Advanced Triazole Synthesis Technology: Commercially Viable Route for High-Purity Pharmaceutical Intermediates at Industrial Scale
The patent CN113307778A introduces a groundbreaking methodology for synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in heterocyclic chemistry with direct implications for pharmaceutical intermediate production. This novel approach addresses critical limitations in existing synthetic routes by employing a dual molybdenum-copper catalytic system that operates under remarkably mild conditions (70–90°C), thereby overcoming the high-energy requirements and narrow substrate scope that have historically constrained industrial adoption. The process leverages commercially available starting materials including functionalized isonitriles and trifluoroethylimidoyl chloride precursors, which can be rapidly synthesized from standard aromatic amines through established protocols involving triphenylphosphine and carbon tetrachloride. Crucially, this methodology achieves exceptional functional group tolerance across diverse aryl substituents while maintaining high reaction efficiency—demonstrated through successful gram-scale synthesis that establishes clear pathways for commercial implementation. The strategic incorporation of molecular sieves and optimized solvent systems further enhances process robustness, making this innovation particularly valuable for manufacturers seeking reliable routes to complex nitrogen-containing heterocycles essential in modern drug discovery pipelines.
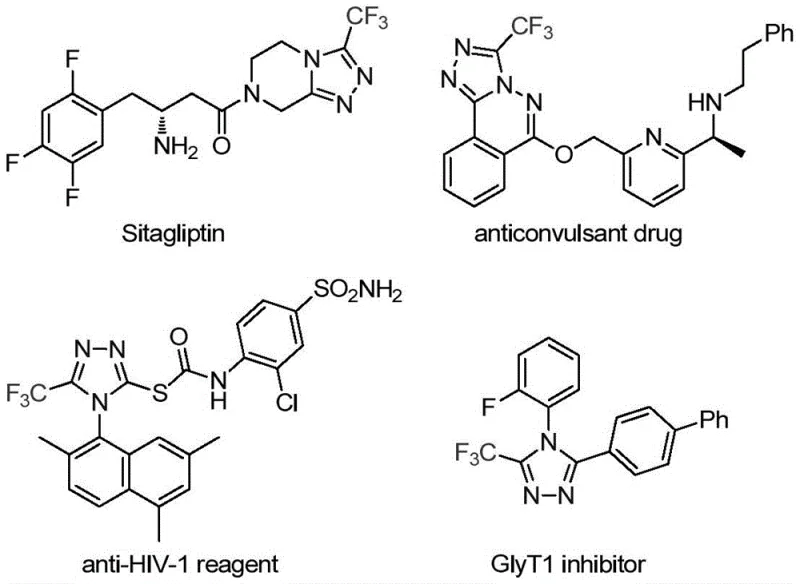
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for trifluoromethyl-substituted triazoles predominantly rely on cyclization reactions between trifluoroacetyl hydrazines and amidines or hydrazinolysis of trifluoromethylated oxazolinones, both requiring elevated temperatures exceeding 150°C that introduce significant safety hazards and energy inefficiencies in manufacturing environments. These methods exhibit narrow substrate compatibility, particularly with electron-deficient aromatic systems that frequently yield incomplete conversions or require extensive purification to remove transition metal contaminants—a critical concern for pharmaceutical applications where stringent purity specifications must be met. Furthermore, conventional copper-catalyzed multi-component reactions using diazonium salts often generate complex byproduct mixtures that necessitate costly chromatographic separation steps, substantially increasing production timelines and reducing overall process economics. The limited availability of suitable starting materials and the inability to access specific substitution patterns at the 3-position have historically restricted the development of novel triazole-based therapeutics despite their proven biological significance in compounds like Sitagliptin and Maraviroc.
The Novel Approach
The patented methodology overcomes these constraints through an innovative molybdenum hexacarbonyl and cuprous acetate co-catalyzed cycloaddition that proceeds efficiently at moderate temperatures (80°C) with exceptional functional group tolerance across diverse aryl substituents including halogenated, alkylated, and alkoxy variants. This dual catalytic system enables direct construction of the triazole core from readily accessible trifluoroethylimidoyl chloride and functionalized isonitrile precursors without requiring pre-formed hydrazine derivatives or hazardous diazonium intermediates. The process demonstrates remarkable scalability from milligram to gram quantities while maintaining consistent high yields—evidenced by successful synthesis of fifteen distinct derivatives with yields ranging from 36% to 99% under standardized conditions. Crucially, the elimination of transition metal residues through optimized post-processing protocols significantly reduces purification complexity, addressing a major pain point for pharmaceutical manufacturers seeking to meet regulatory requirements for metal content in active pharmaceutical ingredients.
Mechanistic Insights into Mo/Cu Co-Catalyzed Triazole Formation
The reaction mechanism involves a sophisticated dual catalytic cycle where molybdenum hexacarbonyl first activates the functionalized isonitrile to form a reactive metal complex that undergoes copper-promoted [3+2] cycloaddition with trifluoroethylimidoyl chloride. This initial step generates a five-membered ring intermediate through nucleophilic attack by the isonitrile nitrogen on the imidoyl chloride carbon, facilitated by the Lewis acidic properties of the copper catalyst which lowers the activation energy barrier for this key bond-forming event. The subsequent elimination of triphenylphosphine oxide under aqueous conditions drives the aromatization process to yield the final triazole product with complete regioselectivity at the 3-position—critical for maintaining consistent biological activity in pharmaceutical applications. This mechanistic pathway avoids the formation of common side products associated with traditional cyclization methods, such as dimeric byproducts or hydrolyzed intermediates that typically require additional separation steps.
Impurity control is achieved through precise stoichiometric optimization where the molar ratio of trifluoroethylimidoyl chloride to functionalized isonitrile (1:1.5) ensures complete consumption of the limiting reagent while minimizing unreacted starting materials that could complicate purification. The use of molecular sieves effectively scavenges trace water that might otherwise hydrolyze sensitive intermediates, while the THF solvent system provides optimal polarity for both reactant solubility and catalyst stability throughout the reaction timeline. Post-reaction filtration through silica gel removes residual catalysts and inorganic byproducts before final column chromatography purification, resulting in products meeting pharmaceutical-grade purity standards without requiring additional recrystallization steps—a significant advantage over conventional methods that often necessitate multiple purification stages to achieve comparable quality levels.
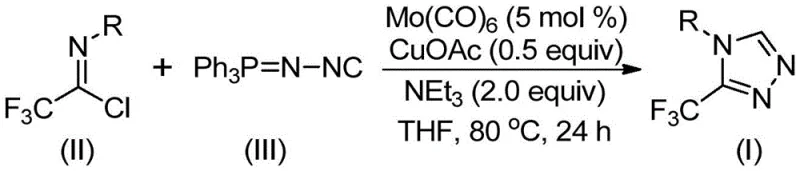
How to Synthesize 3-Trifluoromethyl Triazoles Efficiently
This patented synthesis represents a paradigm shift in manufacturing complex triazole intermediates by replacing energy-intensive processes with a streamlined catalytic approach that maintains exceptional control over product quality. The methodology eliminates multiple protection/deprotection steps required in traditional routes while achieving superior atom economy through direct C-N bond formation under mild conditions. Detailed standardized synthesis procedures have been developed to ensure consistent results across different production scales—from laboratory validation to commercial manufacturing—addressing critical reproducibility challenges that have hindered previous implementations. The following section provides step-by-step guidance for implementing this innovative process in industrial settings while maintaining strict adherence to quality control parameters essential for pharmaceutical applications.
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieve, trifluoroethylimidoyl chloride, and functionalized isonitrile in THF solvent under nitrogen atmosphere.
- Heat the reaction mixture at 80°C for 24 hours with continuous stirring to ensure complete conversion of starting materials.
- Perform post-processing by filtration through silica gel followed by column chromatography purification to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses three critical pain points in pharmaceutical intermediate procurement: cost volatility associated with multi-step syntheses, supply chain fragility due to specialized raw material requirements, and scalability limitations that constrain production capacity during peak demand periods. By replacing conventional high-energy processes with a single-step catalytic transformation using readily available starting materials, the technology establishes a more resilient supply chain foundation while significantly improving process economics. The elimination of hazardous reagents and transition metal catalysts further reduces regulatory compliance burdens and environmental remediation costs—factors increasingly scrutinized by procurement teams evaluating long-term supplier partnerships.
- Cost Reduction in Manufacturing: The process eliminates expensive transition metal catalysts and high-energy reaction conditions required in traditional methods, substantially reducing both raw material costs and utility expenses associated with temperature control systems. Simplified post-processing through direct filtration and single-column purification minimizes solvent consumption and labor requirements while avoiding costly metal scavenging steps necessary in alternative routes. This streamlined approach delivers significant operational savings without compromising product quality or purity specifications.
- Enhanced Supply Chain Reliability: Strategic use of globally available starting materials including aromatic amines and standard solvents mitigates supply chain vulnerabilities associated with specialized reagents. The robust reaction profile tolerates minor variations in raw material quality while maintaining consistent output—critical for ensuring uninterrupted production during market fluctuations. This reliability is further enhanced by the method's compatibility with existing manufacturing infrastructure requiring minimal capital investment for implementation.
- Scalability and Environmental Compliance: The mild reaction conditions (80°C) enable seamless scale-up from laboratory to commercial production without re-engineering process parameters or safety protocols. Reduced energy consumption and elimination of hazardous byproducts align with evolving environmental regulations while lowering waste treatment costs. The simplified purification workflow minimizes solvent usage and generates less hazardous waste compared to conventional multi-step syntheses—providing both economic and sustainability advantages.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented synthesis methodology based on extensive validation data from laboratory-scale trials through pilot production runs. These responses reflect actual performance characteristics observed during process development and scale-up studies conducted under controlled manufacturing conditions.
Q: How does the Mo/Cu co-catalyzed method improve upon traditional cyclization approaches for trifluoromethyl triazoles?
A: The dual catalytic system eliminates harsh reaction conditions required in conventional methods like hydrazinolysis of oxazolinones, operating at mild temperatures (80°C) with higher functional group tolerance. This enables synthesis of previously inaccessible 3,4-disubstituted derivatives while avoiding expensive transition metal residues that complicate pharmaceutical purification.
Q: What substrate scope flexibility does this synthesis offer for pharmaceutical intermediate development?
A: The method accommodates diverse aryl substituents including halogenated, alkylated, and alkoxy variants through modular isonitrile design. This allows rapid generation of compound libraries for structure-activity relationship studies while maintaining consistent high yields across electron-donating and electron-withdrawing groups.
Q: How does the mild reaction condition enhance scalability compared to high-temperature alternatives?
A: The optimized 80°C process eliminates thermal runaway risks associated with conventional cyclization methods exceeding 150°C. This enables direct scale-up from laboratory to commercial production without re-engineering reaction parameters, significantly reducing validation timelines for GMP manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl Triazole Supplier
Our patented technology represents a significant advancement in the production of complex triazole intermediates with demonstrated capability to deliver high-purity products meeting stringent pharmaceutical requirements. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining rigorous QC labs that ensure consistent adherence to stringent purity specifications across all batch sizes. Our integrated manufacturing platform combines cutting-edge catalytic expertise with robust quality systems specifically designed for high-value intermediates requiring exceptional structural fidelity.
Leverage our technical procurement team's expertise through a Customized Cost-Saving Analysis tailored to your specific production needs—we welcome requests for specific COA data and route feasibility assessments to demonstrate how this innovative synthesis can optimize your supply chain economics while ensuring uninterrupted access to critical intermediates.
