Revolutionizing Indolo[3,2-c]quinoline Production: A Cost-Effective Copper-Catalyzed Strategy for Global Supply Chains
Introduction to Advanced Indoloquinoline Synthesis
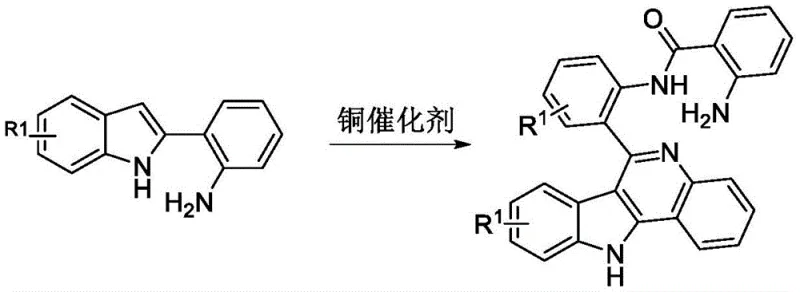
The pharmaceutical industry is constantly seeking robust and economically viable pathways for constructing complex heterocyclic scaffolds, particularly those with proven biological activity. Patent CN114621220B introduces a transformative approach to synthesizing indolo[3,2-c]quinoline compounds, a class of molecules renowned for their potent antimalarial and anticancer properties. This intellectual property details a novel methodology that leverages inexpensive copper salts as catalysts under aerobic conditions, marking a significant departure from traditional precious metal-dependent processes. By utilizing readily available 2-(2-aminophenyl)indole derivatives as starting materials, this technology addresses critical pain points in modern drug development, including high catalyst costs and harsh reaction environments. For R&D directors and procurement specialists, this represents a strategic opportunity to optimize the supply chain for high-value active pharmaceutical ingredients (APIs) and their intermediates.
The significance of this patent extends beyond mere academic interest; it offers a tangible solution for cost reduction in pharmaceutical intermediate manufacturing. The described synthesis operates at moderate temperatures ranging from 70 to 90 degrees Celsius and employs air as the sole oxidant, thereby eliminating the need for hazardous or expensive stoichiometric oxidizing agents. This green chemistry approach not only aligns with increasingly stringent environmental regulations but also simplifies the downstream purification process. As we delve deeper into the technical specifics, it becomes evident that this copper-catalyzed oxidative cyclization is poised to become a preferred route for the commercial production of bioactive indoloquinolines, offering a reliable alternative for global supply chains.
![General molecular structure of the benzamido-containing indolo[3,2-c]quinoline compound](/insights/img/indolo-quinoline-synthesis-copper-catalysis-pharma-supplier-20260304074851-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indolo[3,2-c]quinoline core has relied heavily on transition metal catalysis involving palladium or gold complexes. While effective in laboratory settings, these conventional methods suffer from severe drawbacks when evaluated through the lens of industrial scalability and cost efficiency. Palladium catalysts are notoriously expensive and subject to volatile market pricing, which introduces significant financial risk into long-term production contracts. Furthermore, many existing literature methods require harsh reaction conditions, such as high temperatures or the use of toxic isocyanides, which pose safety hazards and complicate regulatory compliance. The narrow substrate scope often reported in these older methodologies further limits their utility, forcing process chemists to redesign synthetic routes for even minor structural modifications, thereby delaying project timelines and inflating R&D budgets.
The Novel Approach
In stark contrast, the methodology disclosed in CN114621220B presents a streamlined and economically superior alternative. The core innovation lies in the use of copper salts, such as copper bromide or copper chloride, which are orders of magnitude cheaper than their noble metal counterparts. This switch fundamentally alters the cost structure of the synthesis, making it highly attractive for large-scale manufacturing. The reaction proceeds smoothly in common organic solvents like ethanol or dimethyl sulfoxide under an air atmosphere, removing the necessity for inert gas protection or specialized high-pressure equipment. This simplicity translates directly into operational efficiency, allowing for easier scale-up and reduced capital expenditure on reactor infrastructure. The broad applicability of this method across various substituted substrates ensures that it can serve as a versatile platform technology for generating diverse chemical libraries.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between the copper catalyst and the organic substrate under aerobic conditions. Initially, the copper salt coordinates with the amine functionality of the 2-(2-aminophenyl)indole precursor, activating the molecule towards oxidation. In the presence of molecular oxygen from the air, the copper center facilitates the removal of hydrogen atoms, generating reactive radical or cationic intermediates that are primed for cyclization. This oxidative process drives the formation of the new carbon-nitrogen bond that closes the quinoline ring, effectively fusing the indole and benzene moieties into the rigid planar structure characteristic of indoloquinolines. The regeneration of the active copper species by atmospheric oxygen completes the catalytic cycle, ensuring that only catalytic amounts of metal are required to drive the reaction to completion.
From an impurity control perspective, this mechanism offers distinct advantages over non-catalytic thermal cyclizations. The specificity of the copper catalyst minimizes side reactions such as polymerization or over-oxidation, which are common pitfalls in high-temperature processes. The mild reaction window of 70 to 90 degrees Celsius further suppresses the formation of thermal degradation products, resulting in a cleaner crude reaction profile. This high selectivity is crucial for pharmaceutical applications, where strict limits on genotoxic impurities and heavy metal residues must be met. By understanding these mechanistic nuances, process engineers can fine-tune parameters like stirring speed and aeration rates to maximize yield and purity, ensuring that the final product meets the rigorous quality standards demanded by regulatory agencies worldwide.
How to Synthesize Benzamido-containing Indolo[3,2-c]quinoline Efficiently
The practical execution of this synthesis is divided into two logical stages: the preparation of the key indole intermediate and the subsequent copper-catalyzed cyclization. The first stage involves the condensation of o-aminoacetophenone with phenylhydrazine derivatives, followed by an acid-mediated cyclization to yield the 2-(2-aminophenyl)indole scaffold. This precursor is then subjected to the oxidative conditions described previously. The simplicity of the workup procedure, involving basic washing and standard chromatographic separation, makes this protocol accessible even to laboratories with modest equipment. For a detailed breakdown of the specific reagent quantities, temperature profiles, and purification techniques, please refer to the standardized guide below.
- Prepare the 2-(2-aminophenyl)indole precursor by reacting o-aminoacetophenone with phenylhydrazine derivatives in ethanol/acetic acid, followed by acid-mediated cyclization.
- Perform the key oxidative cyclization using the indole precursor and a copper salt catalyst (e.g., CuBr2) in an organic solvent under an air atmosphere at 70-90°C.
- Purify the crude reaction mixture by washing with sodium carbonate, extracting with ethyl acetate, and separating via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed technology offers compelling strategic benefits that extend far beyond the laboratory bench. The primary advantage lies in the drastic reduction of raw material costs associated with the catalyst system. By replacing scarce and expensive palladium or gold with abundant copper, manufacturers can significantly lower the bill of materials for each batch produced. This cost efficiency is compounded by the use of air as the oxidant, which is essentially free and eliminates the logistical burden of storing and handling hazardous chemical oxidizers. These factors combine to create a more resilient and cost-effective supply chain, shielding the organization from the volatility of precious metal markets and ensuring consistent production margins.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes a major cost driver from the production budget. Copper salts are commodity chemicals with stable pricing and widespread availability, ensuring that procurement teams can secure long-term supply contracts without fear of sudden price spikes. Additionally, the mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure alternatives, further contributing to overall operational savings. The simplified purification process also reduces solvent usage and waste disposal costs, enhancing the economic viability of the entire manufacturing lifecycle.
- Enhanced Supply Chain Reliability: The reliance on easily sourced starting materials like o-aminoacetophenone and phenylhydrazine derivatives ensures a robust supply chain foundation. These commodities are produced by numerous global suppliers, reducing the risk of single-source dependency and supply disruptions. The robustness of the reaction conditions means that production can be maintained consistently across different manufacturing sites without requiring highly specialized infrastructure. This flexibility allows supply chain planners to diversify their manufacturing network, improving continuity of supply for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The use of green oxidants (air) and benign solvents aligns perfectly with modern environmental, health, and safety (EHS) standards. Scaling this process does not introduce significant new hazards, as the reaction does not involve explosive reagents or extreme pressures. This ease of scale-up facilitates the rapid transition from pilot plant to commercial production, shortening the time-to-market for new drug candidates. Furthermore, the reduced environmental footprint simplifies regulatory approvals and enhances the company's sustainability profile, which is increasingly important for stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when adopting this route. Understanding these details is essential for making informed decisions about process integration and resource allocation.
Q: What are the primary economic advantages of this copper-catalyzed method over traditional palladium routes?
A: The substitution of expensive palladium or gold catalysts with abundant and low-cost copper salts drastically reduces raw material costs. Furthermore, the use of air as the terminal oxidant eliminates the need for costly stoichiometric oxidants, simplifying waste treatment and lowering overall production expenses.
Q: Does this synthetic route support diverse substrate modifications for drug discovery?
A: Yes, the patent demonstrates excellent functional group tolerance. The method successfully accommodates various substituents such as fluorine, chlorine, and methyl groups on the aromatic rings, allowing medicinal chemists to rapidly generate diverse libraries of indoloquinoline derivatives for SAR studies without changing the core reaction conditions.
Q: Is the process scalable for commercial manufacturing of pharmaceutical intermediates?
A: The process utilizes standard organic solvents like ethanol and operates at moderate temperatures (70-90°C) under ambient air pressure, avoiding high-pressure hydrogenation or cryogenic conditions. These mild parameters, combined with simple workup procedures involving extraction and chromatography, indicate high potential for safe and efficient scale-up to multi-kilogram or ton-level production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolo[3,2-c]quinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of the indolo[3,2-c]quinoline scaffold in next-generation therapeutics. Our team of expert process chemists has thoroughly analyzed the methodology described in CN114621220B and is fully prepared to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from clinical trials to full-scale market launch. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate we deliver adheres to the highest international quality standards.
We invite you to collaborate with us to leverage this cost-effective and scalable synthesis route for your drug development programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can accelerate your path to market while optimizing your production costs. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing.
