Revolutionizing Quinolone Core Synthesis with Green Chemistry and Scalable Efficiency
Revolutionizing Quinolone Core Synthesis with Green Chemistry and Scalable Efficiency
The pharmaceutical industry constantly seeks robust, scalable, and environmentally sustainable pathways for producing critical antibiotic intermediates. A pivotal advancement in this domain is detailed in patent CN101838238A, which discloses a novel method for synthesizing quinolone main ring compounds. This technology addresses the longstanding bottlenecks associated with traditional quinolone synthesis, specifically the management of dimethylamine byproducts and the equilibrium limitations of amine exchange reactions. By integrating a unique CO2 trapping mechanism, this process not only drives the reaction forward to achieve superior yields but also transforms a hazardous waste stream into a recyclable resource. For global procurement and R&D teams, this represents a significant leap towards greener, more cost-effective pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of quinolone antibiotics, such as levofloxacin, has relied on a sequence involving the formation of alpha-substituted aroyl-beta-dimethylamino acrylate followed by amine exchange and cyclization. However, this conventional pathway suffers from severe thermodynamic and environmental drawbacks. The amine exchange reaction is inherently reversible, often failing to proceed to completion due to molecular equilibrium constraints. Furthermore, the byproduct generated is dimethylamine, which poses a significant disposal challenge. Traditional methods typically convert this dimethylamine into dimethylamine hydrochloride using hydrochloric acid. Unfortunately, this salt is notoriously difficult to degrade in biochemical treatment pools, leading to persistent nitrogenous pollution. Alternatively, drying the salt yields a low-purity solid with little utility, ultimately resulting in substantial three-waste generation that complicates regulatory compliance and increases operational overhead.
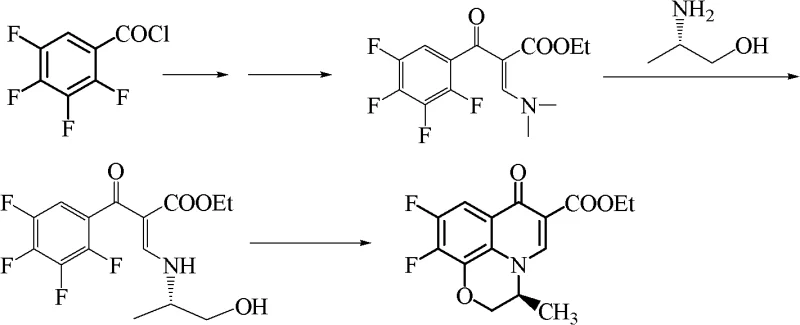
The Novel Approach
The methodology outlined in CN101838238A fundamentally reengineers this workflow by introducing carbon dioxide (CO2) directly into the reaction system. Instead of allowing dimethylamine to accumulate or forming difficult-to-treat salts, the process facilitates the formation of a dimethylamine complex compound (Formula B) through reaction with CO2. This complex possesses a lower boiling point and distinct physical properties that allow it to be easily separated via distillation. Crucially, this removal of the byproduct shifts the chemical equilibrium of the amine exchange reaction towards the product side, significantly enhancing conversion rates. The recovered complex can then be mechanically recycled to regenerate starting materials, creating a closed-loop system that minimizes raw material consumption and eliminates the discharge of unmanageable amine nitrogen pollutants.
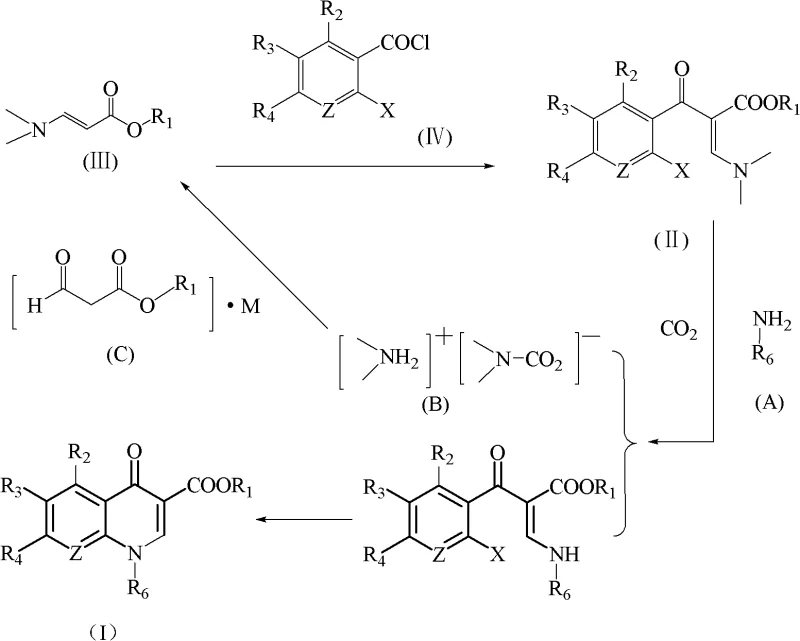
Mechanistic Insights into CO2-Mediated Amine Exchange and Cyclization
The core innovation lies in the precise manipulation of reaction equilibria through in-situ byproduct sequestration. During the initial stage, the alpha-substituted aroyl-beta-dimethylamino acrylate (Formula II) reacts with a primary amine (Formula A) in an organic solvent. As the reaction progresses, dimethylamine is liberated. By feeding CO2 gas at a controlled pressure of 1 to 8 atm, the free dimethylamine is instantly captured to form the carbamate-like complex [Me2NH2]+[Me2N-CO2]-. This chemical transformation effectively lowers the concentration of free dimethylamine in the solution, adhering to Le Chatelier's principle to drive the substitution reaction to near-completion. Following this, the reaction mixture is distilled to recover the volatile complex, leaving behind a residue mother liquor rich in the desired amino-substituted intermediate ready for the next step.
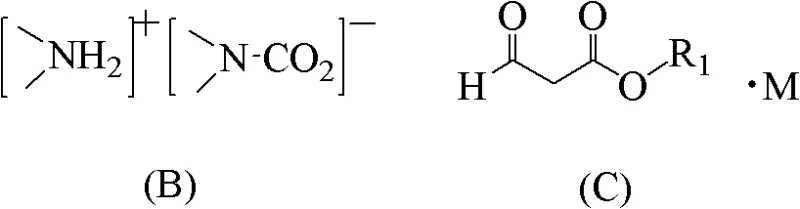
Subsequent cyclization is achieved by treating the residue mother liquor with an acid-binding agent, such as potassium carbonate or tributylamine, at elevated temperatures ranging from 30°C to 160°C. The choice of acid-binding agent is critical for impurity control; for instance, using tributylamine forms a hydrochloride salt that separates cleanly from the organic layer during washing, allowing for the recovery of high-purity tributylamine for reuse. This meticulous control over reaction conditions and byproduct management ensures that the final quinolone main ring compound (Formula I) is obtained with high purity and minimal side products, addressing the stringent quality requirements of high-purity pharmaceutical intermediates needed for downstream API synthesis.
How to Synthesize Quinolone Main Ring Compound Efficiently
Implementing this synthesis route requires careful attention to solvent selection, temperature control, and gas feeding protocols to maximize the efficiency of the CO2 trapping mechanism. The process is designed to be adaptable, allowing for the use of various organic solvents like toluene, DMF, or DMSO depending on the specific substrate solubility and thermal requirements. The following guide outlines the standardized operational framework derived from the patent examples, ensuring reproducibility and safety during scale-up. For detailed standard operating procedures and specific parameter optimization, please refer to the technical guide below.
- Perform amine exchange reaction between alpha-substituted aroyl-beta-dimethylaminoethyl acrylate and an amine in organic solvent, feeding CO2 to trap dimethylamine byproduct.
- Distill the reaction liquid to recover the dimethylamine complex compound (Formula B) and obtain the residual mother liquor containing the intermediate.
- Subject the residual mother liquor to a ring-closure reaction with an acid-binding agent at elevated temperatures to yield the final quinolone main ring compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits beyond mere chemical elegance. The primary value driver is the drastic simplification of waste management protocols. By eliminating the generation of non-biodegradable dimethylamine hydrochloride wastewater, manufacturers can significantly reduce the costs associated with environmental compliance and effluent treatment. Furthermore, the ability to recycle the dimethylamine complex compound means that the effective consumption of amine reagents is lowered, directly contributing to cost reduction in API manufacturing. This circular economy approach insulates the supply chain from volatility in raw material pricing and ensures a more predictable cost structure for long-term contracts.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal catalysts and the recycling of amine byproducts lead to substantial operational savings. By converting a waste liability into a reusable asset, the overall material cost per kilogram of the intermediate is optimized. Additionally, the simplified purification steps reduce solvent consumption and energy usage during distillation and crystallization, further enhancing the economic viability of the process without compromising on yield or quality standards.
- Enhanced Supply Chain Reliability: Traditional methods often face bottlenecks due to waste disposal limits or the availability of specific reagents for salt formation. This novel method utilizes common gases like CO2 and standard organic bases, which are readily available globally. The robustness of the reaction against equilibrium limitations ensures consistent batch-to-batch yields, reducing the risk of production delays. This reliability is crucial for maintaining the continuity of supply for critical antibiotic ingredients in the global market.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates. The use of gas-liquid reactions and distillation is well-understood in large-scale chemical engineering, facilitating a smooth transition from pilot plant to multi-ton production. Moreover, the green chemistry credentials of the process—specifically the reduction of nitrogenous waste—align perfectly with increasingly stringent global environmental regulations, future-proofing the supply chain against regulatory shocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinolone synthesis technology. These insights are derived directly from the experimental data and claims within patent CN101838238A, providing a factual basis for evaluating its potential integration into your manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: How does this method solve the environmental issue of dimethylamine waste?
A: The process introduces CO2 gas during the amine exchange reaction, converting the volatile and polluting dimethylamine byproduct into a stable, low-boiling dimethylamine complex compound (Formula B). This complex can be easily distilled off and recycled, preventing the formation of non-biodegradable dimethylamine hydrochloride wastewater.
Q: What represents the key advantage for large-scale manufacturing?
A: The ability to recycle the dimethylamine complex allows the reaction to proceed cyclically. This significantly reduces raw material consumption and waste treatment costs, making the commercial scale-up of complex quinolone intermediates more economically viable and environmentally compliant.
Q: Can this synthesis method be applied to different quinolone antibiotics?
A: Yes, the patent demonstrates versatility across various substrates. It has been successfully applied to synthesize main ring compounds for Gatifloxacin, Levofloxacin, Ciprofloxacin, and Rufloxacin by adjusting the specific acyl chloride and amine reactants while maintaining the core CO2 trapping mechanism.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolone Intermediate Supplier
The technological breakthroughs described in CN101838238A highlight the immense potential for optimizing quinolone production, yet translating patent chemistry into commercial reality requires deep expertise. NINGBO INNO PHARMCHEM stands as a premier partner in this endeavor, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific pressure and temperature requirements of CO2-mediated reactions, ensuring that the theoretical benefits of this green synthesis are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for pharmaceutical grade APIs.
We invite you to leverage our technical capabilities to enhance your supply chain resilience. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing processes can deliver the high quality and reliability your organization demands.
