Revolutionizing Amlodipine Intermediate Production with Safe Lewis Acid Catalysis and Scalable Manufacturing
Revolutionizing Amlodipine Intermediate Production with Safe Lewis Acid Catalysis and Scalable Manufacturing
The pharmaceutical industry constantly seeks robust synthetic pathways that balance high purity with operational safety, particularly for high-volume cardiovascular drugs like amlodipine. Patent CN111303006A introduces a transformative preparation method for a critical amlodipine intermediate, specifically ethyl (2-(1,3-dioxoisoindolin-2-yl)ethoxy)-3-oxobutanoate. This innovation addresses long-standing safety hazards associated with traditional sodium hydride (NaH) mediated alkylations by employing a sophisticated two-step sequence involving Swern oxidation followed by Lewis acid-catalyzed C-H bond insertion. For R&D directors and procurement managers, this patent represents a pivotal shift towards safer, more cost-effective manufacturing protocols that eliminate pyrophoric reagents while utilizing abundant, low-cost starting materials like diethylene glycol amine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of this key intermediate has relied heavily on a route originating from phthalic anhydride, proceeding through condensation and oxyalkylation steps that necessitate the use of substantial quantities of sodium hydride. As illustrated in the conventional pathway below, the reliance on NaH creates a bottleneck for commercial scale-up due to its extreme pyrophoric nature; contact with moist air can lead to spontaneous combustion, forcing manufacturers to restrict reactor sizes to 1000L or smaller to mitigate explosion risks. Furthermore, the precursor ethyl 4-chloroacetoacetate is derived from chlorine and diketene, raw materials subject to stringent transportation regulations and significant price volatility, leading to unstable supply chains and inflated production costs that hinder the ability to act as a reliable pharmaceutical intermediate supplier.
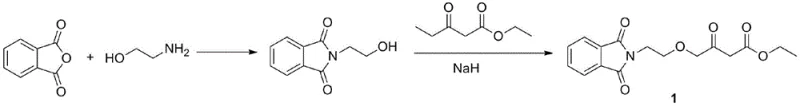
The Novel Approach
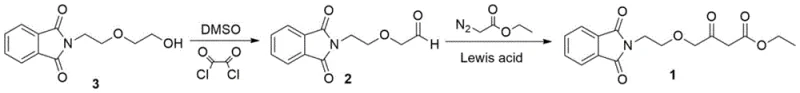
In stark contrast, the methodology disclosed in CN111303006A circumvents these dangers by utilizing a hydroxyethyl isoindoline derivative (Compound 3) which is economically synthesized from phthalic anhydride and diethylene glycol amine, a cheap by-product of morpholine production. The new process replaces the dangerous alkylation with a controlled Swern oxidation to generate an aldehyde intermediate, followed by a catalytic C-H insertion reaction with ethyl diazoacetate. This strategic pivot not only removes the fire hazard associated with NaH but also stabilizes the raw material supply chain, as the new precursors are readily available and do not require special hazardous material handling, thereby facilitating the commercial scale-up of complex pharmaceutical intermediates without the weather-dependent production stoppages seen in older facilities.
Mechanistic Insights into Swern Oxidation and Lewis Acid Catalyzed C-H Insertion
The core of this technological breakthrough lies in the precise execution of the Swern oxidation followed by a metal-catalyzed carbene insertion. In the first stage, Compound 3 reacts with dimethyl sulfoxide (DMSO) and oxalyl chloride at cryogenic temperatures ranging from -78°C to -60°C to form an activated sulfonium species, which subsequently converts the primary alcohol into the corresponding aldehyde (Compound 2) upon treatment with a base like triethylamine. This mild oxidation protocol is crucial for preserving the integrity of the sensitive isoindoline ring system while ensuring high conversion rates, typically exceeding 90% under optimized solvent conditions such as dichloromethane or acetonitrile, providing a clean substrate for the subsequent carbon-carbon bond forming step.
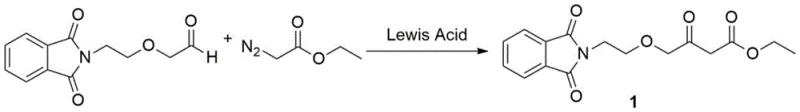
The second stage involves the reaction of the generated aldehyde with ethyl diazoacetate in the presence of a Lewis acid catalyst, such as SnCl2, ZnCl2, or AlCl3, at mild temperatures between 10°C and 30°C. The Lewis acid coordinates with the diazo compound to generate a metal-carbenoid species, which then undergoes a highly selective C-H insertion into the formyl group of the aldehyde. As detailed in the reaction scheme below, this mechanism avoids the harsh basic conditions that cause decomposition in traditional routes, allowing for excellent control over regioselectivity and minimizing side reactions, ultimately delivering the target ester with exceptional purity suitable for downstream API synthesis.
How to Synthesize Amlodipine Intermediate Efficiently
To implement this advanced synthesis in a GMP environment, operators must strictly adhere to the temperature profiles and molar ratios defined in the patent to maximize yield and safety. The process begins with the activation of DMSO at low temperatures to prevent exothermic runaway, followed by the careful addition of the alcohol substrate. Once the aldehyde is formed, the reaction mixture is warmed, and the Lewis acid catalyst is introduced to facilitate the coupling with ethyl diazoacetate. Detailed standardized synthesis steps see the guide below.
- Perform Swern oxidation on compound 3 using DMSO and oxalyl chloride at low temperatures (-78°C to -60°C) to generate the aldehyde intermediate.
- React the resulting aldehyde with ethyl diazoacetate in the presence of a Lewis acid catalyst (such as SnCl2 or ZnCl2) at mild temperatures (10°C to 30°C).
- Purify the final product through standard aqueous workup and solvent removal to obtain the high-purity amlodipine intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patent offers profound logistical and financial benefits by fundamentally altering the risk profile of the manufacturing process. By eliminating the need for sodium hydride, facilities can utilize larger reactor vessels without the fear of pyrophoric incidents, effectively increasing batch sizes and throughput capacity while reducing the unit cost of production through better equipment utilization. Additionally, the substitution of volatile chloroacetoacetate with stable, commodity-grade diethylene glycol amine insulates the supply chain from the regulatory bottlenecks and price spikes associated with chlorine-based chemistry, ensuring consistent delivery schedules for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like NaH and the switch to cheaper bulk chemicals significantly lowers the direct material costs, while the improved safety profile reduces insurance premiums and safety infrastructure investments. The patent explicitly notes a total cost reduction of approximately 30 percent compared to prior art, driven by both raw material savings and the ability to run continuous, uninterrupted campaigns without weather-related shutdowns.
- Enhanced Supply Chain Reliability: Sourcing diethylene glycol amine is far more stable than relying on ethyl 4-chloroacetoacetate, which suffers from upstream supply constraints due to the regulated nature of its precursors. This shift ensures that production schedules are not disrupted by raw material shortages, allowing manufacturers to maintain steady inventory levels and meet the rigorous just-in-time delivery demands of global API producers.
- Scalability and Environmental Compliance: The new route generates less hazardous waste compared to the NaH route, which produces hydrogen gas and requires careful quenching procedures. Furthermore, avoiding the use of heavy metals or toxic chlorinating agents in the final steps simplifies wastewater treatment and aligns with increasingly strict environmental regulations, making it easier to obtain permits for capacity expansion and commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route, based on the specific experimental data and comparative analysis provided in the patent documentation. These insights are designed to clarify the operational feasibility and economic impact of transitioning from legacy methods to this modern catalytic approach.
Q: Why is the new Lewis acid catalyzed route safer than the traditional NaH method?
A: The traditional method relies on sodium hydride (NaH), which is pyrophoric and poses severe fire risks, limiting reactor sizes to 1000L. The new route eliminates NaH entirely, using stable reagents like DMSO and oxalyl chloride, allowing for safe scaling to 100 MT production.
Q: What are the cost advantages of using diethylene glycol amine over chloroacetoacetate?
A: Diethylene glycol amine is an abundant by-product of morpholine production, making it significantly cheaper and more stable in supply compared to ethyl 4-chloroacetoacetate, whose precursors (chlorine and diketene) face strict transportation regulations and price volatility.
Q: Which Lewis acid catalysts provide the highest yield for this C-H insertion reaction?
A: Experimental data indicates that tin(II) chloride (SnCl2) and aluminum chloride (AlCl3) are highly effective, with SnCl2 achieving yields up to 94.9% under optimized conditions in DCE solvent at 25°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amlodipine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition to this Lewis acid-catalyzed process is seamless and compliant with international quality standards. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of amlodipine intermediate meets the exacting requirements of our partners, minimizing impurities and maximizing downstream reaction efficiency.
We invite you to collaborate with us to leverage this patented technology for your supply chain needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this innovative method can enhance your production capabilities and reduce overall manufacturing costs.
