Advanced Synthesis of Benzimidazole Esters for Scalable Candesartan Cilexetil Production
Introduction to Novel Benzimidazole Ester Synthesis
The pharmaceutical industry constantly seeks more efficient pathways for producing Angiotensin II receptor antagonists, specifically Candesartan Cilexetil. Patent CN1207287C discloses a pivotal advancement in this domain by introducing a novel benzimidazole ester compound, specifically 2-ethoxy-1-(p-halophenyl) methyl-1H-benzimidazole-7-carboxylate-1-[[(cyclohexoxy)carbonyl] oxy] ethylester. This intermediate serves as a critical building block that enables a more direct and operationally simple route to the final active pharmaceutical ingredient. By strategically delaying the introduction of the highly reactive tetrazole ring until the final stages of synthesis, this technology circumvents the cumbersome protection and deprotection sequences that have historically plagued traditional manufacturing processes. This innovation represents a significant leap forward for manufacturers aiming to optimize their production lines for cardiovascular medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Candesartan Cilexetil, such as those documented in earlier literature like CN1048486C, often rely on constructing the benzimidazole core around a pre-installed tetrazole moiety. Because the tetrazole group possesses high chemical activity and acidity, its early presence in the molecular scaffold necessitates the use of protecting groups to prevent unwanted side reactions during subsequent cyclization and functionalization steps. This requirement adds substantial complexity to the process, increasing the total number of unit operations, consuming additional reagents for protection and deprotection, and ultimately lowering the overall yield. Furthermore, managing the waste streams associated with these extra steps poses environmental challenges and increases the cost of goods sold, making these legacy routes less attractive for modern, cost-sensitive generic drug manufacturing.
The Novel Approach
In stark contrast, the methodology presented in CN1207287C adopts a convergent strategy where the benzimidazole ester intermediate is fully constructed before the tetrazole fragment is attached. This approach allows chemists to utilize robust and inexpensive starting materials, such as substituted o-phenylenediamines and halobenzyl halides, to build the core structure without interference from sensitive heterocycles. The key intermediate, Formula (I), is synthesized via a straightforward hydrolysis and condensation sequence that avoids the need for delicate protecting group chemistry. By reserving the formation of the biaryl bond for the final step via nickel-catalyzed coupling, the process achieves a higher degree of modularity. This flexibility not only simplifies purification protocols but also enhances the overall atom economy of the synthesis, providing a distinct competitive advantage for large-scale production facilities.
Mechanistic Insights into Late-Stage Aryl Cross-Coupling
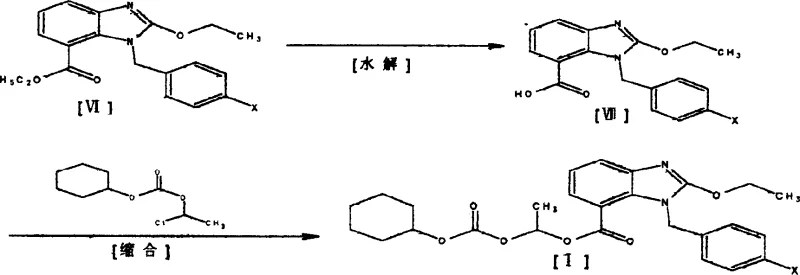
The core of this technological breakthrough lies in the efficient transformation of the benzimidazole carboxylic acid derivative into the final ester intermediate, followed by its coupling with the tetrazole fragment. The synthesis begins with the hydrolysis of the ethyl ester precursor (VI) under alkaline conditions, typically employing sodium or potassium hydroxide in mixed solvent systems like ethanol-water or THF-water. This step cleanly generates the free carboxylic acid (VII), which is then subjected to a condensation reaction with 1-chloroethyl-cyclohexyloxy carbonic ether. This specific acylation installs the crucial prodrug ester moiety required for oral bioavailability in the final drug product. The reaction conditions are mild, often proceeding at room temperature or with slight heating in polar aprotic solvents such as DMF or DMSO, ensuring high conversion rates while minimizing thermal degradation of the sensitive benzimidazole core.
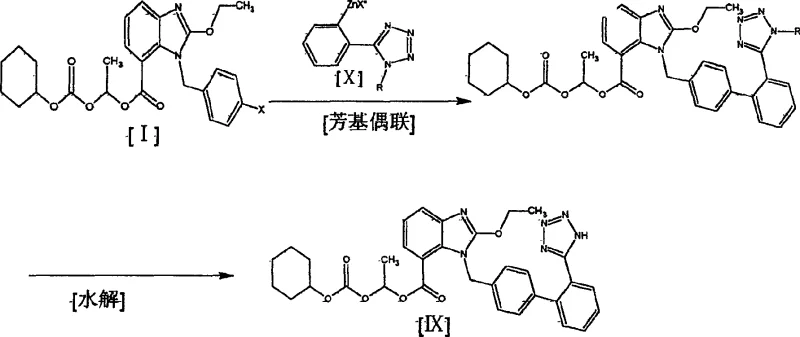
Following the preparation of the benzimidazole ester, the final assembly involves a sophisticated transition metal-catalyzed cross-coupling reaction. The patent details the use of nickel-based catalysts, specifically bis(triphenylphosphine)nickel(II) chloride, to couple the halogenated benzimidazole intermediate with a tetrazole-containing organometallic reagent, such as a Grignard or organozinc species. This mechanistic pathway is superior to older methods because it tolerates the diverse functional groups present on both coupling partners without requiring extensive protection. The use of nickel offers a cost-effective alternative to palladium catalysis, which is critical for reducing raw material costs in bulk manufacturing. Moreover, the reaction can be conducted in common ether solvents like THF or diethyl ether under inert atmosphere, providing a safe and scalable protocol that maintains high stereochemical integrity and minimizes the formation of homocoupling byproducts.
How to Synthesize Benzimidazole Ester Intermediate Efficiently
The synthesis of this high-value intermediate relies on a precise sequence of hydrolysis, condensation, and coupling reactions that have been optimized for industrial feasibility. The process begins with readily available nitrobenzoate derivatives or benzimidazole esters, which are converted into the key halogenated precursor through alkylation and cyclization steps. Once the precursor is secured, the hydrolysis step must be carefully controlled to ensure complete conversion to the acid without hydrolyzing other sensitive ester groups if present. Subsequent condensation with the cyclohexyl carbonate reagent requires strict moisture control to maximize yield. For the final coupling, the preparation of the organometallic tetrazole reagent must be handled under anhydrous conditions to prevent quenching of the reactive species. Detailed standardized operating procedures for each of these critical stages are essential for maintaining batch-to-batch consistency and meeting stringent regulatory purity specifications.
- Hydrolyze the ethyl ester precursor (VI) under alkaline conditions using sodium or potassium hydroxide in alcohol/water mixtures to obtain the carboxylic acid (VII).
- Perform a condensation reaction between the resulting carboxylic acid (VII) and 1-chloroethyl-cyclohexyloxy carbonic ether in polar aprotic solvents like DMF or DMSO.
- Execute a nickel-catalyzed aryl cross-coupling reaction between the benzimidazole ester intermediate and a tetrazole-containing organometallic reagent to finalize the API structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthetic route described in CN1207287C offers profound strategic benefits beyond mere technical elegance. The primary advantage is the drastic simplification of the manufacturing workflow, which directly translates to reduced operational expenditures. By eliminating the need for multiple protection and deprotection steps, manufacturers can significantly reduce the consumption of auxiliary chemicals, solvents, and labor hours. This streamlining effect shortens the overall production cycle time, allowing facilities to increase throughput and respond more agilely to market demand fluctuations. Furthermore, the reliance on commodity chemicals such as nickel salts and common organic solvents, rather than exotic or supply-constrained reagents, mitigates the risk of raw material shortages and price volatility.
- Cost Reduction in Manufacturing: The elimination of protecting group chemistry removes entire stages of the synthesis, thereby reducing the total volume of solvents and reagents required per kilogram of product. This reduction in material intensity leads to substantial cost savings in waste treatment and disposal, as fewer hazardous byproducts are generated. Additionally, the switch to nickel catalysis for the final coupling step avoids the high capital lock-up associated with precious metal catalysts like palladium, further optimizing the cost structure. These cumulative efficiencies result in a more competitive cost of goods sold, enabling suppliers to offer more attractive pricing models to downstream pharmaceutical clients without compromising margin.
- Enhanced Supply Chain Reliability: The versatility of this synthetic route allows for the use of multiple starting materials, such as different halobenzyl halides (chloro, bromo, or iodo derivatives), providing flexibility in sourcing. If the price or availability of one specific halogenated precursor fluctuates, the process can be adapted to use an alternative without redesigning the entire workflow. This redundancy builds resilience into the supply chain, ensuring continuous production even when specific raw material markets experience disruptions. Moreover, the robustness of the reaction conditions means that the process is less susceptible to minor variations in utility quality or environmental conditions, guaranteeing consistent delivery schedules.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plant to commercial scale. The use of aqueous workups and standard crystallization techniques simplifies isolation and purification, reducing the need for complex chromatographic separations that are difficult to scale. From an environmental perspective, the reduced step count and avoidance of toxic protecting groups align with green chemistry principles, lowering the E-factor of the process. This facilitates easier compliance with increasingly stringent environmental regulations regarding solvent emissions and hazardous waste discharge, safeguarding the manufacturer against regulatory risks and potential fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzimidazole ester synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is crucial for R&D teams evaluating technology transfer and for procurement officers assessing supplier capabilities. The responses highlight the practical implications of the chemical mechanisms discussed earlier, bridging the gap between laboratory theory and industrial application.
Q: How does this patent improve upon conventional Candesartan synthesis routes?
A: Conventional methods often introduce the reactive tetrazole moiety early in the synthesis, necessitating complex protection and deprotection steps. This patent utilizes a late-stage coupling strategy, significantly simplifying the operational workflow and reducing the total number of synthetic steps required.
Q: What are the critical reaction conditions for the hydrolysis step?
A: The hydrolysis of the ethyl ester precursor is effectively carried out under alkaline conditions using common bases such as sodium hydroxide or potassium hydroxide. The reaction proceeds efficiently in standard solvent systems including water, alcohols like methanol or ethanol, and ethers such as THF.
Q: Is the nickel-catalyzed coupling suitable for large-scale manufacturing?
A: Yes, the described aryl cross-coupling utilizes robust nickel catalysts like Cl2Ni(PPh3)2 in common ether solvents. This methodology avoids expensive palladium systems where possible and operates under mild thermal conditions, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Candesartan Cilexetil Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust intermediate supply chains in the global pharmaceutical market. Our technical team has extensively analyzed the pathways disclosed in CN1207287C and possesses the expertise to implement this advanced synthesis at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of benzimidazole ester intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage these technological advancements for your product portfolio. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your manufacturing operations. Let us be your trusted partner in navigating the complexities of fine chemical synthesis.
