Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Production: A Safe, Scalable, and Metal-Free Commercial Route
Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Production: A Safe, Scalable, and Metal-Free Commercial Route
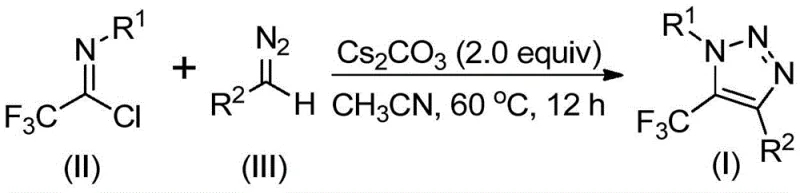
The landscape of heterocyclic chemistry is constantly evolving to meet the rigorous demands of modern pharmaceutical and agrochemical manufacturing. A significant breakthrough in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift away from hazardous traditional methodologies, offering a streamlined, base-promoted pathway that eliminates the need for transition metal catalysts and explosive azide reagents. For industry leaders seeking a reliable pharmaceutical intermediate supplier, this innovation addresses critical pain points regarding safety, purity, and scalability. The ability to construct these valuable fluorinated heterocycles under mild conditions using commercially available starting materials positions this technology as a cornerstone for next-generation drug discovery and process development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,3-triazoles has been fraught with significant operational and safety challenges that hinder efficient commercial scale-up. The most prevalent literature methods typically involve copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation, or organocatalytic 1,3-dipolar cycloadditions involving azides and trifluoromethyl ketones. These conventional routes suffer from severe drawbacks, primarily the reliance on organic azides, which are notoriously toxic and possess high explosion risks, necessitating specialized containment facilities and driving up insurance and handling costs. Furthermore, the use of copper catalysts introduces the risk of heavy metal contamination, requiring extensive and costly downstream purification processes to meet stringent regulatory limits for active pharmaceutical ingredients (APIs). The combination of hazardous reagents and difficult purification creates a bottleneck for cost reduction in electronic chemical manufacturing and pharma sectors alike.
The Novel Approach
In stark contrast, the methodology described in patent CN113121462B offers a transformative solution by utilizing cheap and readily available diazo compounds and trifluoroethylimidoyl chloride as starting materials. This base-promoted synthesis operates without the participation of metals, azides, or specialized trifluoromethylating reagents, fundamentally altering the safety profile of the production line. The reaction proceeds efficiently in common aprotic solvents like acetonitrile at moderate temperatures ranging from 50 to 70°C. By bypassing the need for explosive azides and toxic copper catalysts, this approach not only enhances workplace safety but also simplifies the workflow, making it highly suitable for the commercial scale-up of complex polymer additives and fine chemicals. The robustness of this method allows for easy extension to gram-scale and beyond, providing a viable pathway for industrial application that was previously difficult to achieve with older technologies.
Mechanistic Insights into Base-Promoted Cyclization
Understanding the mechanistic underpinnings of this reaction is crucial for R&D directors focused on impurity control and process optimization. The transformation likely initiates with a base-promoted intermolecular nucleophilic addition-elimination process where the diazo compound attacks the trifluoroethylimidoyl chloride. This step facilitates the formation of a critical carbon-carbon bond, setting the stage for the subsequent ring closure. Following this initial coupling, the intermediate undergoes an intramolecular 5-endo-dig cyclization reaction. This specific cyclization mode is energetically favorable under the promoted conditions and leads directly to the formation of the final 5-trifluoromethyl substituted 1,2,3-triazole core. The absence of metal coordination complexes simplifies the reaction coordinate, reducing the likelihood of metal-induced side reactions that often complicate purification profiles in transition-metal catalyzed systems.
From an impurity management perspective, the use of cesium carbonate as a mild inorganic base ensures a clean reaction environment. Unlike strong organic bases or reactive metal catalysts, cesium carbonate minimizes the formation of decomposition byproducts often associated with sensitive diazo species. The reaction demonstrates excellent functional group tolerance, accommodating various substituents on both the aryl and aroyl moieties, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens. This tolerance suggests that the electronic nature of the substrates does not drastically alter the mechanistic pathway, allowing for a predictable impurity spectrum across different derivatives. Such predictability is invaluable for establishing robust quality control protocols in high-purity OLED material or API intermediate production.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The operational simplicity of this synthesis makes it an attractive candidate for immediate adoption in pilot and production plants. The process involves mixing the key reactants—trifluoroethylimidoyl chloride and the specific diazo compound—with cesium carbonate in an organic solvent. The reaction is then heated to facilitate the cyclization. Detailed standard operating procedures regarding stoichiometry, specific solvent volumes, and workup techniques are essential for reproducibility. For a comprehensive guide on executing this synthesis with maximum efficiency and yield, please refer to the standardized protocol below.
- Combine cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature range of 50-70°C and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this metal-free, azide-free methodology translates into tangible strategic advantages beyond mere chemical curiosity. The elimination of hazardous azides significantly reduces the regulatory burden and safety infrastructure costs associated with storage and transport. Moreover, the removal of copper catalysts eradicates the need for expensive scavenger resins or complex extraction protocols designed to lower heavy metal residues, directly impacting the bottom line through simplified downstream processing. This streamlining of the manufacturing workflow ensures a more reliable supply chain with reduced lead times for high-purity intermediates, as fewer unit operations are required to bring the product to specification.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substitution of expensive and hazardous reagents with commodity chemicals. By avoiding the use of precious metal catalysts like copper, the raw material costs are inherently lower. Additionally, the simplified purification process, which avoids the need for rigorous metal removal steps, reduces solvent consumption and waste disposal costs. The overall process efficiency means that less energy and labor are required per kilogram of product, resulting in substantial cost savings that can be passed down the supply chain or reinvested in further R&D initiatives.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, specifically trifluoroethylimidoyl chloride and various diazo compounds, are either commercially available or easily synthesized from abundant precursors like aromatic amines and acid chlorides. This accessibility mitigates the risk of supply disruptions often caused by the limited availability of specialized organometallic catalysts or unstable azide reagents. A supply chain built on stable, shelf-stable reagents is far more resilient to market fluctuations, ensuring consistent delivery schedules for critical pharmaceutical and agrochemical projects.
- Scalability and Environmental Compliance: The mild reaction conditions (50-70°C) and the use of common solvents like acetonitrile make this process highly amenable to scale-up from laboratory to multi-ton production. The absence of toxic heavy metals and explosive azides aligns perfectly with modern green chemistry principles and increasingly strict environmental regulations. This compliance reduces the environmental footprint of the manufacturing process, lowering the costs associated with waste treatment and permitting, while enhancing the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of this method for producing diverse triazole derivatives.
Q: Why is this new synthesis method safer than traditional triazole production?
A: Traditional methods often rely on organic azides, which are toxic and potentially explosive. This patented route utilizes stable diazo compounds and imidoyl chlorides, eliminating the safety hazards associated with handling high-energy azide intermediates.
Q: Does this process require expensive transition metal catalysts?
A: No, the process is metal-free. It uses cesium carbonate as a base promoter, which removes the need for costly copper catalysts and the subsequent complex purification steps required to remove trace heavy metals from pharmaceutical intermediates.
Q: What is the substrate scope for R1 and R2 groups in this reaction?
A: The method demonstrates broad functional group tolerance. R1 can be alkyl or various substituted aryl groups (e.g., methyl, methoxy, halogen), while R2 accommodates aroyl, phospholipid, alkoxycarbonyl, or trifluoromethyl groups, allowing for diverse molecular scaffolding.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
As the demand for fluorinated heterocycles continues to surge in the development of novel therapeutics and advanced materials, having a manufacturing partner with deep technical expertise is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging cutting-edge synthetic methodologies like the one described in CN113121462B to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 5-trifluoromethyl-1,2,3-triazole meets the highest international standards for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization.
