Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Advanced Pharmaceutical Applications
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Advanced Pharmaceutical Applications
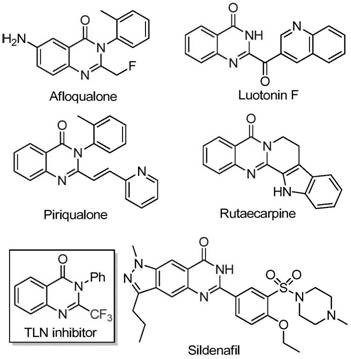
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds remains a pivotal challenge in modern medicinal chemistry, particularly for enhancing the metabolic stability and lipophilicity of drug candidates. Quinazolinone derivatives, renowned for their broad spectrum of biological activities including anticancer, anticonvulsant, and anti-inflammatory properties, represent a high-value class of intermediates in the pharmaceutical sector. However, traditional synthetic routes often suffer from limitations such as severe reaction conditions, narrow substrate scope, and the reliance on expensive fluorinating agents. Addressing these critical bottlenecks, the recent intellectual property disclosed in patent CN111675662B introduces a transformative preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This innovation leverages a cost-effective iron-catalyzed cyclization strategy that not only simplifies the operational workflow but also significantly expands the chemical space accessible to process chemists.
This novel methodology utilizes readily available trifluoroethylimidoyl chloride and isatin derivatives as key building blocks, circumventing the need for complex pre-functionalized synthons. By employing ferric trichloride as a Lewis acid catalyst in conjunction with sodium hydride, the process achieves high conversion rates under relatively mild thermal conditions. For global procurement teams and R&D directors seeking reliable pharmaceutical intermediate suppliers, this technology represents a substantial opportunity to optimize manufacturing costs while ensuring the high purity required for downstream API synthesis. The following analysis dissects the technical merits and commercial implications of this breakthrough.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has relied heavily on the cyclization of anthranilamide or isatoic anhydride derivatives with trifluoroacetylating agents such as trifluoroacetic anhydride or ethyl trifluoroacetate. While these classical approaches are well-documented in literature, they are frequently plagued by significant drawbacks that hinder their utility in large-scale commercial manufacturing. The reaction conditions often necessitate strong bases or high temperatures that can lead to the decomposition of sensitive functional groups, thereby limiting the diversity of substituents that can be tolerated on the aromatic ring. Furthermore, the atom economy of these traditional routes is often suboptimal, generating substantial amounts of waste and requiring extensive purification steps to remove unreacted starting materials and byproducts.
Another critical limitation lies in the cost and availability of the trifluoromethyl synthons themselves. Reagents like trifluoroacetic anhydride are not only expensive but also pose handling challenges due to their corrosive nature and moisture sensitivity. In an industrial setting, the requirement for stringent anhydrous conditions and specialized equipment to handle such reagents drives up capital expenditure and operational complexity. Additionally, the yields reported for these conventional methods are frequently inconsistent, particularly when scaling up from milligram to kilogram quantities, leading to unpredictable production timelines and increased cost of goods sold (COGS). These factors collectively create a bottleneck for the efficient production of high-purity pharmaceutical intermediates needed for clinical and commercial supply.
The Novel Approach
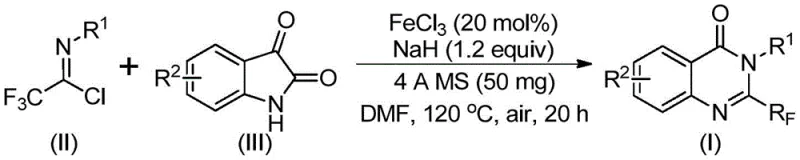
In stark contrast to the legacy methods, the invention detailed in CN111675662B proposes a streamlined and robust synthetic pathway that fundamentally alters the retrosynthetic logic. By shifting the focus to the reaction between trifluoroethylimidoyl chloride and isatin, the process utilizes precursors that are both commercially accessible and chemically stable. The core of this innovation is the use of an inexpensive iron catalyst system, specifically ferric chloride, which activates the substrates for cyclization without the need for precious metals like palladium or rhodium. This switch to base-metal catalysis is a game-changer for cost reduction in pharmaceutical intermediate manufacturing, as it eliminates the risk of heavy metal contamination that often requires costly scavenging steps in final drug substances.
The operational simplicity of this new approach is equally compelling. The reaction proceeds efficiently in polar aprotic solvents such as DMF, with the addition of 4A molecular sieves to manage water content and drive the equilibrium forward. The protocol tolerates a wide range of electronic and steric environments on the isatin and imidoyl chloride components, allowing for the rapid generation of diverse libraries of quinazolinone analogs. This broad substrate scope is essential for medicinal chemists engaged in structure-activity relationship (SAR) studies, as it enables the exploration of chemical space that was previously inaccessible or prohibitively expensive. Moreover, the reaction conditions are scalable, having been demonstrated effectively at the gram level with the potential for further expansion to multi-kilogram batches, thereby securing the supply chain for future commercial demands.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between the iron catalyst and the basic promoter, sodium hydride. Initially, the sodium hydride serves to deprotonate the isatin nitrogen, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step forms a key trifluoroacetamidine intermediate, which sets the stage for the subsequent ring closure. The presence of ferric chloride is crucial at this juncture; it acts as a Lewis acid to coordinate with the carbonyl oxygen and the imine nitrogen, facilitating the intramolecular decarbonylation and cyclization steps. This iron-mediated activation lowers the energy barrier for the formation of the quinazolinone core, allowing the reaction to proceed at moderate temperatures compared to thermal cyclizations.
Furthermore, the inclusion of 4A molecular sieves plays a non-trivial role in the reaction kinetics and thermodynamics. By sequestering trace amounts of water generated during the condensation or present in the solvent, the molecular sieves prevent the hydrolysis of the sensitive imidoyl chloride and the intermediate amidine species. This ensures that the reaction equilibrium is driven decisively towards the product side, resulting in the high yields observed across various substrates. The tolerance of the catalytic system towards different substituents, such as halogens, alkyl groups, and methoxy groups on the aromatic rings, suggests a mechanism that is not overly sensitive to electronic effects. This robustness is a hallmark of a mature industrial process, providing R&D teams with the confidence to design complex molecules without fear of reaction failure due to subtle structural changes.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed to be straightforward, minimizing the need for specialized equipment or exotic reagents. The general procedure involves charging a reaction vessel with the catalyst system and substrates in a suitable solvent, followed by a controlled heating profile. The initial phase of the reaction occurs at a lower temperature to facilitate the formation of the intermediate, followed by a higher temperature phase to drive the cyclization to completion. This two-stage thermal profile optimizes the reaction kinetics, ensuring high conversion while minimizing side reactions. The work-up procedure is equally simple, typically involving filtration to remove the solid catalyst and molecular sieves, followed by standard chromatographic purification to isolate the pure product.
- Charge a reaction vessel with ferric chloride (20 mol%), sodium hydride (1.2 equiv), and 4A molecular sieves in anhydrous DMF.
- Add trifluoroethylimidoyl chloride and the substituted isatin derivative to the mixture under air atmosphere.
- Heat the reaction mixture to 120°C for 20 hours, then filter and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed methodology offers tangible benefits that extend beyond mere chemical elegance. The primary driver for value creation here is the drastic simplification of the raw material portfolio. By replacing expensive and hazardous fluorinating agents with stable, commodity-grade chemicals, the overall cost structure of the synthesis is significantly improved. This reduction in input costs directly translates to better margins for the final API or a more competitive pricing strategy for the intermediate supplier. Additionally, the use of iron, an earth-abundant metal, eliminates the volatility associated with the pricing and supply of precious metal catalysts, thereby stabilizing long-term procurement planning.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of inexpensive iron salts result in a substantial decrease in catalyst costs per kilogram of product. Furthermore, the simplified work-up procedure reduces the consumption of solvents and silica gel during purification, leading to lower waste disposal costs and a smaller environmental footprint. The high yields reported across a broad range of substrates mean that less starting material is wasted, maximizing the efficiency of every batch produced and driving down the effective cost of goods.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials such as isatins and trifluoroethylimidoyl chlorides ensures a robust supply chain that is less susceptible to disruptions. Unlike specialized reagents that may have long lead times or single-source suppliers, the key inputs for this process are widely produced by multiple chemical manufacturers globally. This redundancy in the supply base provides a critical safety net for production schedules, ensuring that manufacturing timelines are met consistently even in volatile market conditions.
- Scalability and Environmental Compliance: The process is inherently scalable, having been validated from small-scale optimization to gram-level synthesis with clear pathways for tonnage production. The use of DMF, a common industrial solvent, allows for easy integration into existing manufacturing infrastructure without the need for major capital investment in new reactor types. Moreover, the avoidance of toxic heavy metals aligns with increasingly stringent environmental regulations and green chemistry principles, reducing the regulatory burden and facilitating smoother approval processes for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when adopting this route. Understanding these nuances is essential for making informed decisions about process development and sourcing strategies.
Q: What are the primary advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride offers a distinct economic and operational advantage by replacing expensive transition metal catalysts with an abundant, low-cost iron salt. Furthermore, this method demonstrates superior functional group tolerance, allowing for the synthesis of diverse derivatives without harsh conditions that might degrade sensitive moieties.
Q: How does this method improve the supply chain stability for trifluoromethylated heterocycles?
A: By utilizing readily available starting materials such as isatins and trifluoroethylimidoyl chlorides, this process mitigates reliance on scarce or highly regulated reagents. The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, which is critical for maintaining uninterrupted supply chains in API manufacturing.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the method's scalability from gram-level laboratory synthesis to potential industrial application. The simplicity of the work-up procedure, involving standard filtration and chromatography, combined with the use of common solvents like DMF, facilitates straightforward scale-up processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has thoroughly analyzed the potential of the FeCl3-catalyzed route described in CN111675662B and is fully prepared to leverage this technology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone we deliver meets the highest industry standards.
We invite you to collaborate with us to unlock the full potential of this innovative chemistry for your pipeline. Whether you require custom synthesis of novel analogs or reliable supply of known intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to quality and efficiency can become a cornerstone of your supply chain strategy.
