Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Drug Discovery
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Drug Discovery
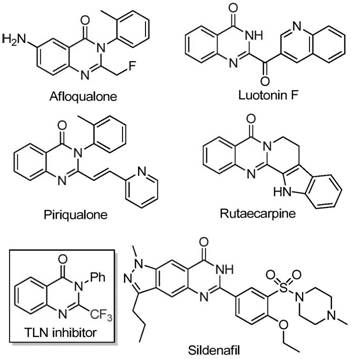
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for nitrogen-containing heterocycles, particularly quinazolinones, due to their pervasive presence in bioactive molecules. As detailed in the groundbreaking patent CN111675662B, a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds has been developed, addressing critical bottlenecks in modern medicinal chemistry. This technology leverages an inexpensive iron-catalyzed cyclization strategy that transforms readily available isatin derivatives and trifluoroethylimidoyl chlorides into high-value scaffolds. The introduction of the trifluoromethyl group is strategically vital, as it significantly enhances the electronegativity, metabolic stability, and lipophilicity of the target drug candidates, thereby improving their overall bioavailability. For R&D directors and procurement specialists alike, this methodology represents a paradigm shift from costly, low-yielding traditional syntheses to a streamlined, cost-effective process suitable for both gram-scale discovery and multi-ton commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functionalities has relied heavily on cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with anthranilamides or isatoic anhydrides. While chemically feasible, these conventional pathways are plagued by severe operational disadvantages that hinder their utility in a commercial setting. The reaction conditions are often excessively harsh, requiring stringent temperature controls and specialized equipment that increase capital expenditure. Furthermore, the starting materials, particularly the fluorinated synthons, are frequently expensive and subject to volatile market pricing, which destabilizes supply chains. Perhaps most critically for process chemists, these older methods suffer from narrow substrate scope and consistently low yields, generating significant chemical waste and complicating downstream purification efforts. The inability to tolerate diverse functional groups limits the structural diversity accessible to medicinal chemists, slowing down the optimization of lead compounds.
The Novel Approach
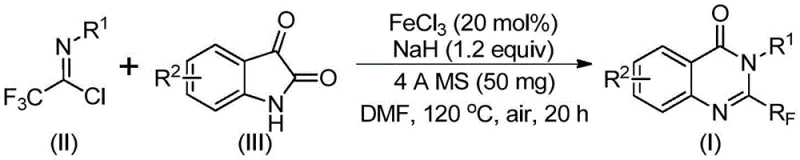
In stark contrast, the innovative method disclosed in the patent utilizes a synergistic combination of ferric chloride and sodium hydride to catalyze the coupling of trifluoroethylimidoyl chloride with isatin derivatives. This approach fundamentally alters the economic and operational landscape of quinazolinone synthesis by employing earth-abundant iron as the catalyst, eliminating the need for precious metals like palladium or rhodium. The reaction proceeds through a tandem sequence involving initial carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization, effectively constructing the heterocyclic core in a single pot. This one-pot strategy not only simplifies the workflow by reducing unit operations but also dramatically improves atom economy. The resulting process is characterized by mild reaction conditions, typically operating between 40°C and 120°C in common solvents like DMF, and demonstrates exceptional functional group tolerance, allowing for the incorporation of halogens, alkyls, and nitro groups without protective group strategies.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of base-promoted nucleophilic attack and transition metal catalysis. Initially, sodium hydride acts as a strong base to deprotonate the isatin substrate, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step forms a transient trifluoroacetamidine intermediate, setting the stage for the subsequent cyclization. The ferric chloride catalyst then plays a dual role: it facilitates the decarbonylation of the intermediate and promotes the intramolecular cyclization required to close the quinazolinone ring. This iron-mediated step is crucial for overcoming the kinetic barriers associated with forming the fused ring system under mild thermal conditions. The use of 4A molecular sieves in the reaction mixture further drives the equilibrium forward by sequestering moisture, ensuring that the sensitive imidoyl chloride reagent remains intact and reactive throughout the prolonged heating phase.
From an impurity control perspective, this mechanism offers distinct advantages over acid-catalyzed alternatives. The basic conditions employed minimize the risk of acid-sensitive side reactions, such as the hydrolysis of the trifluoromethyl group or the degradation of the lactam ring. The high selectivity of the iron catalyst ensures that the cyclization occurs regioselectively, preventing the formation of isomeric byproducts that are notoriously difficult to separate. Moreover, the broad substrate scope observed in the experimental data suggests that the catalytic cycle is robust against steric hindrance and electronic variations on the aromatic rings. Whether the substrate bears electron-withdrawing groups like fluorine and chlorine or electron-donating groups like methyl and methoxy, the reaction maintains high efficiency, yielding pure products with minimal chromatographic effort. This predictability is essential for maintaining a clean impurity profile, a key requirement for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory research and pilot plant scaling. The protocol involves a straightforward addition of reagents into a reaction vessel, followed by a two-stage heating profile that optimizes conversion while minimizing energy consumption. Detailed standard operating procedures regarding stoichiometry, solvent selection, and workup protocols are critical for reproducing the high yields reported in the patent literature. Researchers should pay close attention to the quality of the sodium hydride and the dryness of the solvent to ensure optimal catalyst performance. For a comprehensive guide on the specific molar ratios and temperature ramps required for different substrates, please refer to the standardized synthesis steps outlined below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C for 18-20 hours under air.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed methodology translates directly into tangible strategic benefits that enhance the bottom line. The shift from precious metal catalysts to inexpensive ferric chloride eliminates a major cost driver in the bill of materials, while simultaneously removing the complex and costly heavy metal scavenging steps typically required in downstream processing. This simplification of the purification train reduces solvent consumption and waste disposal costs, contributing to a greener and more sustainable manufacturing footprint. Furthermore, the reliance on commodity chemicals like isatin and simple aromatic amines as starting materials insulates the supply chain from the volatility associated with specialized fluorinated reagents. This stability ensures consistent availability of raw materials, reducing the risk of production delays caused by supplier shortages or logistical bottlenecks.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the use of cheap, commercially available starting materials drastically lower the direct material costs per kilogram of product. Additionally, the simplified one-pot procedure reduces labor hours and utility consumption by consolidating multiple reaction steps into a single operation. The high conversion rates and yields minimize the loss of valuable intermediates, ensuring that the maximum amount of input material is converted into saleable product. These factors combine to create a highly cost-competitive process that allows for significant margin improvement in the final API or intermediate pricing.
- Enhanced Supply Chain Reliability: By utilizing widely sourced reagents such as isatin and ferric chloride, the manufacturing process becomes less dependent on niche suppliers with long lead times. The robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality, further stabilizing the supply chain. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery demands of global pharmaceutical clients. The ability to source materials from multiple vendors reduces single-source risk and provides greater negotiating leverage in procurement contracts.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, having been demonstrated to work efficiently from gram scales up to potential multi-kilogram batches without loss of performance. The use of iron, a non-toxic and environmentally benign metal, aligns with increasingly stringent environmental regulations regarding heavy metal residues in pharmaceutical products. The reduced generation of hazardous waste and the lower energy requirements of the mild heating profile contribute to a smaller carbon footprint. This alignment with green chemistry principles not only facilitates regulatory approval but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the method. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing workflows or new drug development programs.
Q: What are the key advantages of this iron-catalyzed method over traditional synthesis?
A: This method utilizes inexpensive and readily available starting materials like isatin and trifluoroethylimidoyl chloride, avoiding expensive trifluoroacetic anhydride. It operates under milder conditions with higher yields and broader substrate tolerance.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the protocol is designed for scalability, utilizing cheap iron catalysts and simple post-treatment procedures like filtration and column chromatography, making it viable for commercial scale-up.
Q: What types of substituents are tolerated on the aromatic rings?
A: The reaction exhibits excellent functional group tolerance, successfully accommodating alkyl, halogen, methoxy, and nitro groups at ortho-, meta-, and para-positions without significant yield loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis for the next generation of therapeutic agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting the most stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest international standards. We are committed to leveraging our technical expertise to optimize this novel route, ensuring maximum yield and minimal impurity levels for your specific application.
We invite you to engage with our technical team to explore how this cost-effective synthesis can accelerate your drug development timeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this iron-catalyzed process for your specific project needs. Please contact our technical procurement team today to obtain specific COA data for our catalog compounds or to discuss custom route feasibility assessments tailored to your unique molecular targets. Let us be your partner in turning complex chemical challenges into commercial successes.
