Revolutionizing Quinolone Intermediate Production: A Streamlined 2-Step Synthetic Route for Commercial Scale-Up
Introduction to Advanced Quinolone Synthesis Technology
The landscape of antibiotic intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic pathways. Patent CN100999493A introduces a groundbreaking methodology for the synthesis of quinolone main ring compounds, which serve as the critical backbone for a vast array of fluoroquinolone antibiotics. This technology addresses long-standing bottlenecks in the industry by condensing what was traditionally a complex, multi-step sequence into a highly streamlined two-step process. By leveraging a novel acylation-amination strategy followed by a robust cyclization step, this approach not only enhances atom economy but also drastically reduces the environmental footprint associated with legacy manufacturing protocols. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and economically viable production models that align with modern regulatory standards.
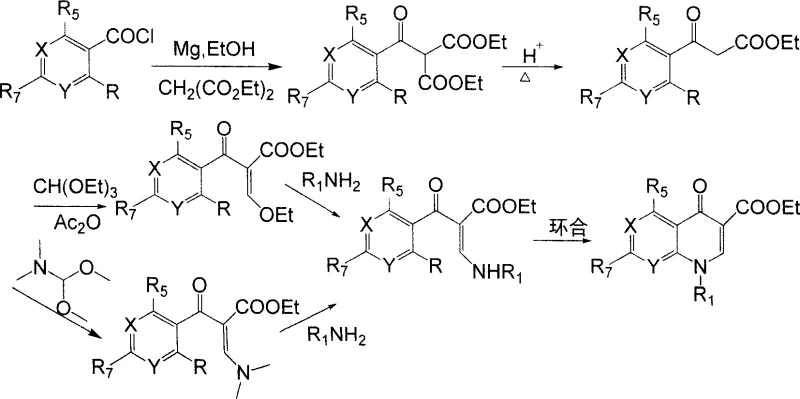
The core innovation lies in the direct conversion of o-haloaroyl chlorides and alkali metal salts of formylacetic esters into alpha-substituted aroyl-beta-substituted amino acrylates, which are subsequently cyclized to form the target quinolone structure. This eliminates the necessity for protective group chemistry that has historically plagued quinolone synthesis, specifically avoiding the use of dimethylamine as a protecting agent. The implications for supply chain stability are profound, as fewer steps translate to reduced lead times and lower cumulative loss of material. As we delve deeper into the technical specifics, it becomes evident that this patent offers a reliable quinolone intermediate supplier with a distinct competitive advantage in terms of both purity and cost-efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of quinolone intermediates has been fraught with inefficiencies and safety hazards that complicate large-scale production. Traditional routes, particularly those prevalent in earlier Chinese industrial practices, often necessitated a cumbersome five-step sequence that suffered from poor atom economy and generated substantial quantities of hazardous waste. A major pain point in these legacy processes was the reliance on dangerous reagents such as sodium hydride (NaH) and triethyl orthoformate, which pose significant fire and explosion risks in a manufacturing environment. Furthermore, the decarboxylation and esterification steps inherent in these older methods frequently resulted in difficult product separations and low overall yields, driving up the cost of goods sold.
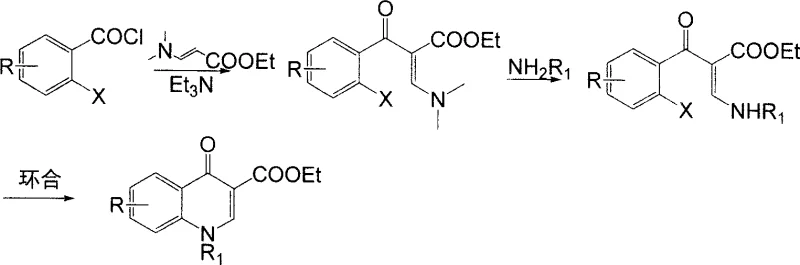
Even more recent advancements, such as the technology described in foreign patent US6229017 by Bayer A.G., while an improvement, still retained critical flaws that limited their commercial optimality. This prior art utilized N,N-dimethylamino ethyl propenoate and required a dimethylamine protection strategy, which inevitably led to the generation of dimethylamine waste during the deprotection phase. This not only created environmental compliance issues due to atmospheric emissions but also added unnecessary complexity to the purification workflow. The reliance on such protective groups inherently lowers the utilization rate of raw materials and extends the production cycle, creating bottlenecks for procurement managers seeking to optimize inventory turnover and reduce working capital tied up in WIP.
The Novel Approach
In stark contrast to these convoluted legacy pathways, the methodology outlined in CN100999493A offers a elegantly simple yet powerful alternative that bypasses the need for protective groups entirely. By directly reacting o-haloaroyl chlorides with sodium formylacetate in an organic solvent, the process achieves acylation and subsequent amination in a single pot operation to generate the key acrylate intermediate. This strategic simplification removes the entire protection-deprotection cycle, thereby eliminating the associated waste streams and reducing the consumption of auxiliary reagents. The result is a process that is not only chemically superior in terms of selectivity but also operationally simpler, requiring less equipment and shorter reaction times to achieve high-purity outputs.
The second stage of this novel approach involves a straightforward cyclization reaction facilitated by mild acid-binding agents such as potassium carbonate or triethylamine. This step proceeds efficiently at temperatures ranging from 30°C to 220°C, offering flexibility in process optimization depending on the specific substrate. The ability to avoid carcinogenic and flammable materials like dimethyl sulfate and sodium metal further underscores the safety profile of this new route. For supply chain heads, this translates to a more resilient manufacturing process that is less susceptible to disruptions caused by the strict regulation or scarcity of hazardous controlled substances, ensuring a steady flow of high-quality intermediates to downstream API manufacturers.
Mechanistic Insights into Acylation-Amination and Cyclization
The mechanistic elegance of this synthesis lies in the precise orchestration of nucleophilic attacks and elimination reactions that construct the quinolone core with high fidelity. Initially, the enolate derived from the alkali metal salt of formylacetic ester acts as a potent nucleophile, attacking the electrophilic carbonyl carbon of the o-haloaroyl chloride. This acylation event is immediately followed by the introduction of a primary amine, which displaces the ethoxy group or reacts with the intermediate to form the beta-amino acrylate structure. This tandem sequence is crucial as it sets up the necessary geometric arrangement for the subsequent intramolecular cyclization, ensuring that the nitrogen atom is correctly positioned to attack the carbonyl carbon of the benzoyl group.
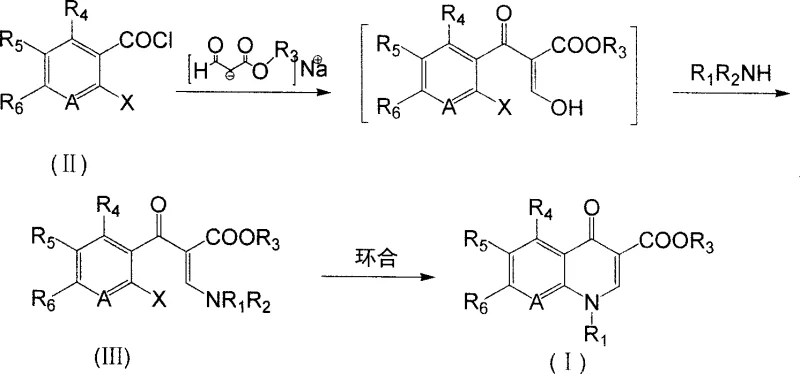
Following the formation of the alpha-substituted aroyl-beta-amino acrylate, the cyclization step is driven by the presence of a base which deprotonates the amine nitrogen, enhancing its nucleophilicity. This activated nitrogen then performs an intramolecular nucleophilic aromatic substitution or addition-elimination sequence onto the aromatic ring, displacing the halogen substituent ortho to the carbonyl group. This ring-closing step is thermodynamically favorable and results in the formation of the stable quinolone-3-carboxylate skeleton. The robustness of this mechanism allows for a wide scope of substituents on the aromatic ring and the amine component, making it a versatile platform for generating diverse libraries of quinolone derivatives without compromising on yield or purity specifications.
How to Synthesize Quinolone Intermediates Efficiently
Implementing this synthetic route in a commercial setting requires careful attention to solvent selection and stoichiometric ratios to maximize the benefits of the atom-economical design. The process typically begins with the suspension of sodium formylacetate in a solvent such as toluene, followed by the controlled addition of the acyl chloride at moderate temperatures to manage exotherms. Once the acylation is complete, the amine is introduced, and the mixture is heated to facilitate the condensation and water removal, often using a Dean-Stark trap. The resulting intermediate is then subjected to cyclization conditions using a base like potassium carbonate, followed by standard workup procedures involving filtration and recrystallization to achieve pharmaceutical-grade purity.
- Perform acylation by reacting o-haloaroyl chloride with sodium formylacetate in an organic solvent like toluene at 25-35°C, followed by amination with a primary amine.
- Purify the resulting alpha-substituted aroyl-beta-amino acrylate intermediate through filtration and washing to remove solid salts.
- Execute the ring-closure reaction by heating the purified intermediate with a base such as K2CO3 in toluene under reflux for 6-8 hours to obtain the final quinolone compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this streamlined synthesis protocol offers compelling advantages that directly impact the bottom line and operational resilience of chemical manufacturing enterprises. By eliminating multiple synthetic steps and hazardous reagents, the process significantly reduces the consumption of raw materials and the associated costs of waste disposal and environmental compliance. This efficiency gain is particularly valuable in the context of cost reduction in pharmaceutical manufacturing, where margin pressures are intense and the price of goods is a critical determinant of market competitiveness. The simplified workflow also reduces the burden on quality control laboratories, as fewer intermediates need to be tested and validated, accelerating the overall release time for finished products.
- Cost Reduction in Manufacturing: The removal of the dimethylamine protection and deprotection steps fundamentally alters the cost structure of quinolone production by eliminating the need for expensive protecting group reagents and the solvents required to handle them. Furthermore, the avoidance of hazardous materials like sodium hydride removes the necessity for specialized handling equipment and rigorous safety protocols that drive up operational expenditures. This leaner chemical footprint allows manufacturers to achieve substantial cost savings through reduced utility consumption and lower waste treatment fees, creating a more profitable production model that can withstand market volatility.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as toluene, potassium carbonate, and readily available acyl chlorides ensures that the supply chain remains robust and less vulnerable to geopolitical or logistical disruptions. Unlike processes dependent on niche or highly regulated reagents, this method utilizes materials that are widely sourced and easily stocked, minimizing the risk of production stoppages due to raw material shortages. This stability is crucial for maintaining consistent delivery schedules to API customers, fostering long-term partnerships based on trust and reliability in the face of global supply chain challenges.
- Scalability and Environmental Compliance: The inherent safety of avoiding pyrophoric reagents and carcinogenic alkylating agents makes this process exceptionally well-suited for scale-up from pilot plant to multi-ton commercial production. The reduced generation of hazardous waste aligns perfectly with increasingly stringent environmental regulations, mitigating the risk of fines and shutdowns while enhancing the corporate sustainability profile. This green chemistry approach not only future-proofs the manufacturing asset but also appeals to environmentally conscious stakeholders and customers who prioritize sustainable sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented quinolone synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear picture of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios and for procurement professionals assessing the long-term value proposition.
Q: How does this new synthetic route improve upon traditional quinolone manufacturing methods?
A: The patented process reduces the synthesis from five steps to just two, eliminating the need for hazardous reagents like sodium hydride and dimethyl sulfate, thereby significantly improving atom economy and operational safety.
Q: What are the typical yields achieved with this cyclization method?
A: Experimental embodiments within the patent demonstrate consistent yields ranging from 90% to 93% for key intermediates such as the Gatifloxacin precursor, indicating high process efficiency.
Q: Is this process scalable for industrial production of antibiotic intermediates?
A: Yes, the use of common organic solvents like toluene and standard bases like potassium carbonate, along with moderate temperature requirements, makes this route highly suitable for commercial scale-up and continuous manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthetic methodology and have integrated similar high-efficiency protocols into our own manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical, large-volume operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of quinolone intermediate meets the exacting standards required by global pharmaceutical regulators, providing our partners with absolute confidence in product quality.
We invite forward-thinking organizations to collaborate with us to leverage these technological advancements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and obtain specific COA data and route feasibility assessments. Let us help you optimize your antibiotic intermediate sourcing strategy with a partner dedicated to innovation, quality, and sustainable growth in the fine chemical sector.
