Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharma
Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharma
The integration of fluorine atoms into heterocyclic scaffolds represents a cornerstone strategy in modern medicinal chemistry, particularly for enhancing the metabolic stability and lipophilicity of drug candidates. As detailed in the groundbreaking patent CN113307778A, a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds has been developed, addressing critical bottlenecks in the synthesis of these valuable pharmacophores. The 1,2,4-triazole ring is a privileged structure found in numerous bioactive molecules, including prominent pharmaceuticals such as Sitagliptin, Maraviroc, and Deferasirox, which serve as vital treatments for diabetes, HIV, and iron overload respectively. The introduction of a trifluoromethyl group at the 3-position further amplifies the physicochemical properties of these molecules, offering superior electronegativity and bioavailability profiles that are highly sought after by R&D teams globally.
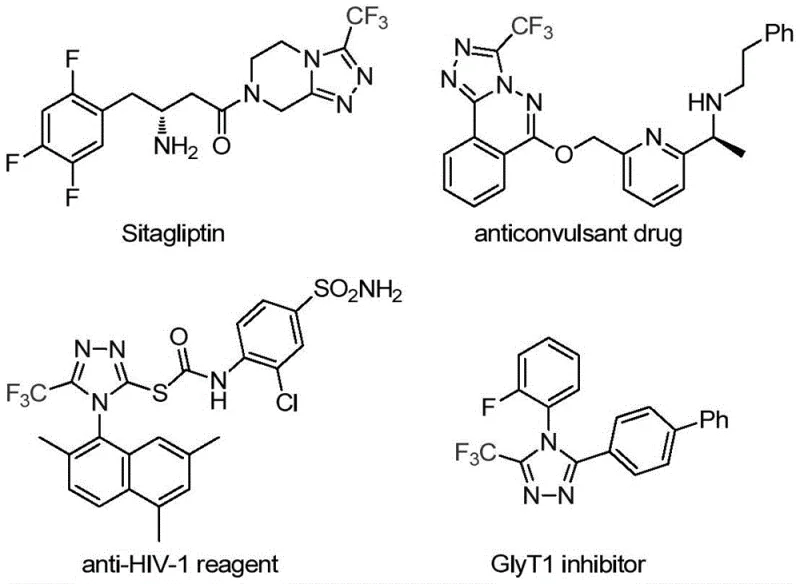
This innovative synthetic route leverages a dual-catalytic system involving molybdenum hexacarbonyl and cuprous acetate to facilitate a efficient cycloaddition reaction. By utilizing functionalized isonitriles and trifluoroethylimidoyl chloride as key building blocks, the process achieves high reaction efficiency under remarkably mild conditions. For procurement managers and supply chain directors, this translates to a robust and reliable pharmaceutical intermediate supplier capability, where the complexity of synthesis does not compromise the consistency or cost-effectiveness of the final output. The ability to access these complex heterocycles through a streamlined process is a significant advancement for the industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant synthetic challenges that hinder large-scale manufacturing. Traditional literature methods often rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, processes that frequently demand harsh reaction conditions and involve multiple tedious steps. Furthermore, alternative approaches utilizing copper-catalyzed multi-component reactions with diazonium salts and trifluorodiazoethane pose severe safety risks due to the explosive nature of diazo compounds, creating substantial liability and operational hazards for production facilities. These legacy methods often suffer from poor atom economy and limited substrate scope, making it difficult to introduce diverse functional groups without drastically reducing yields or requiring extensive purification efforts.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN113307778A offers a transformative solution by employing a molybdenum and copper co-catalyzed cycloaddition of trifluoroethylimidoyl chloride and functionalized isonitrile. This new route operates under mild thermal conditions, typically between 70°C and 90°C, eliminating the need for cryogenic temperatures or high-pressure equipment often associated with older protocols. The use of commercially available and inexpensive starting materials, such as triethylamine and cuprous acetate, drastically simplifies the supply chain logistics and reduces the overall cost of goods sold. Moreover, the reaction demonstrates exceptional functional group tolerance, allowing for the seamless incorporation of electron-donating and electron-withdrawing substituents, thereby enabling the rapid generation of diverse compound libraries for drug discovery programs without the need for protecting group strategies.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this synthesis lies in the synergistic interaction between the molybdenum and copper catalysts, which orchestrate the formation of the triazole ring through a sophisticated mechanistic pathway. Initially, the molybdenum hexacarbonyl acts as a metal activator, coordinating with the functionalized isonitrile to form a reactive metal-isocyanide complex. This activation enhances the nucleophilicity of the isonitrile carbon, priming it for the subsequent cycloaddition event. Simultaneously, the cuprous acetate promotes a [3+2] cycloaddition reaction between the activated isonitrile species and the trifluoroethylimidoyl chloride. This concerted process efficiently constructs the five-membered triazole ring intermediate with high regioselectivity, ensuring that the trifluoromethyl group is positioned precisely at the 3-position of the heterocycle.
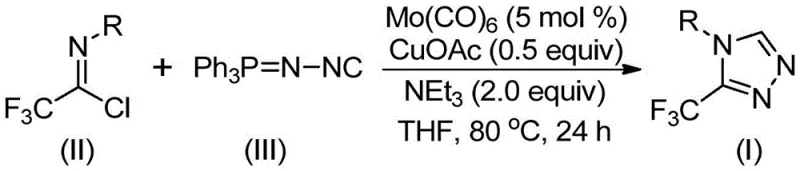
Following the ring closure, the reaction proceeds through a hydrolysis step where water present in the system facilitates the removal of the triphenylphosphine oxide moiety, yielding the final stable 3-trifluoromethyl-substituted 1,2,4-triazole product. This mechanistic elegance ensures minimal formation of side products, resulting in a clean impurity profile that is crucial for meeting stringent pharmaceutical quality standards. The robustness of this catalytic cycle allows for the conversion of various substrates, including those with sterically demanding groups like naphthyl or t-butyl, achieving yields that are consistently high across a broad range of derivatives. For R&D directors, understanding this mechanism provides confidence in the scalability and reproducibility of the process, as the fundamental chemical steps are well-defined and controllable.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it highly attractive for both laboratory-scale optimization and pilot-plant production. The standard protocol involves charging a reaction vessel with the requisite amounts of molybdenum hexacarbonyl, cuprous acetate, triethylamine, and molecular sieves in an aprotic organic solvent, with tetrahydrofuran (THF) being the preferred medium due to its superior solvation properties. To this mixture, the trifluoroethylimidoyl chloride and the functionalized isonitrile are added, and the system is heated to maintain a steady temperature within the optimal range. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to ensure consistent replication of the high yields reported in the patent data.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the crude sample with silica gel, and perform column chromatography purification to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers profound advantages that directly impact the bottom line and operational resilience of chemical manufacturing enterprises. The shift away from hazardous diazonium salts and expensive precious metal catalysts towards a base-metal catalytic system represents a strategic move towards greener and more sustainable chemistry. This transition not only mitigates safety risks but also aligns with increasingly rigorous environmental regulations, reducing the burden of waste disposal and compliance monitoring. For supply chain heads, the reliance on commodity chemicals like triethylamine and cuprous acetate ensures a stable and continuous supply of raw materials, insulating production schedules from the volatility often seen in the market for specialized reagents.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals such as palladium or rhodium, which are traditionally used in cross-coupling reactions, leads to a substantial decrease in catalyst costs. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling, while the simplified post-treatment process minimizes the usage of silica gel and solvents during purification. These factors collectively contribute to a significantly lower cost of production, allowing for more competitive pricing in the global market for pharmaceutical intermediates without sacrificing quality.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically the functionalized isonitriles and trifluoroethylimidoyl chlorides, are either commercially available or can be synthesized in high yields from readily accessible precursors. This accessibility ensures that production lead times are not extended by the scarcity of niche reagents. Additionally, the robustness of the reaction against moisture and air, facilitated by the use of molecular sieves and standard Schlenk techniques, reduces the risk of batch failures, thereby guaranteeing a consistent and reliable delivery schedule for downstream customers.
- Scalability and Environmental Compliance: The process has been validated to operate effectively at the gram level with straightforward scalability potential to kilogram and ton scales. The use of THF as a solvent, which is easily recoverable and recyclable, supports closed-loop manufacturing systems that minimize environmental impact. The absence of toxic heavy metal residues in the final product simplifies the regulatory approval process for API manufacturing, as extensive metal scavenging steps are rendered unnecessary, streamlining the path from intermediate to final drug substance.
Frequently Asked Questions (FAQ)
To assist our partners in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the technical specifications and experimental data provided in the patent documentation. These insights address common concerns regarding reaction scope, safety, and scalability, providing a clear picture of what can be expected when adopting this synthetic route. We encourage technical teams to review these details carefully to understand how this method can be integrated into existing workflows to enhance efficiency and product quality.
Q: What are the key advantages of this Mo/Cu catalytic system over traditional methods?
A: Unlike traditional methods that often require harsh conditions or dangerous diazonium salts, this novel approach utilizes mild temperatures (70-90°C) and readily available starting materials. The co-catalytic system significantly improves reaction efficiency and substrate tolerance, allowing for the synthesis of diverse derivatives with high yields.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly demonstrates that the method can be expanded to gram-level reactions with simple post-treatment procedures. The use of inexpensive catalysts like cuprous acetate and common solvents like THF facilitates easier scale-up compared to precious metal-catalyzed alternatives.
Q: What types of substituents are tolerated on the aromatic ring?
A: The method exhibits broad substrate scope, tolerating various substituents including alkyl groups (methyl, ethyl, t-butyl), alkoxy groups, halogens (fluoro, chloro), and electron-withdrawing groups like nitro. This versatility allows for the design of specific molecular frameworks required for different pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development pipeline, and we are committed to delivering excellence in every batch we produce. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both early-stage clinical trials and full-scale commercial manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every shipment of 3-trifluoromethyl-1,2,4-triazole compounds meets the highest international standards, providing our clients with the confidence they need to advance their projects.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and accelerate your time to market. Contact us today to discuss how we can support your goals as a trusted partner in fine chemical synthesis.
