Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Scale-Up
Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and safe methodologies for constructing complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. These fluorinated heterocycles are critical structural motifs found in numerous bioactive molecules, offering enhanced metabolic stability, lipophilicity, and electronegativity compared to their non-fluorinated counterparts. The patent outlines a transformative approach that bypasses traditional limitations, utilizing a base-promoted cyclization between trifluoroethylimidoyl chlorides and diazo compounds. This innovation represents a paradigm shift for reliable pharmaceutical intermediate suppliers aiming to streamline the production of high-value building blocks without the safety hazards associated with azide chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
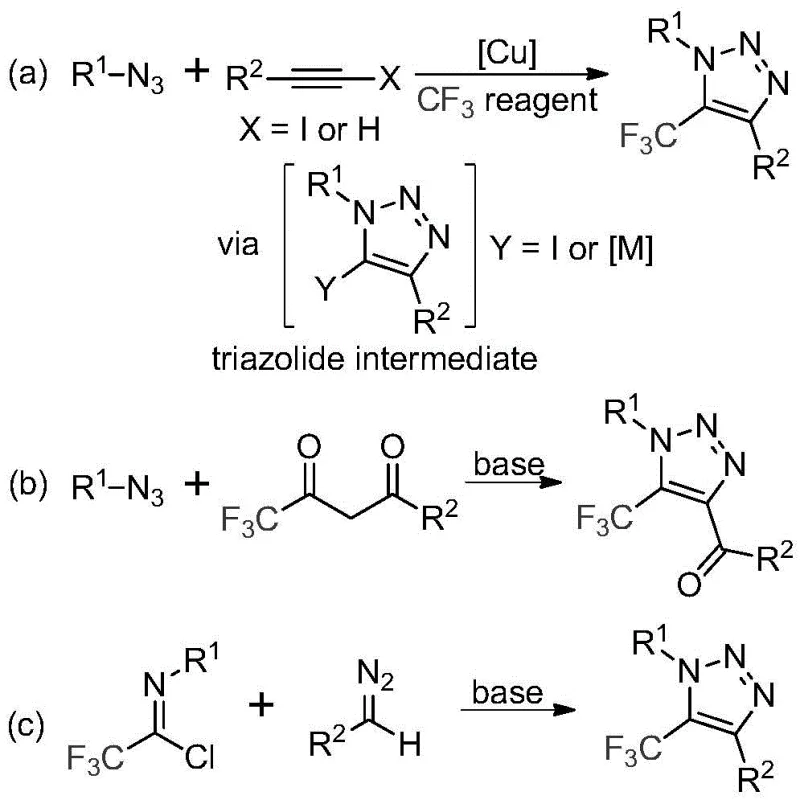
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on two primary synthetic strategies, both of which present substantial drawbacks for large-scale manufacturing. The first common route involves copper-catalyzed [3+2] cycloaddition between alkynes and organic azides, followed by trifluoromethylation. The second strategy employs organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones. The most critical bottleneck in both legacy methods is the mandatory use of organic azides. Azides are notoriously unstable, toxic, and potentially explosive, posing severe safety risks during storage, handling, and reaction, particularly when scaling up to industrial volumes. Furthermore, the copper-catalyzed variant introduces heavy metal contamination issues, necessitating rigorous and expensive purification steps to meet stringent regulatory limits for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these hazardous legacy protocols, the technology described in CN113121462B introduces a metal-free, azide-free pathway that fundamentally alters the risk profile of the synthesis. As illustrated in the comparative reaction schemes below, the new method (pathway c) replaces the dangerous azide component with stable diazo compounds and utilizes trifluoroethylimidoyl chlorides as the electrophilic partner. This strategic substitution not only eliminates the explosion hazard but also removes the dependency on transition metal catalysts. The reaction proceeds efficiently under mild basic conditions, offering a cleaner reaction profile and simplifying the downstream processing workflow. This advancement is pivotal for achieving cost reduction in API manufacturing by removing the need for specialized explosion-proof equipment and expensive metal scavenging resins.
Mechanistic Insights into Base-Promoted Cyclization
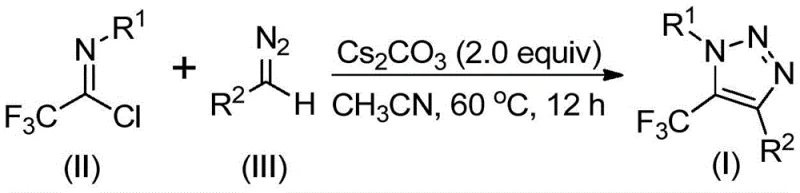
The core of this innovative synthesis lies in a sophisticated base-promoted cascade reaction that ensures high regioselectivity and yield. The mechanism initiates with the deprotonation of the diazo compound by cesium carbonate, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This intermolecular nucleophilic addition-elimination process facilitates the formation of a critical carbon-carbon bond. Subsequently, the intermediate undergoes an intramolecular 5-endo-dig cyclization to close the triazole ring. The choice of cesium carbonate is deliberate; its moderate basicity and solubility profile in polar aprotic solvents like acetonitrile create an optimal environment for this transformation without promoting excessive decomposition of the sensitive diazo reagent. This precise control over reaction kinetics is essential for maintaining a clean impurity profile, which is a top priority for R&D Directors focused on process robustness.
Furthermore, the structural integrity of the final product is preserved through the mild nature of the reaction conditions, typically ranging from 50°C to 70°C. The specific reaction scheme highlights the direct conversion of starting materials (II) and (III) into the target scaffold (I) with high atom economy. The tolerance for diverse functional groups on both the imidoyl chloride and the diazo compound—ranging from electron-donating methoxy groups to electron-withdrawing halogens—demonstrates the versatility of this catalytic system. By avoiding harsh acidic or oxidative conditions, the method minimizes side reactions such as hydrolysis or polymerization, thereby ensuring that the commercial scale-up of complex pharmaceutical intermediates remains feasible and economically viable.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and solvent selection to maximize yield and purity. The patent specifies that the diazo compound should be used in slight excess relative to the imidoyl chloride to drive the reaction to completion, given the reactive nature of the diazo species. The process is operationally simple, involving the mixing of reagents in a standard reactor followed by a controlled heating phase. For detailed procedural specifics regarding exact molar ratios, workup procedures, and purification techniques, please refer to the standardized guide below.
- Combine cesium carbonate, 4 Å molecular sieves, trifluoroethylimidoyl chloride, and diazo compound in an organic solvent such as acetonitrile.
- Heat the reaction mixture to 50-70°C (optimally 60°C) and stir for 8-16 hours to ensure complete conversion.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final 5-trifluoromethyl-1,2,3-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical benefits. The elimination of transition metal catalysts immediately reduces the raw material bill of materials (BOM) and removes the entire unit operation dedicated to metal removal, which often involves expensive scavengers or complex crystallization steps. Additionally, the avoidance of azides drastically lowers insurance premiums and facility compliance costs associated with handling energetic materials. The use of commodity chemicals like cesium carbonate and acetonitrile further stabilizes the supply chain against volatility, ensuring consistent production capabilities.
- Cost Reduction in Manufacturing: The removal of copper catalysts and azide reagents leads to substantial cost savings by simplifying the purification train. Without the need for heavy metal clearance, manufacturers can reduce solvent consumption and waste disposal costs, directly improving the gross margin of the final intermediate. The use of inexpensive inorganic bases instead of precious metal complexes further drives down the variable cost per kilogram, making this route highly competitive for large-volume production.
- Enhanced Supply Chain Reliability: The starting materials, specifically trifluoroethylimidoyl chlorides and diazo compounds, are derived from readily available bulk chemicals, reducing the risk of supply bottlenecks. Unlike specialized azide reagents which may have limited suppliers and long lead times, the precursors for this method are commoditized. This accessibility ensures reducing lead time for high-purity intermediates and allows for more flexible inventory management strategies.
- Scalability and Environmental Compliance: The reaction operates under mild thermal conditions and generates minimal hazardous waste, aligning with modern green chemistry principles. The absence of heavy metals simplifies wastewater treatment and reduces the environmental footprint of the manufacturing process. This ease of scale-up from gram to multi-kilogram batches ensures that production can be ramped up quickly to meet market demand without requiring significant capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on safety, scalability, and applicability for potential licensees or manufacturing partners.
Q: Does this synthesis method require toxic azide reagents?
A: No, unlike conventional methods that rely on explosive and toxic organic azides, this patented process utilizes stable diazo compounds and trifluoroethylimidoyl chlorides, significantly enhancing operational safety.
Q: Is a transition metal catalyst required for this reaction?
A: No, the reaction is metal-free. It employs cesium carbonate as a base promoter, which eliminates the need for expensive copper catalysts and the subsequent costly steps required to remove heavy metal residues from the final product.
Q: What is the substrate scope for this trifluoromethyl triazole synthesis?
A: The method demonstrates broad functional group tolerance, accommodating various substituents on the aromatic rings including alkyl, alkoxy, halogens, and trifluoromethyl groups, making it highly versatile for diverse pharmaceutical intermediate applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting cutting-edge synthetic methodologies like the one described in CN113121462B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped to handle metal-free chemistries with precision, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest global regulatory standards.
We invite you to collaborate with us to leverage this efficient, azide-free technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this advanced synthesis can optimize your supply chain and accelerate your time to market.
