Advanced Acid-Catalyzed Synthesis of Quinoxaline-2-one Derivatives for Pharmaceutical Manufacturing
Introduction to Patent CN110981820B
The landscape of heterocyclic chemistry is constantly evolving, driven by the relentless demand for higher purity and greater efficiency in the production of pharmaceutical intermediates. A significant breakthrough in this domain is documented in patent CN110981820B, which discloses a novel method for synthesizing quinoxaline-2-one derivatives under acidic conditions. This technology addresses a longstanding challenge in organic synthesis: the control of regioselectivity during the condensation of asymmetric substrates. Quinoxaline scaffolds are ubiquitous in medicinal chemistry, serving as critical cores for compounds exhibiting antibacterial, antitumor, antiviral, and anti-inflammatory activities. However, traditional synthetic routes often suffer from poor selectivity, generating complex mixtures of regioisomers that are difficult and costly to separate. The disclosed method utilizes a simple yet effective organic acid catalysis system to drive the reaction towards the desired 3-substituted quinoxaline-2-one with exceptional precision.
By leveraging the intrinsic electronic properties of the substrates in the presence of an organic acid promoter, this process eliminates the need for harsh reaction conditions or exotic reagents. The result is a streamlined synthetic pathway that not only improves the chemical yield but also drastically enhances the purity profile of the final product. For R&D directors and process chemists, this represents a paradigm shift from brute-force synthesis to intelligent, mechanism-driven design. The ability to achieve such high selectivity without auxiliary reagents suggests a robust platform technology that can be adapted for the commercial scale-up of complex pharmaceutical intermediates, ensuring a reliable supply chain for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoxaline derivatives has relied on several established strategies, each carrying inherent drawbacks that complicate industrial application. One common approach involves microwave-assisted reactions between aromatic diamines and dicarbonyl compounds. While this method can achieve high conversion rates, it notoriously fails to control regioselectivity when asymmetric substrates are employed. As illustrated in the comparative data, such microwave protocols often result in a regioisomer ratio of approximately 1:1, creating a nightmare for purification teams who must separate structurally similar impurities.
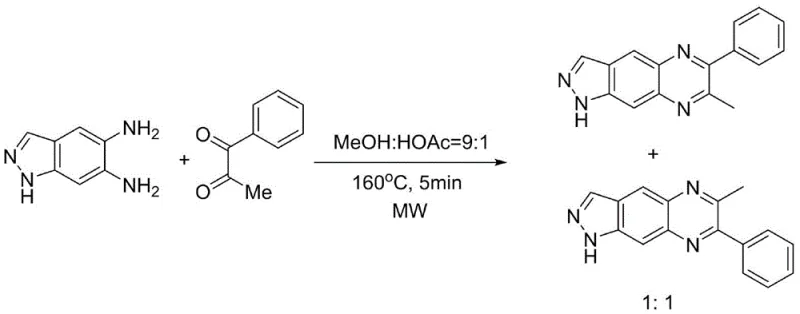
Another prevalent technique involves in-situ iodination and oxidation using copper oxide and iodine in DMSO. This multi-step cascade is not only operationally complex but also introduces heavy metal contaminants that require rigorous removal to meet pharmaceutical standards. Furthermore, alternative methods utilizing aniline-mediated alpha-imine esters often require prolonged reaction times exceeding 24 hours and necessitate the use of additional auxiliary reagents, inflating both the material costs and the environmental footprint of the process. These conventional limitations underscore the urgent need for a more direct, selective, and environmentally benign synthetic strategy.
The Novel Approach
The innovation presented in patent CN110981820B offers a compelling solution by employing a direct condensation reaction promoted solely by organic acids. This novel approach reacts 5-substituted o-phenylenediamines with ortho-dicarbonyl compounds, specifically alpha-ketoesters, in a mild alcohol solvent system. The key differentiator is the remarkable regioselectivity achieved; the reaction preferentially generates the 3-substituted quinoxaline-2-one (product 3) over the isomeric quinoxaline-3-one (product 4). Experimental data indicates that the ratio of the target product to the regioisomer can reach as high as 19:1 under optimized conditions.
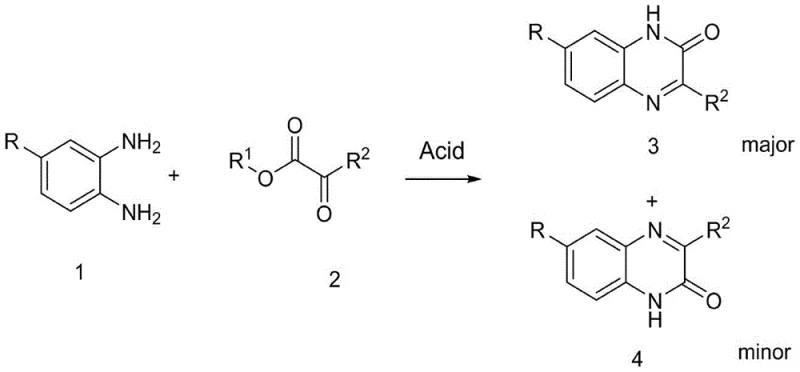
This high selectivity is achieved without the introduction of any auxiliary reagents or transition metal catalysts, relying instead on the subtle interplay between the organic acid catalyst and the electronic nature of the reactants. The operational simplicity is another major advantage; the reaction proceeds efficiently at room temperature, eliminating the energy costs associated with high-temperature reflux or microwave irradiation. For procurement managers, this translates to a significant reduction in raw material complexity and energy consumption, while for supply chain heads, the mild conditions imply a safer and more easily scalable process suitable for large-volume manufacturing.
Mechanistic Insights into Acid-Catalyzed Regioselective Cyclization
The success of this synthetic method lies in the precise mechanistic control exerted by the organic acid catalyst. In the absence of strong Lewis acids or oxidizing agents, the reaction proceeds through a nucleophilic attack of the amine groups on the carbonyl carbons of the alpha-ketoester. The presence of the acid likely protonates the more reactive ketone carbonyl oxygen, enhancing its electrophilicity and directing the initial nucleophilic attack by the more nucleophilic amine nitrogen of the o-phenylenediamine. This kinetic control is crucial for establishing the correct regiochemistry early in the reaction pathway.
Furthermore, the electronic effects of the substituents on the phenylenediamine ring play a pivotal role in stabilizing the transition state leading to the major isomer. Electron-donating groups, such as methoxy or ethoxy, enhance the nucleophilicity of the adjacent amine, further biasing the cyclization towards the formation of the quinoxaline-2-one scaffold. The acid medium also facilitates the dehydration steps required to aromatize the dihydro-intermediate, driving the equilibrium towards the final stable heterocyclic product. This mechanistic understanding allows chemists to predict and optimize outcomes for a wide range of substrates, ensuring consistent quality across different batches.
From an impurity control perspective, the high intrinsic selectivity means that the formation of the undesired regioisomer is kinetically suppressed rather than just thermodynamically disfavored. This minimizes the burden on downstream purification processes. Instead of requiring preparative HPLC or complex chromatographic separations, the crude product mixture often contains the target isomer in such high predominance that simple recrystallization is sufficient to achieve pharmaceutical-grade purity. This reduction in processing steps is a critical factor in maintaining cost efficiency and throughput in a commercial setting.
How to Synthesize Quinoxaline-2-one Efficiently
Implementing this acid-catalyzed protocol requires careful attention to stoichiometry and solvent choice to maximize the regioselectivity advantage. The standard procedure involves dissolving the substituted o-phenylenediamine in a lower alcohol solvent, such as methanol or ethanol, under an inert atmosphere. The addition of the organic acid, preferably glacial acetic acid, is a critical step that activates the system prior to the introduction of the electrophile. Following this activation, the alpha-ketoester is added slowly to manage the exotherm and ensure homogeneous mixing. The detailed standardized synthesis steps for this high-efficiency route are provided in the guide below.
- Dissolve 5-substituted o-phenylenediamine in an alcohol solvent (methanol or ethanol) under nitrogen protection.
- Add organic acid (preferably acetic acid, 5.0 equivalents) and stir, followed by the slow addition of the ortho-dicarbonyl compound (1.2 equivalents).
- Stir at room temperature until completion (0.5-2.0h), then pour into cold water, extract, and purify the major isomer via recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the shift to this acid-catalyzed methodology offers profound strategic benefits. The elimination of expensive and potentially hazardous reagents like iodine and copper oxide directly impacts the cost of goods sold (COGS). By replacing these with commodity chemicals like acetic acid, the raw material bill is significantly reduced, and the supply chain becomes less vulnerable to fluctuations in the price of specialty catalysts. Moreover, the avoidance of heavy metals simplifies the regulatory compliance landscape, reducing the testing burden and the risk of batch rejection due to residual metal limits.
- Cost Reduction in Manufacturing: The economic advantages of this process are multifaceted, stemming primarily from the simplification of the reaction workflow. By operating at room temperature, the process eliminates the substantial energy costs associated with heating large reactors to reflux or maintaining high-pressure microwave systems. Additionally, the high regioselectivity means that yield losses during purification are minimized; there is no need to discard large fractions of product to remove stubborn isomers. The use of simple recrystallization instead of column chromatography for purification further drives down solvent consumption and labor costs, resulting in a leaner, more cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes directly to supply chain stability. The raw materials required—substituted phenylenediamines and alpha-ketoesters—are widely available commodity chemicals with established global supply networks. Unlike processes dependent on proprietary ligands or scarce transition metals, this method does not face single-source bottlenecks. The mild reaction conditions also reduce the wear and tear on equipment and lower the safety risks associated with high-temperature operations, ensuring consistent production uptime and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with green chemistry principles. The absence of heavy metal catalysts means there is no toxic metal waste to treat or dispose of, significantly lowering the environmental footprint of the manufacturing site. The solvent systems used are primarily alcohols, which are easier to recover and recycle compared to chlorinated solvents or polar aprotic solvents like DMSO. This ease of waste management facilitates smoother regulatory approvals and supports the company's sustainability goals, making the commercial scale-up of complex pharmaceutical intermediates both feasible and responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this acid-catalyzed synthesis technology. These insights are derived directly from the experimental data and advantageous effects reported in the patent literature, providing a clear picture of what partners can expect when adopting this route for their own manufacturing needs.
Q: What is the regioselectivity advantage of this acid-catalyzed method?
A: Unlike traditional microwave or iodine-mediated methods which often yield a 1:1 mixture of isomers, this acid-catalyzed process achieves a regioisomer ratio of up to 19:1, significantly simplifying downstream purification.
Q: Does this process require expensive transition metal catalysts?
A: No, the method relies solely on inexpensive organic acids like acetic acid as the promoter, eliminating the need for costly transition metals like copper or iodine and avoiding heavy metal residue issues.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions (room temperature) with readily available raw materials and simple workup procedures (recrystallization), making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxaline-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and proven scalability. Our team has extensively analyzed the potential of the acid-catalyzed route described in CN110981820B and is fully prepared to leverage this technology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high selectivity and purity observed in the lab are maintained at the plant level. Our rigorous QC labs and stringent purity specifications guarantee that every batch of quinoxaline-2-one intermediate meets the exacting standards required for API synthesis.
We invite you to collaborate with us to optimize this process for your specific portfolio. Whether you require a Customized Cost-Saving Analysis to evaluate the economic impact of switching to this greener route or need specific COA data to validate the impurity profile, our technical procurement team is ready to assist. Contact us today to request route feasibility assessments and discover how our advanced synthesis capabilities can accelerate your drug development timeline while reducing overall manufacturing costs.
