Revolutionizing Azabicyclic Intermediate Production: A Deep Dive into Patent CN114031542A for Commercial Scale-Up
The pharmaceutical industry is currently witnessing a paradigm shift in the synthesis of complex heterocyclic intermediates, driven by the urgent need for efficient supply chains for antiviral therapeutics. Patent CN114031542A introduces a groundbreaking methodology for the preparation of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane, a critical scaffold found in potent protease inhibitors such as Boceprevir and the novel coronavirus treatment PF-07321332. This technology leverages a sophisticated copper-catalyzed intramolecular cyclization strategy that fundamentally redefines the efficiency of constructing the azabicyclic core. By transitioning from traditional multi-step oxidative pathways to a direct cyclization approach, this innovation addresses long-standing bottlenecks in purity, yield, and environmental compliance. For R&D directors and procurement strategists, understanding the mechanistic nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity APIs at scale.
![Chemical structures of Boceprevir and PF-07321332 highlighting the 6,6-dimethyl-3-azabicyclo[3.1.0]hexane core](/insights/img/6-6-dimethyl-3-azabicyclo-hexane-cu-catalysis-pharma-supplier-20260307170528-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane has relied heavily on starting materials such as ethyl chrysanthemate or protected prenol derivatives. These legacy routes, as illustrated in prior art documents like CN104163759B, necessitate a cumbersome sequence involving the initial formation of caronic acid through extensive oxidation protocols. This traditional pathway is fraught with inefficiencies; it demands the use of large quantities of oxidizing agents to convert carenic acid or its lactones into caronic acid, inevitably generating substantial volumes of wastewater and saline waste that complicate environmental management. Furthermore, the crude caronic acid obtained is typically a mixture of cis and trans isomers, requiring an energy-intensive high-temperature isomerization step to enrich the desired cis-configuration before proceeding to anhydride formation. The subsequent reduction of the anhydride to the target amine often relies on expensive and hazardous reducing agents like lithium aluminum hydride in stoichiometric excess, driving up production costs and safety risks significantly.
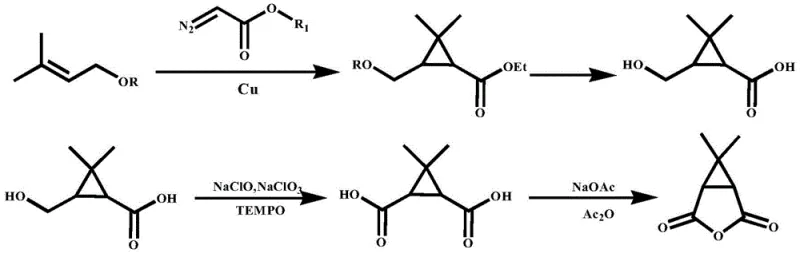
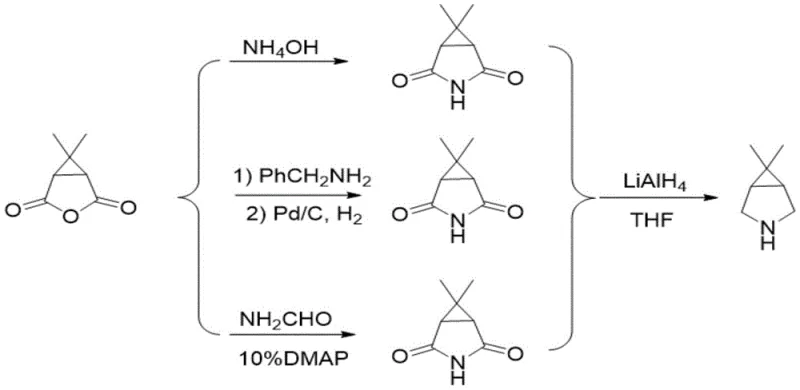
The Novel Approach
In stark contrast, the methodology disclosed in patent CN114031542A circumvents these oxidative hurdles entirely by employing a direct intramolecular cyclization of diazo-functionalized olefinic precursors. This novel route constructs the heteroatom bicyclic framework in a single catalytic step, effectively bypassing the need for external oxidants and the associated waste streams. The process initiates with a general formula (1) compound, which undergoes a metal-catalyzed cyclopropanation to form the bicyclic core with exceptional stereocontrol. By eliminating the oxidation-reduction loop inherent in older methods, this approach not only shortens the synthetic timeline but also drastically improves the atom economy. The ability to access the target scaffold directly from diazo precursors allows for greater flexibility in introducing functional groups early in the synthesis, facilitating the production of diverse derivatives required for next-generation drug discovery programs without the baggage of legacy purification challenges.
Mechanistic Insights into Cu-Catalyzed Intramolecular Cyclization
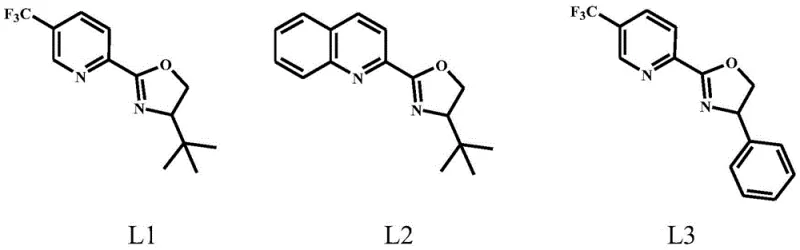
The heart of this technological advancement lies in the precise orchestration of the copper-catalyzed cyclization reaction. The process utilizes a catalytic system comprising copper salts—such as cuprous chloride, cuprous trifluoromethanesulfonate, or copper sulfate—paired with specialized organic ligands. These ligands, falling under general formulas La through Le, are critical for modulating the electronic and steric environment around the metal center. Specifically, ligands like L1, L2, and L3, which feature nitrogen-oxygen or nitrogen-nitrogen multidentate coordination motifs, play a pivotal role in stabilizing the reactive copper-carbenoid intermediate generated from the diazo precursor. This stabilization is key to controlling the trajectory of the carbene insertion into the proximal olefinic bond, ensuring that the cyclopropane ring forms with the correct geometry relative to the existing substituents.
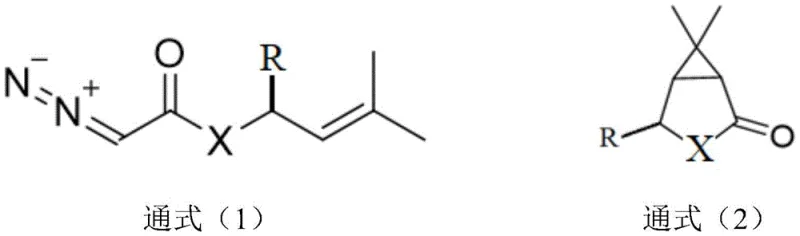
The reaction mechanism proceeds through the decomposition of the diazo group to form a metal-carbene species, which then undergoes an intramolecular attack on the alkene moiety. The choice of solvent, such as dichloroethane (DCE), toluene, or acetonitrile, and the maintenance of reaction temperatures between 60°C and 80°C are optimized to balance reaction kinetics with the stability of the diazo functionality. Crucially, this catalytic system achieves a diastereomeric ratio (dr) of greater than 100:1 in favor of the cis-isomer. This high level of stereoselectivity is a direct result of the ligand-controlled transition state, which energetically disfavors the formation of the trans-configured byproduct. Consequently, the need for downstream isomerization or difficult chromatographic separations is rendered obsolete, streamlining the path to high-purity intermediates.
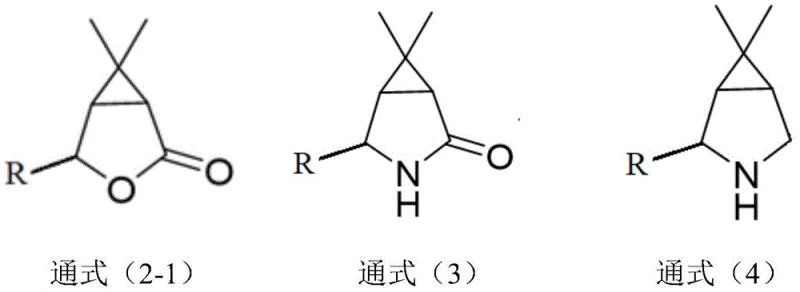
How to Synthesize 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Efficiently
Implementing this synthesis requires careful attention to the preparation of the diazo starting material and the precise control of the catalytic cycle. The process begins with the generation of the diazo-olefin precursor, which can be derived from aminoacetates via diazotization under acidic conditions. Once the precursor is secured, the cyclization is executed in the presence of the copper-ligand complex under inert atmosphere to prevent side reactions. Following the successful formation of the bicyclic lactone or lactam intermediate, the pathway diverges slightly depending on the specific derivative required, generally proceeding through an amination step using ammonia in methanol followed by a controlled reduction. The detailed standardized synthesis steps see the guide below.
- Perform intramolecular cyclization of general formula (1) compounds using a copper salt catalyst and organic ligand (e.g., L1-L3) in solvents like DCE at 60-80°C to obtain high cis-selectivity bicyclic intermediates.
- Subject the resulting lactone intermediate (formula 2-1) to an amination reaction using ammonia in methanol at low temperatures (-10 to 5°C) to form the lactam structure (formula 3).
- Reduce the lactam carbonyl group using lithium aluminum hydride or borohydride systems in aprotic solvents like THF to yield the final 6,6-dimethyl-3-azabicyclo[3.1.0]hexane product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN114031542A translates into tangible strategic advantages beyond mere chemical elegance. The primary value proposition lies in the radical simplification of the manufacturing workflow, which directly impacts cost structures and supply reliability. By removing the oxidation and isomerization stages, the process significantly reduces the consumption of raw materials and utilities, leading to substantial cost savings in manufacturing operations. The elimination of heavy oxidant usage also means a drastic reduction in waste treatment costs, aligning production with increasingly stringent environmental regulations and sustainability goals without compromising output volume.
- Cost Reduction in Manufacturing: The new route eliminates the need for expensive stoichiometric oxidants and reduces the reliance on high-cost reducing agents like lithium aluminum hydride by optimizing the reduction step to target only specific carbonyl groups. This qualitative shift in reagent usage lowers the bill of materials significantly. Furthermore, the high yield of the cyclization step, reported to reach up to 98.5 percent, ensures that less starting material is wasted, maximizing the throughput of existing reactor capacity and improving the overall return on investment for production campaigns.
- Enhanced Supply Chain Reliability: Traditional routes often suffer from supply bottlenecks due to the limited availability of specialized starting materials like kung fu acid or the complexities of managing hazardous oxidation steps. This novel method utilizes more accessible diazo-olefin precursors that can be synthesized from commodity chemicals. The robustness of the copper-catalyzed system, which tolerates a range of solvents and conditions, ensures consistent batch-to-batch quality. This reliability is critical for maintaining continuous supply lines for high-demand antiviral medications, mitigating the risk of production stoppages due to raw material shortages or process failures.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, operating at moderate temperatures and avoiding the extreme conditions associated with high-temperature isomerization. The reduction in wastewater and saline waste generation simplifies effluent treatment, making it easier for manufacturing sites to maintain compliance with local environmental standards. This 'green' profile not only reduces regulatory risk but also enhances the corporate social responsibility standing of the supply chain, a factor increasingly weighted in vendor selection by major pharmaceutical companies seeking sustainable partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the process capabilities and limitations for potential adopters.
Q: How does the new Cu-catalyzed route improve upon traditional synthesis methods for 6,6-dimethyl-3-azabicyclo[3.1.0]hexane?
A: Traditional methods often require lengthy oxidation steps to convert carene derivatives into caronic acid anhydrides, generating significant waste salts and water. The new patent CN114031542A utilizes a direct intramolecular cyclization of diazo-olefin precursors, bypassing these oxidation steps entirely. This results in higher atom economy, reduced environmental impact, and eliminates the need for energy-intensive high-temperature isomerization of trans-isomers.
Q: What represents the key advantage regarding stereoselectivity in this process?
A: The process achieves exceptional cis-selectivity with a diastereomeric ratio (dr) greater than 100:1. This high selectivity is crucial because it avoids the formation of unwanted trans-isomers that typically require difficult and costly separation or high-energy isomerization processes in conventional routes, thereby simplifying purification and boosting overall yield.
Q: Is this synthesis method scalable for industrial production of antiviral intermediates?
A: Yes, the method is specifically designed for industrial scalability. By utilizing readily available copper catalysts and avoiding hazardous or expensive stoichiometric oxidants, the process offers a safer and more cost-effective profile. The high yields (up to 98.5% in cyclization) and simplified downstream processing make it highly suitable for meeting the large-scale demand for intermediates used in drugs like Boceprevir and PF-07321332.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Supplier
As the global demand for antiviral therapeutics continues to surge, the ability to produce key intermediates like 6,6-dimethyl-3-azabicyclo[3.1.0]hexane efficiently and sustainably has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this chemical evolution, leveraging advanced catalytic technologies to deliver superior quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of top-tier pharmaceutical clients. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the exacting standards required for GMP-grade API synthesis, providing you with a secure and dependable source for your critical supply chain needs.
We invite you to explore how our technical expertise can optimize your procurement strategy. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of patent CN114031542A can drive value and security into your pharmaceutical manufacturing operations.
