Advanced Manufacturing of Candesartan Intermediates via Novel Ethoxycarbonyl Protection Strategy
Introduction to Patent CN102391254A and Strategic Process Innovation
The pharmaceutical industry continuously seeks robust synthetic routes for Angiotensin II receptor blockers, specifically Candesartan cilexetil and its active acid form, TCV-116. Patent CN102391254A discloses a groundbreaking preparation method that fundamentally alters the traditional synthetic landscape for this critical antihypertensive agent. By strategically replacing tert-butyl alcohol with ethanol in the initial alkylation of the active nitrogen atom on the benzene ring, this invention introduces an ethoxycarbonyl group that serves a dual purpose as both a protecting group and a cyclization precursor. This ingenious modification allows the intermediate to undergo nucleophilic substitution and reduction reactions before directly entering a cyclization phase to generate 1-[(2'-cyanobiphenyl-4-yl)methyl]-2-ethoxy-1H-benzimidazole-7-ethyl formate. The elimination of the hydrolysis step required to remove protecting groups prior to cyclization, coupled with the avoidance of expensive reagents like tetraethyl orthoformate, represents a significant leap forward in process chemistry efficiency. For global procurement and R&D teams, this patent offers a viable pathway to reduce production cycles and enhance overall yield while maintaining stringent purity standards required for API manufacturing.
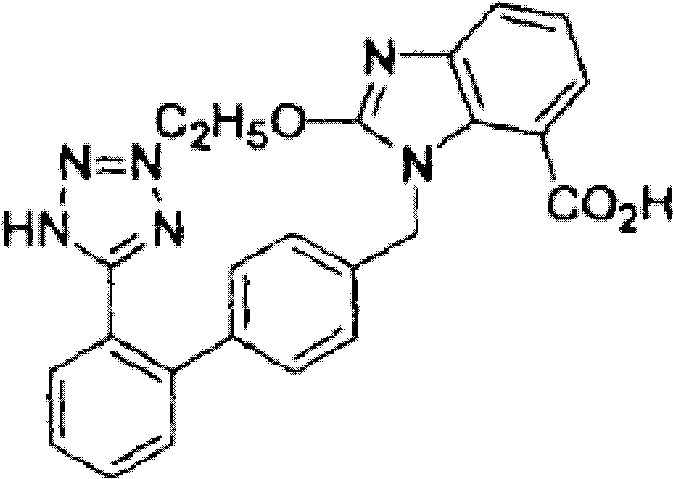
This technological advancement addresses the critical bottlenecks found in legacy synthesis routes, particularly the cumbersome protection-deprotection sequences that inflate manufacturing costs and extend lead times. The core innovation lies in the stability and reactivity profile of the ethoxycarbonyl moiety, which remains intact during the harsh conditions of nucleophilic substitution yet facilitates ring closure under acidic conditions without external carbon sources. This not only streamlines the material flow but also minimizes waste generation, aligning with modern green chemistry principles. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic nuances is essential for evaluating the commercial viability and scalability of the proposed route. The patent explicitly details a sequence starting from 3-nitrophthalic acid derivatives, progressing through acyl chloride and azide intermediates, and culminating in the formation of the benzimidazole core with high regioselectivity. This comprehensive approach ensures that the final product meets the rigorous quality specifications demanded by regulatory bodies worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Candesartan intermediates have long been plagued by inefficiencies inherent in multi-step protection strategies. Historically, the synthesis involves the introduction of a tert-butoxycarbonyl (Boc) group onto the nitrogen atom of the phthalic acid derivative to prevent side reactions during subsequent alkylation. While effective for protection, this group necessitates a dedicated deprotection step involving hydrolysis before the molecule can undergo cyclization to form the benzimidazole ring. Furthermore, the cyclization step itself traditionally relies on expensive and moisture-sensitive reagents such as tetraethyl orthoformate to provide the necessary carbon atom for ring closure. These additional unit operations not only extend the production cycle significantly but also introduce opportunities for yield loss at each stage. The requirement for strict anhydrous conditions for orthoformate reactions and the handling of large volumes of solvents for deprotection washes contribute to elevated operational expenditures. Moreover, the removal of the Boc group often generates volatile byproducts and requires careful pH control, adding complexity to the waste treatment protocols. For a procurement manager, these factors translate into higher raw material costs, increased energy consumption for heating and cooling across multiple steps, and a larger environmental footprint, all of which erode the profit margins of the final API.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the unique reactivity of the ethoxycarbonyl group to bypass these traditional hurdles. By utilizing ethanol for the initial alkylation, the process installs an ethoxycarbonyl protecting group that is sufficiently stable to withstand the nucleophilic substitution with 4'-bromomethyl-2-cyanobiphenyl but reactive enough to participate directly in the cyclization event. After the nucleophilic substitution and the subsequent reduction of the nitro group to an amine using hydrazine hydrate and activated carbon, the intermediate undergoes cyclization simply upon treatment with glacial acetic acid. This eliminates the need for a separate deprotection step entirely, as the ethoxycarbonyl group provides the carbon necessary for the benzimidazole ring formation in situ. Consequently, the reliance on costly tetraethyl orthoformate is completely removed, leading to substantial cost reduction in pharmaceutical intermediates manufacturing. The streamlined sequence reduces the total number of isolation and purification steps, thereby minimizing product loss and solvent usage. This approach not only accelerates the time-to-market for the intermediate but also enhances the overall atom economy of the process. For supply chain stakeholders, this translates to a more resilient production schedule with fewer potential points of failure, ensuring a consistent supply of high-purity materials essential for downstream API synthesis.
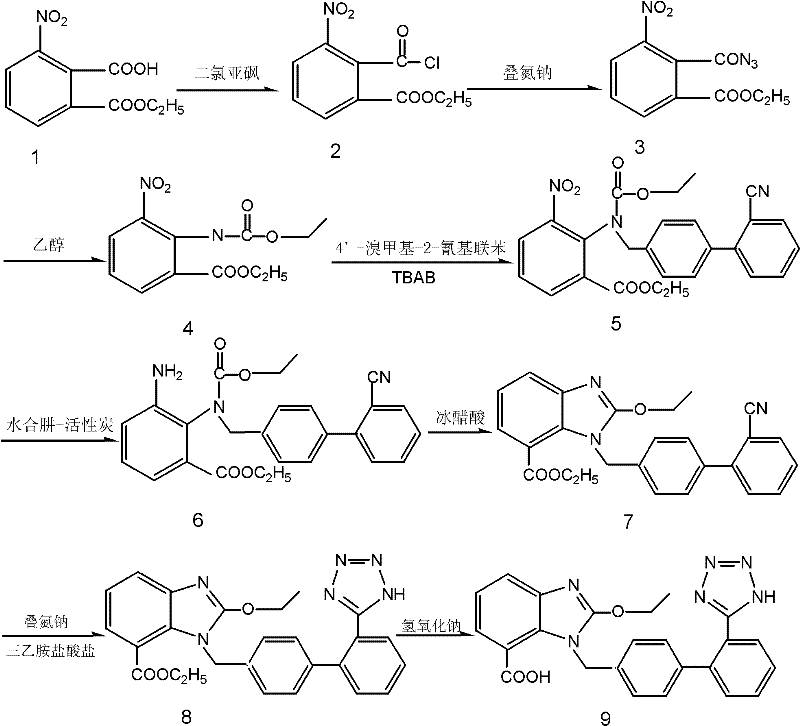
Mechanistic Insights into Ethoxycarbonyl-Mediated Cyclization
The mechanistic elegance of this synthesis lies in the dual functionality of the ethoxycarbonyl group introduced in step 3. Initially, the reaction of the acyl azide intermediate with ethanol generates an ethyl carbamate (urethane) linkage at the nitrogen position. Unlike the bulky tert-butyl group, the ethyl group offers a balance of steric protection and electronic activation. During the nucleophilic substitution in step 4, the nitrogen atom, now part of the carbamate, acts as a nucleophile attacking the benzylic bromide of the biphenyl derivative. The presence of a phase-transfer catalyst like tetrabutylammonium bromide (TBAB) facilitates this reaction in a heterogeneous dichloromethane-water system by shuttling the hydroxide ions into the organic phase, thereby enhancing the nucleophilicity of the nitrogen without requiring harsh homogeneous basic conditions. Following the reduction of the ortho-nitro group to an amine in step 5, the molecule is primed for cyclization. In step 6, the addition of glacial acetic acid protonates the amine and activates the carbonyl of the adjacent ethoxycarbonyl group. An intramolecular nucleophilic attack by the amine nitrogen on the carbamate carbonyl ensues, followed by the elimination of ethanol. This cyclization effectively constructs the benzimidazole core while simultaneously removing the protecting group in a single thermodynamic sink, driving the reaction to completion without the need for external dehydrating agents or carbon sources.
Impurity control is another critical aspect where this mechanism offers distinct advantages. In traditional routes, incomplete deprotection or side reactions with orthoformates can lead to difficult-to-remove impurities that persist through to the final API. In this novel route, the cyclization is driven by the inherent proximity of the reacting groups and the stability of the aromatic benzimidazole system. The use of hydrazine hydrate and activated carbon for the reduction step is particularly noteworthy; activated carbon not only catalyzes the reduction but also adsorbs colored impurities and heavy metal residues, resulting in a cleaner amine intermediate prior to cyclization. The heterogeneous nature of the nucleophilic substitution further aids in purity; since the inorganic salts formed during the reaction remain in the aqueous phase, the organic layer containing the product is inherently cleaner upon separation. This reduces the burden on downstream crystallization steps, which are often the bottleneck in achieving the high purity levels (>99%) required for regulatory filing. By minimizing the formation of side products associated with over-alkylation or incomplete cyclization, this process ensures a robust impurity profile that simplifies the analytical validation required by quality control laboratories.
How to Synthesize 1-[(2'-cyanobiphenyl-4-yl)methyl]-2-ethoxy-1H-benzimidazole-7-ethyl formate Efficiently
The synthesis of this key benzimidazole intermediate is achieved through a concise six-step sequence that maximizes yield and operational simplicity. The process begins with the conversion of 2-carboxyl-3-ethyl nitrobenzoate to its corresponding acid chloride using thionyl chloride, followed by transformation into an acyl azide. The critical innovation occurs when this azide reacts with ethanol to form the ethoxycarbonyl protected amine, setting the stage for the subsequent coupling. The nucleophilic substitution with the biphenyl bromide is conducted under mild phase-transfer conditions, ensuring high conversion without degradation. Following the reduction of the nitro group, the final cyclization is triggered by acetic acid, delivering the target intermediate in high purity. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and work-up procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up.
- Convert 2-carboxyl-3-ethyl nitrobenzoate to acid chloride using thionyl chloride, followed by azide formation with sodium azide.
- React the acyl azide with ethanol to form the ethoxycarbonyl amino protected intermediate, replacing traditional tert-butyl protection.
- Perform nucleophilic substitution with 4'-bromomethyl-2-cyanobiphenyl in a heterogeneous system using TBAB, followed by nitro reduction and acid-catalyzed cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers transformative economic and logistical benefits. The primary advantage stems from the drastic simplification of the synthetic route, which directly correlates to reduced manufacturing costs. By eliminating the deprotection step and removing the requirement for expensive cyclization reagents like tetraethyl orthoformate, the bill of materials is significantly optimized. The replacement of tert-butyl alcohol with ethanol, a commodity chemical with stable pricing and abundant availability, further insulates the production cost from market volatility. Additionally, the reduction in the number of unit operations means less energy is consumed for heating, cooling, and solvent recovery, contributing to a lower carbon footprint and reduced utility expenses. The heterogeneous reaction system employed in the nucleophilic substitution step allows for the use of cheaper phase-transfer catalysts in minimal quantities, contrasting with the stoichiometric amounts of strong bases often required in homogeneous systems. These cumulative efficiencies result in a manufacturing process that is not only cost-effective but also environmentally sustainable, aligning with the increasing corporate mandates for green chemistry practices.
- Cost Reduction in Manufacturing: The elimination of the hydrolysis deprotection step and the avoidance of expensive tetraethyl orthoformate lead to substantial savings in raw material costs. The streamlined process reduces solvent consumption and waste disposal fees, while the use of ethanol as a reagent instead of specialized alcohols lowers the overall input cost. Furthermore, the higher overall yield resulting from fewer processing steps means more product is generated per batch, effectively lowering the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals such as ethanol, thionyl chloride, and hydrazine hydrate ensures a stable supply chain不受 limited by niche reagent availability. The robustness of the heterogeneous reaction conditions reduces the risk of batch failures due to sensitive moisture or temperature fluctuations, ensuring consistent output. This reliability allows for better production planning and inventory management, reducing the need for safety stock and minimizing the risk of supply disruptions for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the use of standard industrial solvents like dichloromethane and ethyl acetate which are easily recovered and recycled. The reduction in reaction steps decreases the total volume of waste generated, simplifying effluent treatment and ensuring compliance with stringent environmental regulations. The use of activated carbon in the reduction step aids in decolorization and impurity removal, reducing the need for extensive chromatographic purification which is difficult to scale. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates from pilot plant to multi-ton production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the practical aspects of adopting this technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing manufacturing lines.
Q: How does the ethoxycarbonyl group improve the synthesis of Candesartan intermediates?
A: The ethoxycarbonyl group serves a dual function: it protects the active nitrogen atom during nucleophilic substitution and simultaneously acts as the carbon source for the subsequent cyclization. This eliminates the need for a separate deprotection step and avoids the use of expensive cyclization reagents like tetraethyl orthoformate.
Q: What are the advantages of the heterogeneous nucleophilic substitution method described in the patent?
A: The use of a dichloromethane-water heterogeneous system with a phase-transfer catalyst (TBAB) significantly improves the solubility of the basic catalyst. This allows for milder reaction conditions (room temperature), reduces the consumption of base and catalyst, and simplifies the work-up procedure compared to homogeneous systems.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable. It utilizes common solvents like ethanol and dichloromethane, avoids cryogenic conditions, and reduces the total number of unit operations by combining protection and cyclization precursors, making it ideal for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Candesartan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of optimized synthetic routes like the one described in patent CN102391254A for the production of high-value antihypertensive intermediates. Our team of expert chemists has extensively analyzed this technology and possesses the capability to implement these advanced protocols at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this process are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Candesartan intermediate we deliver meets the highest international standards. We are committed to leveraging our technical expertise to provide our partners with a competitive edge in the global marketplace.
We invite you to collaborate with us to explore the full potential of this cost-effective synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this process can improve your bottom line. We encourage you to contact us to request specific COA data from our recent pilot batches and to discuss route feasibility assessments for your upcoming projects. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally dedicated to driving efficiency and quality in your pharmaceutical supply chain.
