the cyclohexyl rings. This steric congestion is not merely a passive feature but an active design element that governs the regioselectivity of the hydroformylation. When the cyclohexene carboxaldehyde coordinates to the cobalt center, the bulky ligand shields certain approach vectors, effectively forcing the incoming syngas (CO/H2) to react at the less hindered position—specifically, the carbon atom opposite to the existing aldehyde group. This steric steering mechanism suppresses the formation of the 1,3-isomer and other branched byproducts, ensuring that the reaction pathway funnels efficiently toward the desired 1,4-cyclohexane dicarbaldehyde intermediate, which is subsequently hydrogenated to the final diol. For technical teams, understanding this ligand-metal conical angle is crucial for replicating the high yields (>88% GC yield reported in examples) and minimizing the load on purification columns.
Furthermore, the choice of solvent plays a pivotal role in maintaining catalyst stability and substrate solubility. The patent highlights the efficacy of using the reaction products themselves, such as cyclohexanedimethanol or intermediate aldehydes, as part of the solvent matrix. This "self-solvating" approach minimizes the introduction of extraneous impurities that could poison the sensitive cobalt catalyst or complicate the final distillation. It also simplifies the recycling of unreacted materials, contributing to a greener process profile. The ability to operate without additional transition metal promoters or harsh acidic conditions further underscores the robustness of this catalytic cycle, making it highly attractive for continuous flow manufacturing environments where catalyst longevity is paramount.
How to Synthesize 1,4-Cyclohexanedimethanol Efficiently
Implementing this synthesis route requires careful attention to the preparation of the active catalyst species and the control of reaction parameters. The process begins with the generation of the hydrogenated cobalt polycarbonyl complex, typically achieved by treating a cobalt carbonyl precursor with synthesis gas in the presence of the phosphine ligand. Once the active catalyst is formed, the cyclohexene carboxaldehyde substrate is introduced. The reaction is then driven by maintaining a specific partial pressure of CO and H2, usually in a ratio of 1:2 to 2:1, at temperatures ranging from 120°C to 180°C. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to ensure reproducibility and safety.
- Prepare the active cobalt catalyst by hydrogenating a cobalt carbonyl precursor (e.g., C3H7CCo3(CO)9) in the presence of a bulky trialkylphosphine ligand.
- Introduce cyclohexene carboxaldehyde (derived from acrolein and 1,3-butadiene) into the reactor containing the catalyst and solvent mixture.
- Maintain reaction conditions at 110-180°C and 5-10 MPa under a synthesis gas (CO/H2) atmosphere to effect simultaneous hydroformylation and hydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain leaders, the adoption of this cobalt-catalyzed route offers multifaceted benefits that extend beyond simple yield improvements. The most significant advantage is the decoupling of CHDM production from the terephthalic acid supply chain. PTA is a high-volume commodity subject to massive fluctuations in price driven by the textile industry's demand for PET. By shifting to a feedstock base of acrolein and butadiene—chemicals with different market drivers—manufacturers can stabilize their raw material costs and mitigate the risk of supply shortages. This diversification is a critical risk management strategy for any organization aiming for long-term operational continuity in the volatile specialty chemicals sector.
- Cost Reduction in Manufacturing: The elimination of the aromatic hydrogenation step, which traditionally requires expensive noble metal catalysts (like ruthenium or rhodium) and extreme pressures, results in significant operational savings. Cobalt is substantially more abundant and cost-effective than noble metals, and the ligand system described allows for lower catalyst loading while maintaining high activity. Additionally, the potential for a one-pot process reduces the number of reactors, separators, and drying units required, leading to lower utility consumption (steam and electricity) and reduced maintenance overheads. The simplified workflow also translates to lower labor costs and reduced downtime between batches, driving down the overall cost of goods sold (COGS) without compromising on quality.
- Enhanced Supply Chain Reliability: The raw materials for this process, specifically acrolein and 1,3-butadiene, are bulk petrochemicals produced in vast quantities globally, ensuring a robust and redundant supply network. Unlike niche intermediates that might be sourced from a single supplier, these feedstocks are available from multiple integrated refineries, reducing the risk of logistics disruptions. Furthermore, the high selectivity of the catalyst means that less raw material is wasted on byproducts, improving the overall material efficiency of the plant. This efficiency allows manufacturers to meet increased demand without proportionally increasing their raw material intake, providing a buffer against upstream supply shocks.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior to the traditional PTA method. It avoids the generation of large volumes of CO2 associated with the oxidation of xylenes to terephthalic acid. The atom economy of the hydroformylation reaction is inherently high, meaning fewer waste streams are generated per kilogram of product. The use of recyclable solvents and the absence of heavy metal contaminants in the final product simplify wastewater treatment and reduce the regulatory burden. This alignment with green chemistry principles facilitates easier permitting for capacity expansions and enhances the sustainability profile of the final polymer products, a key selling point for downstream customers in the automotive and electronics sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent literature, providing a factual basis for feasibility assessments.
Q: What is the primary advantage of this cobalt-catalyzed route over traditional PTA-based methods?
A: The primary advantage is the complete avoidance of terephthalic acid (PTA) as a raw material. Traditional PTA routes involve energy-intensive oxidation steps with significant CO2 emissions and difficult separation processes. This novel olefin-based route utilizes abundant bulk chemicals like acrolein and butadiene, offering a more atom-economical and environmentally friendly pathway with simplified downstream purification.
Q: How does the specific phosphine ligand improve selectivity for 1,4-CHDM?
A: The use of bulky trialkylphosphine ligands, such as tris(2,4-di-tert-butylcyclohexyl)phosphine, creates a specific steric environment around the cobalt center. This steric hindrance inhibits the formation of isomeric aldehydes during the hydroformylation step, directing the reaction preferentially to the carbon atom opposite the existing aldehyde group. This results in high selectivity for the 1,4-isomer, which is critical for producing high-performance polyesters.
Q: Can this process be scaled for industrial production?
A: Yes, the process is designed for scalability. It operates as a one-pot reaction where hydroformylation and hydrogenation occur sequentially or simultaneously in the same reactor. This eliminates the need for intermediate isolation steps, reducing equipment investment and operational complexity. The use of robust cobalt catalysts and standard high-pressure reactor conditions (3-30 MPa) aligns well with existing industrial infrastructure for fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Cyclohexanedimethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-purity monomers like 1,4-cyclohexanedimethanol in the production of next-generation engineering plastics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial realities. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chromatographic systems to guarantee that every batch meets the exacting standards required for polymerization. Our commitment to quality assurance ensures that our CHDM delivers consistent performance in your final polyester formulations, minimizing defects and maximizing material properties.
We invite you to collaborate with us to leverage this advanced cobalt-catalyzed technology for your supply chain. Whether you require a Customized Cost-Saving Analysis to evaluate the economic viability of switching from PTA-based routes or need specific COA data to validate our product against your internal specifications, our technical procurement team is ready to assist. Contact us today to request route feasibility assessments and discover how our optimized manufacturing processes can drive value and reliability for your organization.
