Optimizing Candesartan Cilexetil Production: A Safer, High-Yield Synthetic Route for Global API Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with operational safety, particularly for blockbuster antihypertensive agents like Candesartan Cilexetil. Patent CN101781286B introduces a transformative synthetic methodology that addresses critical bottlenecks in the production of this angiotensin II receptor antagonist. By utilizing 2-amino-3-nitrobenzoic acid as a strategic starting material, the disclosed process circumvents the reliance on hazardous azide chemistry and toxic organotin reagents that have long plagued conventional manufacturing. This technical breakthrough not only streamlines the reaction sequence but also fundamentally alters the impurity profile, offering a cleaner, more efficient route to the active pharmaceutical ingredient (API). For global supply chain stakeholders, this represents a pivotal shift towards sustainable and reliable pharmaceutical intermediates sourcing, ensuring that the final drug product meets stringent regulatory standards while minimizing environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Candesartan Cilexetil has been hindered by complex, multi-step routes that introduce significant safety and efficiency liabilities. Traditional Method One, often cited in prior art, relies on a lengthy reaction scheme that necessitates the use of sodium azide for tetrazole ring formation. Sodium azide is notoriously unstable and explosive, posing severe risks during scale-up and requiring specialized containment infrastructure. Furthermore, these legacy processes frequently employ tributyltin chloride for cyclization, a heavy metal reagent that leaves difficult-to-remove toxic residues in the final API, complicating purification and increasing waste disposal costs. The cumulative effect of these factors is a lower total yield, typically hovering around 25% to 27%, and a production timeline that is vulnerable to regulatory scrutiny regarding heavy metal limits and worker safety.
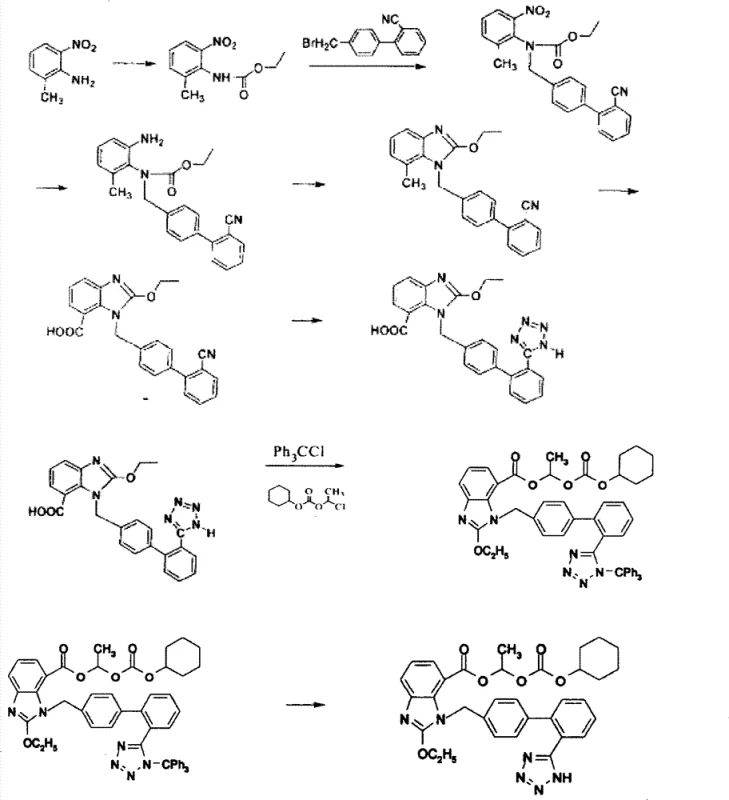
The Novel Approach
In stark contrast, the novel approach detailed in the patent data重构 s the synthetic logic by connecting the tetrazole-containing biphenyl intermediate directly to the amino-benzoate scaffold early in the sequence. This strategic disconnection allows for the formation of the benzimidazole core through a safer cyclization using tetraethyl orthocarbonate, completely bypassing the need for toxic tin catalysts. The process flow is significantly condensed, reducing the number of isolation steps and minimizing material loss at each stage. By shifting the tetrazole introduction to a pre-formed, stable trityl-protected intermediate, the method avoids the thermal instability issues associated with direct azide reactions on the benzoic acid backbone. This results in a more robust process capable of achieving higher purity profiles and a markedly improved total yield, positioning it as a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into the Optimized Synthetic Pathway
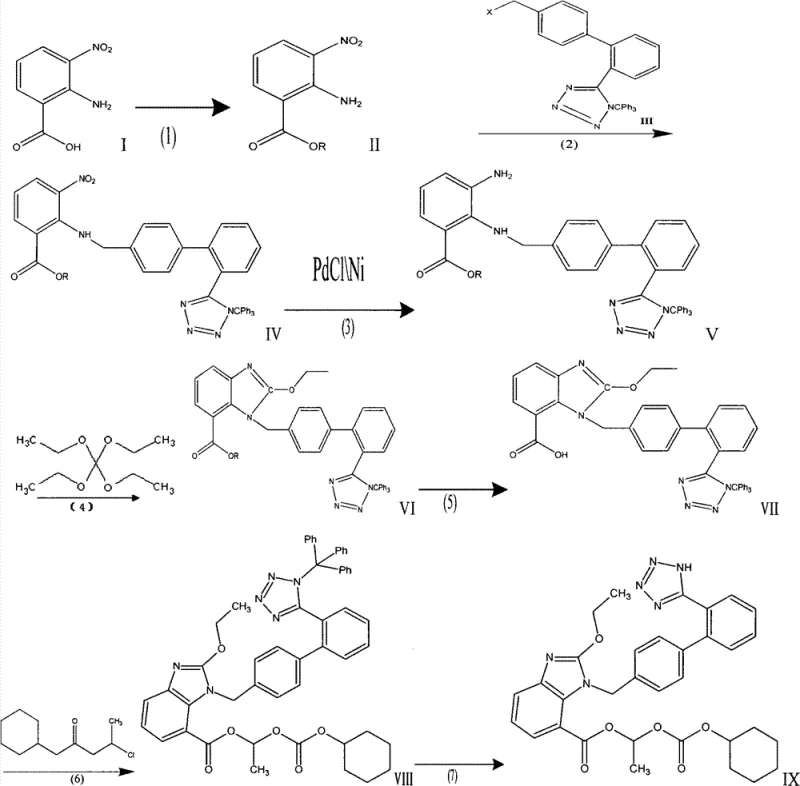
The core of this technological advancement lies in the precise orchestration of the N-alkylation and subsequent cyclization steps. The process initiates with the esterification of 2-amino-3-nitrobenzoic acid, protecting the carboxyl group as a methyl or ethyl ester to prevent side reactions during the subsequent nucleophilic attack. The critical N-alkylation step involves the reaction of this nitro-benzoate with N-trityl-5-(4'-bromomethylbiphenyl-2-yl)tetrazole in an anhydrous polar aprotic solvent like DMF. The use of strong bases such as sodium hydride facilitates the deprotonation of the aniline nitrogen, driving the substitution reaction efficiently. Following this, the nitro group is reduced to an amine, either via catalytic hydrogenation using palladium on carbon or through metal reduction, setting the stage for the intramolecular condensation. The cyclization with tetraethyl orthocarbonate in the presence of acetic acid is a mild yet effective method to close the benzimidazole ring, avoiding the harsh conditions and toxic byproducts associated with traditional thiourea or cyanogen bromide methods.
Impurity control is inherently built into this mechanism by eliminating sources of heavy metal contamination and unstable intermediates. In conventional routes, the thermal decomposition of azido-intermediates can generate brown gases and unpredictable exotherms, leading to variable product quality and the formation of complex polymeric byproducts. The new route's reliance on stable trityl-protected tetrazoles ensures that the biphenyl-tetrazole moiety remains intact throughout the harsh reduction and cyclization conditions. Furthermore, the final deprotection step, which removes the trityl group via acid-catalyzed alcoholysis, is highly selective. This specificity minimizes the formation of des-ethyl or hydrolyzed byproducts that often plague the final esterification stages, thereby simplifying the downstream purification process and ensuring a final product that consistently meets high-purity pharmaceutical intermediates specifications required by major regulatory bodies.
How to Synthesize Candesartan Cilexetil Efficiently
The implementation of this synthesis requires careful attention to anhydrous conditions and temperature control, particularly during the alkylation and cyclization phases. The patent outlines a clear seven-step progression that transforms simple raw materials into the complex prodrug structure with high fidelity. Operators must ensure the complete removal of water before introducing sodium hydride to prevent quenching of the base, and the hydrogenation step requires monitoring to avoid over-reduction or catalyst poisoning. The following guide summarizes the critical operational parameters derived from the patent examples, providing a roadmap for laboratory validation and pilot plant scaling.
- Perform esterification of 2-amino-3-nitrobenzoic acid using methanol or ethanol under sulfuric acid catalysis to form the nitro-benzoate ester.
- Conduct N-alkylation with N-trityl-5-(4'-bromomethylbiphenyl-2-yl)tetrazole in anhydrous DMF using sodium hydride as the base.
- Execute nitro reduction via catalytic hydrogenation (Pd/C) or metal reduction to convert the nitro group to an amine.
- Induce cyclization using tetraethyl orthocarbonate and acetic acid to form the benzimidazole core.
- Hydrolyze the ester group using mineral alkali (NaOH/KOH) to obtain the free carboxylic acid intermediate.
- React with cyclohexyl 1-chloroethyl carbonate to install the prodrug ester moiety.
- Finalize the synthesis by removing the trityl protecting group via acid-catalyzed alcoholysis to yield Candesartan Cilexetil.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into enhanced operational resilience and risk mitigation. The elimination of sodium azide and tributyltin chloride removes two of the most significant regulatory and safety hurdles in API manufacturing. This shift not only lowers the barrier for facility compliance but also drastically reduces the costs associated with hazardous waste treatment and specialized handling equipment. By simplifying the chemical landscape of the production floor, manufacturers can achieve faster turnaround times and more predictable batch cycles, which are essential for maintaining continuity in the global supply of antihypertensive medications.
- Cost Reduction in Manufacturing: The streamlined nature of this process offers substantial economic benefits by reducing the total number of unit operations. Fewer reaction steps mean less solvent consumption, lower energy usage for heating and cooling, and reduced labor hours per kilogram of output. Additionally, the avoidance of expensive and toxic reagents like organotin compounds eliminates the need for costly scavenging resins or complex chromatographic purification steps to meet heavy metal specifications. The improved total yield, rising from the industry average of roughly 25% to over 36% in this method, means that less raw material is required to produce the same amount of finished API, directly lowering the cost of goods sold (COGS) and improving margin potential for generic manufacturers.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term API contracts, and this route enhances reliability by utilizing widely available, commodity-grade starting materials. 2-amino-3-nitrobenzoic acid and standard biphenyl derivatives are produced at scale by multiple global suppliers, reducing the risk of single-source bottlenecks. Furthermore, the robustness of the intermediates, particularly the trityl-protected tetrazole species, allows for safer storage and transportation compared to thermally unstable azides. This stability reduces the likelihood of batch failures due to degraded starting materials, ensuring that production schedules remain on track and delivery commitments to downstream formulation partners are met without interruption.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding green chemistry and waste reduction, this process offers a distinct advantage for commercial scale-up. The absence of explosive hazards allows for larger batch sizes in standard reactor vessels without the need for expensive blast-proof infrastructure. The reduction in toxic waste generation aligns with increasingly strict environmental protection laws, minimizing the liability and permitting delays often associated with expanding production capacity. This 'cleaner' production profile facilitates smoother technology transfer between sites and accelerates the timeline for regulatory approval of new manufacturing facilities, securing the long-term viability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the comparative data and experimental results presented in the patent documentation, offering clarity on how this method outperforms legacy technologies in terms of safety, yield, and scalability.
Q: How does this synthesis route improve safety compared to traditional methods?
A: This method eliminates the use of highly toxic and explosive sodium azide and tributyltin chloride, which are common in conventional tetrazole formation and coupling steps, significantly reducing workplace hazards and environmental waste.
Q: What is the impact of this route on the total yield of Candesartan Cilexetil?
A: By shortening the reaction sequence and optimizing key steps like N-alkylation and cyclization, this process achieves an average total yield of approximately 36-37%, which is a substantial improvement over the 25-27% typical of older methodologies.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the route utilizes standard industrial reagents and avoids difficult-to-handle intermediates like thermally unstable azides, making it highly scalable and compliant with modern GMP and environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Candesartan Cilexetil Supplier
The technical superiority of this non-azide synthetic route underscores the importance of partnering with a CDMO that possesses deep expertise in complex heterocyclic chemistry. NINGBO INNO PHARMCHEM stands at the forefront of API intermediate manufacturing, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific anhydrous and hydrogenation requirements of this process, ensuring that every batch adheres to stringent purity specifications. With rigorous QC labs and a commitment to green chemistry principles, we are uniquely positioned to deliver high-quality Candesartan Cilexetil intermediates that meet the exacting demands of the global pharmaceutical market.
We invite forward-thinking pharmaceutical companies to collaborate with us to optimize their supply chains. By adopting this safer, high-yield methodology, we can jointly achieve significant efficiencies and cost savings. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data, route feasibility assessments, and volume pricing to support your next project milestone.
