Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Pharmaceutical Manufacturing
Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Pharmaceutical Manufacturing
The rapid evolution of medicinal chemistry demands increasingly sophisticated heterocyclic scaffolds, particularly those incorporating fluorine motifs to enhance metabolic stability and bioavailability. A groundbreaking development in this sector is detailed in patent CN113121462B, which discloses a highly efficient preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift from traditional transition-metal catalyzed processes to a streamlined, base-promoted cyclization strategy. For R&D directors and procurement specialists alike, this innovation offers a compelling solution to the longstanding challenges of safety, cost, and scalability in producing these vital pharmacophores. The ability to construct these complex nitrogen-containing heterocycles without relying on hazardous azides or expensive copper catalysts positions this methodology as a cornerstone for next-generation API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed azide-alkyne cycloaddition (CuAAC) reactions or organocatalytic 1,3-dipolar cycloadditions involving trifluoromethyl ketones. As illustrated in the comparative reaction schemes below, these conventional pathways present significant logistical and safety hurdles for industrial application. The reliance on organic azides is particularly problematic due to their inherent toxicity and potential for explosive decomposition, necessitating specialized handling protocols and rigorous safety engineering controls that drive up capital expenditure. Furthermore, the use of copper catalysts introduces the risk of heavy metal contamination in the final product, requiring additional purification steps such as scavenging or recrystallization to meet stringent regulatory limits for residual metals in pharmaceutical ingredients.
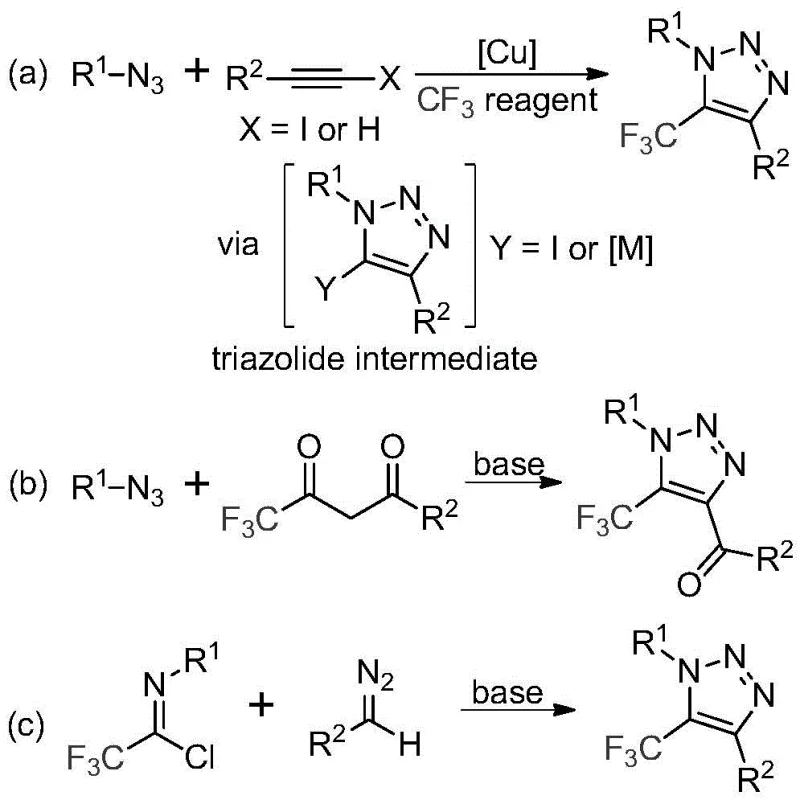
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in the patent utilizes a metal-free, azide-free strategy that leverages readily available diazo compounds and trifluoroethylimidoyl chlorides. This base-promoted reaction proceeds under mild thermal conditions, typically between 50°C and 70°C, utilizing cesium carbonate as a benign promoter. The reaction scheme depicted below highlights the elegance of this transformation, where the carbon-carbon bond formation and subsequent ring closure occur seamlessly without the need for transition metals. This not only mitigates the safety risks associated with azide chemistry but also simplifies the downstream processing workflow. By eliminating the catalyst removal step, manufacturers can achieve significant reductions in production time and waste generation, thereby enhancing the overall sustainability and economic viability of the synthesis process for high-purity pharmaceutical intermediates.
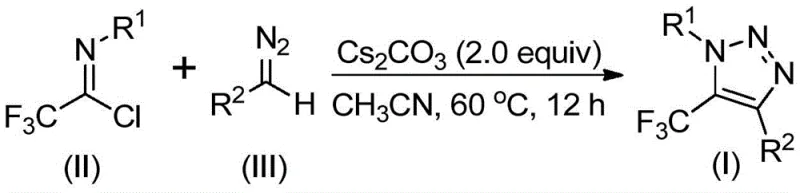
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this transformation offers profound insights into its efficiency and selectivity, which are critical for R&D teams optimizing process parameters. The reaction is hypothesized to initiate with a base-promoted intermolecular nucleophilic addition-elimination process, where the diazo compound attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This initial step facilitates the formation of a key carbon-carbon bond, setting the stage for the subsequent ring closure. The presence of the electron-withdrawing trifluoromethyl group enhances the electrophilicity of the imidoyl chloride, thereby accelerating the nucleophilic attack and ensuring high conversion rates even under relatively mild conditions. This mechanistic understanding allows chemists to fine-tune the electronic properties of the substrates to maximize yield and minimize side reactions.
Following the initial addition, the intermediate undergoes an intramolecular 5-endo-dig cyclization to generate the final 5-trifluoromethyl substituted 1,2,3-triazole core. This specific cyclization mode is favored by the geometric arrangement of the intermediate and the stabilization provided by the conjugated system. The use of cesium carbonate is pivotal here, as it acts not only as a base to deprotonate the diazo species but also potentially stabilizes the transition state through cation-pi interactions or coordination with the nitrogen atoms. This dual role ensures that the reaction proceeds with high regioselectivity, avoiding the formation of unwanted isomers that often plague triazole syntheses. Such precise control over the reaction trajectory is essential for maintaining a clean impurity profile, which is a top priority for regulatory compliance in drug substance manufacturing.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to ensure reproducibility and safety. The protocol involves combining the trifluoroethylimidoyl chloride and the diazo compound in an aprotic organic solvent, with acetonitrile being the preferred medium due to its ability to dissolve reactants effectively while promoting the reaction kinetics. The addition of molecular sieves is recommended to maintain anhydrous conditions, preventing the hydrolysis of the sensitive imidoyl chloride starting material. Detailed standard operating procedures regarding stoichiometry, temperature ramping, and workup techniques are essential for successful scale-up.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent like acetonitrile.
- Heat the reaction mixture to 50-70°C and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via silica gel column chromatography to obtain the final triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic advantages that extend beyond simple yield metrics. The shift away from hazardous azides and expensive transition metals fundamentally alters the cost structure and risk profile of the supply chain. By utilizing commodity chemicals like cesium carbonate and commercially available diazo precursors, the dependency on specialized, high-cost reagents is drastically reduced. This simplification of the bill of materials enhances supply chain resilience, as the sourcing of raw materials becomes less vulnerable to geopolitical disruptions or single-supplier bottlenecks that often affect niche catalysts.
- Cost Reduction in Manufacturing: The elimination of copper catalysts removes the necessity for costly metal scavenging resins and the associated validation testing for residual metals, leading to substantial operational savings. Furthermore, the mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure alternatives, contributing to a lower carbon footprint and reduced utility costs. The simplified workup procedure, often requiring only filtration and standard chromatography, minimizes solvent usage and waste disposal fees, driving down the overall cost of goods sold for these complex intermediates.
- Enhanced Supply Chain Reliability: The robustness of this base-promoted method allows for greater flexibility in manufacturing scheduling and inventory management. Since the starting materials are stable and non-hazardous compared to organic azides, they can be stored in larger quantities without triggering strict hazardous material storage regulations. This capability enables manufacturers to build strategic stockpiles of key intermediates, ensuring continuity of supply even during periods of market volatility. The reduced safety risk also lowers insurance premiums and regulatory compliance burdens, further stabilizing the long-term supply economics.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, having been validated from gram-scale experiments to potential multi-kilogram production runs without significant loss in efficiency. The absence of heavy metals simplifies environmental permitting and wastewater treatment requirements, aligning with increasingly stringent global environmental regulations. This green chemistry approach not only future-proofs the manufacturing process against tightening regulatory standards but also enhances the brand reputation of the end-product as being sustainably sourced, a factor of growing importance to downstream pharmaceutical customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is this new synthesis method safer than traditional triazole production?
A: Traditional methods often rely on toxic and explosive organic azides. This patented process utilizes stable diazo compounds and imidoyl chlorides, eliminating significant safety hazards associated with azide handling and storage.
Q: Does this metal-free process impact the purity of the final API intermediate?
A: Yes, positively. By avoiding copper or other transition metal catalysts, the process removes the need for complex and costly heavy metal scavenging steps, resulting in a cleaner impurity profile suitable for stringent pharmaceutical specifications.
Q: What represents the primary cost advantage of this manufacturing route?
A: The primary cost drivers are reduced through the use of inexpensive, commercially available starting materials and the elimination of expensive noble metal catalysts. Additionally, simplified downstream processing lowers overall operational expenditures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole delivered meets the highest industry standards for potency and impurity control.
We invite you to engage with our technical procurement team to discuss how this innovative chemistry can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this azide-free protocol. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that optimize both your R&D timeline and your bottom line.
