Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the demand for safer, more efficient, and environmentally benign synthetic routes. A pivotal advancement in this domain is detailed in patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. These structures are critical pharmacophores found in numerous bioactive molecules, including potent β3 adrenergic receptor agonists and various agrochemical agents. The introduction of the trifluoromethyl group is particularly strategic, as it markedly enhances the metabolic stability, lipophilicity, and electronegativity of the parent molecule, thereby improving bioavailability. This patent represents a paradigm shift away from hazardous traditional methodologies, offering a robust platform for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
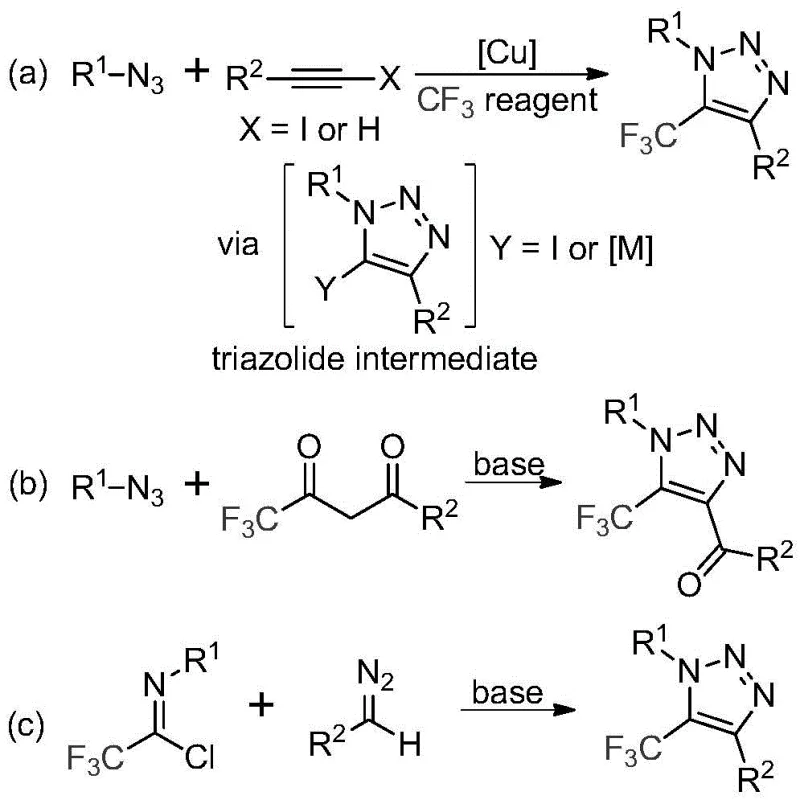
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed azide-alkyne cycloadditions or organocatalytic reactions involving trifluoromethyl ketones. As illustrated in the prior art comparison, these conventional pathways present substantial hurdles for industrial application. The primary concern is the reliance on organic azides, which are notoriously toxic and possess high explosive potential, posing severe safety risks during storage and handling on a large scale. Furthermore, copper-catalyzed variants necessitate the use of transition metals, which introduces the complex and costly challenge of removing heavy metal residues to meet stringent regulatory limits for active pharmaceutical ingredients (APIs). These factors collectively inflate production costs and complicate the supply chain logistics for high-purity intermediates.
The Novel Approach
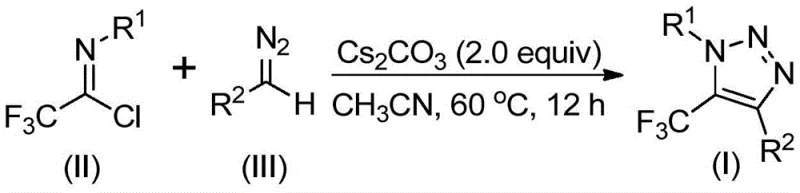
In stark contrast to these legacy techniques, the methodology described in CN113121462B utilizes a base-promoted cyclization between readily available trifluoroethylimidoyl chlorides and diazo compounds. This innovative strategy completely eliminates the need for transition metal catalysts and avoids the use of unstable azide reagents. The reaction proceeds under mild thermal conditions, typically between 50°C and 70°C, utilizing cesium carbonate as an inexpensive and effective promoter. By shifting the synthetic logic to a metal-free, azide-free protocol, this approach not only mitigates safety hazards but also drastically simplifies the downstream purification process. The result is a streamlined workflow that facilitates the efficient construction of diverse triazole scaffolds with excellent functional group tolerance.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation offers fascinating insights into the reactivity of diazo species under basic conditions. The reaction is hypothesized to initiate with a base-promoted intermolecular nucleophilic addition-elimination process. Specifically, the diazo compound acts as a nucleophile attacking the electrophilic carbon of the trifluoroethylimidoyl chloride. This step promotes the formation of a critical carbon-carbon bond while eliminating a chloride ion. Following this initial coupling, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This cyclization step is energetically favorable and leads directly to the formation of the five-membered 1,2,3-triazole ring system. The presence of the trifluoromethyl group at the 5-position is retained throughout this sequence, ensuring the final product possesses the desired electronic properties essential for biological activity.
From an impurity control perspective, this mechanism is advantageous because it avoids the formation of metal-complexed side products that are common in copper-catalyzed variants. The use of cesium carbonate ensures a homogeneous reaction environment in polar aprotic solvents like acetonitrile, which enhances the solubility of ionic intermediates and drives the reaction to completion. The broad substrate scope allows for significant structural diversity; R1 groups can range from simple phenyl rings to substituted aryls with electron-donating or withdrawing groups, while R2 can accommodate esters, ketones, or phosphonates. This versatility makes the method highly attractive for medicinal chemists seeking to rapidly generate libraries of analogues for structure-activity relationship (SAR) studies without being constrained by harsh reaction conditions.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The operational simplicity of this synthesis makes it highly amenable to both laboratory-scale optimization and industrial manufacturing. The protocol requires standard equipment and does not demand specialized high-pressure or cryogenic setups. Typically, the reactants are combined in an organic solvent such as acetonitrile, toluene, or 1,4-dioxane, with acetonitrile proving to be the optimal medium for maximizing conversion rates. The reaction mixture is heated to approximately 60°C and maintained for 8 to 16 hours. Upon completion, the workup involves straightforward filtration to remove inorganic salts, followed by concentration and purification via column chromatography. For those seeking a standardized operational procedure to implement this technology immediately, the detailed steps are outlined below.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C (optimally 60°C) and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits. The elimination of explosive azides significantly reduces the regulatory burden and insurance costs associated with hazardous material handling. Moreover, the absence of transition metals means that manufacturers can bypass expensive scavenging processes and rigorous heavy metal testing, leading to substantial cost savings in the overall production budget. The use of commodity chemicals like cesium carbonate and commercially available diazo precursors ensures a stable and reliable supply chain, reducing the risk of raw material shortages that often plague specialty reagent markets.
- Cost Reduction in Manufacturing: The removal of copper catalysts eliminates the need for costly ligands and the subsequent purification steps required to reduce metal content to ppm levels. This simplification of the downstream processing directly translates to lower operational expenditures. Additionally, the high atom economy and efficient conversion rates minimize waste generation, further reducing disposal costs and enhancing the overall sustainability profile of the manufacturing process.
- Enhanced Supply Chain Reliability: By relying on stable, non-explosive starting materials, the process mitigates the risks associated with the storage and transport of hazardous azides. This stability allows for larger batch sizes and more flexible inventory management, ensuring consistent delivery schedules for downstream API manufacturers. The robustness of the reaction conditions also means that production is less susceptible to minor fluctuations in environmental parameters, guaranteeing consistent quality output.
- Scalability and Environmental Compliance: The mild reaction temperatures and ambient pressure conditions make this process inherently scalable from gram to multi-ton quantities without requiring significant engineering modifications. The reduced toxicity profile aligns well with increasingly stringent environmental regulations, facilitating easier permitting and compliance auditing. This green chemistry approach positions manufacturers favorably in markets where sustainability credentials are becoming a key differentiator for supplier selection.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: Why is this new synthesis method safer than traditional triazole production?
A: Traditional methods often rely on organic azides, which are toxic and potentially explosive. This patented route utilizes stable diazo compounds and imidoyl chlorides, eliminating high-risk reagents and enhancing operational safety.
Q: Does this process require expensive transition metal catalysts?
A: No, the process is entirely metal-free. It uses cesium carbonate as a promoter, which removes the need for costly copper catalysts and the subsequent complex purification steps required to remove heavy metal residues.
Q: What is the substrate scope for R1 and R2 groups in this reaction?
A: The method demonstrates broad functional group tolerance. R1 can be alkyl or various substituted aryl groups, while R2 accommodates aroyl, phospholipid, alkoxycarbonyl, or trifluoromethyl groups, allowing for diverse molecular scaffolding.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement metal-free and azide-free processes underscores our dedication to safety, quality, and environmental stewardship.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Whether you require custom synthesis services or bulk supply of 5-trifluoromethyl-1,2,3-triazole derivatives, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific needs. Contact us today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for the future.
