Revolutionizing Azoxystrobin Production: A Deep Dive into Lewis Acid Catalyzed Formylation for Commercial Scale-up
Revolutionizing Azoxystrobin Production: A Deep Dive into Lewis Acid Catalyzed Formylation for Commercial Scale-up
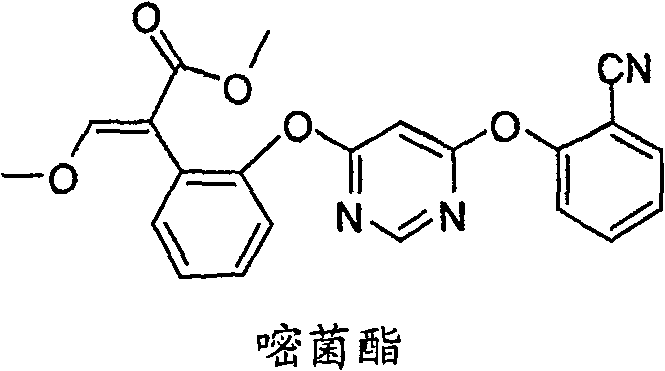
The global demand for high-efficiency fungicides continues to surge, driven by the critical need to secure crop yields against devastating fungal pathogens. At the forefront of this agricultural revolution is Azoxystrobin, a broad-spectrum strobilurin fungicide renowned for its ability to inhibit mitochondrial respiration in fungi. However, the complexity of its chemical structure has historically posed significant challenges for manufacturers seeking cost-effective and scalable production routes. A pivotal breakthrough in this domain is detailed in patent CN100564362C, which discloses a novel preparation method that fundamentally alters the synthetic landscape for Azoxystrobin and its analogues. This technology leverages a sophisticated Lewis acid catalyzed formylation strategy, bypassing the cumbersome multi-step sequences associated with legacy processes. By streamlining the construction of the critical methoxyacrylate pharmacophore, this innovation offers a robust pathway for producing high-purity agrochemical intermediates. For R&D directors and procurement strategists alike, understanding the nuances of this patented methodology is essential for optimizing supply chains and reducing manufacturing overheads in the competitive agrochemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Azoxystrobin has been plagued by inefficiencies inherent in traditional organic synthesis strategies. As outlined in the background art of the patent, conventional routes typically rely on o-hydroxy phenylacetic acid as a starting material, necessitating a dehydration step to form benzofuranone intermediates. This approach, while chemically valid, introduces significant operational friction. The key bottleneck lies in the methoxy metheneization of the alpha-methyl group within the benzofuranone structure, a transformation that often suffers from moderate yields and requires harsh reaction conditions. Furthermore, alternative routes involving the protection of phenolic hydroxyl groups using benzyl moieties add at least two additional reaction steps—protection and subsequent deprotection—to the overall sequence. These extra steps not only consume valuable raw materials and solvents but also generate increased waste streams, complicating environmental compliance and driving up the cost of goods sold. For a procurement manager, these inefficiencies translate directly into higher purchase prices and longer lead times, as the cumulative yield losses across multiple steps erode the overall process economics.
The Novel Approach
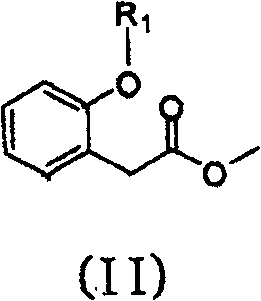
In stark contrast to these legacy methods, the technology disclosed in patent CN100564362C introduces a paradigm shift by utilizing a direct formylation-methylation sequence on phenylacetate derivatives. This novel approach eliminates the need for benzofuranone formation and the associated protection-deprotection cycles entirely. Instead, it employs a general formula (II) compound, which serves as a versatile precursor, and subjects it to a Lewis acid catalyzed formylation reaction. This transformation installs the necessary carbon framework for the methoxyacrylate side chain in a single, highly efficient operation. The subsequent methylation step finalizes the structure, delivering the target general formula (I) compound with remarkable precision. By collapsing what was previously a multi-step sequence into a concise two-stage process, this method drastically simplifies the production technique. The result is a synthetic route that is not only chemically elegant but also industrially superior, offering improved yields and a significantly reduced environmental footprint compared to the state-of-the-art methods previously available.
Mechanistic Insights into Titanium Tetrachloride Catalyzed Formylation
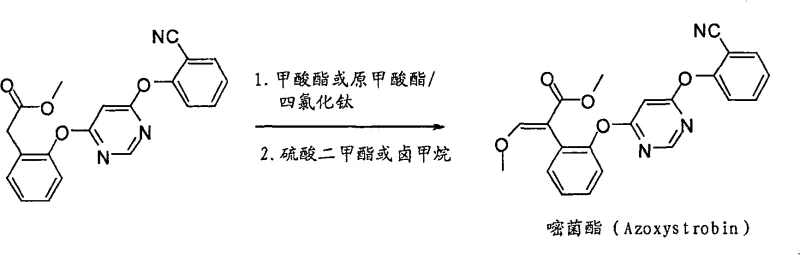
To fully appreciate the technical superiority of this process, one must delve into the mechanistic underpinnings of the Lewis acid catalyzed formylation. The core of this transformation relies on the activation of the formylation reagent, such as trimethyl orthoformate or methyl formate, by a potent Lewis acid. In the preferred embodiments, titanium tetrachloride (TiCl4) is utilized due to its strong electrophilic character. When introduced into an aprotic solvent system, typically a halogenated alkane like methylene dichloride, the titanium center coordinates with the oxygen atoms of the formylation reagent. This coordination significantly enhances the electrophilicity of the formyl carbon, making it susceptible to nucleophilic attack by the enolizable position of the phenylacetate substrate. The reaction is carefully controlled at temperatures ranging from -20°C to 50°C to manage the exothermic nature of the Lewis acid complexation and ensure regioselectivity. Following the initial formylation, the addition of a tertiary amine, such as triethylamine, serves a dual purpose: it neutralizes the acidic byproducts generated during the reaction and facilitates the elimination steps required to establish the conjugated double bond system essential for the biological activity of the final fungicide.
The subsequent methylation step is equally critical for defining the purity profile of the final product. Conducted at temperatures between -10°C and 100°C, this phase involves the reaction of the formylated intermediate with a methylating reagent like dimethyl sulfate in the presence of a mineral alkali. The choice of base and the precise control of stoichiometry are paramount here to prevent over-alkylation or hydrolysis of the sensitive ester functionalities. The patent data indicates that this sequence can achieve yields as high as 92% for the final Azoxystrobin product, a figure that underscores the efficiency of the catalytic cycle. From an impurity control perspective, the directness of this route minimizes the formation of side products typically associated with protection group chemistry, such as benzyl ethers or incomplete deprotection residues. This inherent cleanliness of the reaction pathway simplifies downstream purification, allowing manufacturers to meet stringent purity specifications required for regulatory approval in major agricultural markets without resorting to costly and yield-damaging recrystallization protocols.
How to Synthesize Azoxystrobin Efficiently
Implementing this advanced synthesis route requires a precise adherence to the reaction parameters defined in the patent to ensure reproducibility and safety. The process begins with the preparation of the key intermediate, 2-(2-(6-(2-cyano-benzene oxygen) pyrimidine-4-oxygen base) phenyl) methyl acetate, which acts as the substrate for the formylation. Operators must maintain strict anhydrous conditions, typically under nitrogen protection, to prevent the premature hydrolysis of the Lewis acid catalyst. The formylation is initiated by adding the Lewis acid to the solvent and formylation reagent mixture before introducing the substrate, ensuring the active catalytic species is generated in situ. Following the formylation and workup, the crude intermediate is immediately subjected to the methylation conditions. The detailed standardized synthesis steps, including specific molar ratios, stirring times, and quenching procedures, are critical for maximizing the 92% yield reported in the exemplary embodiments. For a comprehensive guide on executing this protocol in a pilot or production setting, please refer to the structured instructions below.
- Conduct a formylation reaction on the general formula (II) compound using a formylation reagent and a Lewis acid catalyst (such as titanium tetrachloride) in an aprotic solvent at temperatures between -20°C and 100°C, followed by the addition of a tertiary amine.
- Perform a methylation reaction on the resulting product from step 1 using a mineral alkali and a methylating reagent at temperatures between -10°C and 100°C to obtain the final general formula (I) compound.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and operational continuity, the adoption of this patented synthesis method offers compelling strategic advantages. The primary value proposition lies in the drastic simplification of the manufacturing workflow. By eliminating the need for protecting group chemistry, the process removes entire unit operations from the production schedule. This reduction in step count translates directly into lower capital expenditure on reactor time and a significant decrease in the consumption of solvents and auxiliary reagents. Furthermore, the avoidance of benzyl protection groups removes the necessity for hydrogenation or harsh acidic deprotection steps, which are often bottlenecks in batch processing. Consequently, the overall throughput of the manufacturing facility can be substantially increased without the need for new infrastructure investment. This efficiency gain allows suppliers to offer more competitive pricing structures while maintaining healthy margins, a critical factor for procurement managers negotiating long-term contracts for agrochemical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, driven primarily by the elimination of expensive reagents and the reduction of waste disposal costs. Traditional routes requiring benzyl protection involve the use of benzyl halides and subsequent removal steps that generate stoichiometric amounts of salt waste. In contrast, the Lewis acid catalyzed route utilizes commodity chemicals like titanium tetrachloride and dimethyl sulfate, which are readily available at scale. The higher yields observed in the patent examples, reaching up to 92%, mean that less raw material is required to produce the same amount of finished product. This atom economy, combined with the reduced solvent usage from fewer isolation steps, results in a significantly lower cost of goods sold. Additionally, the simplified process reduces the burden on quality control laboratories, as there are fewer intermediates to test and fewer potential impurities to monitor, further driving down operational expenses.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the robustness of the underlying chemistry. Complex multi-step syntheses with low-yielding protection steps are inherently fragile; a failure in any single step can halt the entire production campaign. The streamlined nature of this novel method mitigates such risks by reducing the number of critical control points. Moreover, the reagents employed are standard industrial chemicals with stable global supply chains, reducing the risk of raw material shortages. The ability to operate effectively within a broad temperature range (-20°C to 100°C) also provides operational flexibility, allowing manufacturers to adapt to varying utility conditions without compromising product quality. This reliability ensures consistent delivery schedules for downstream formulators, safeguarding their own production plans against upstream disruptions.
- Scalability and Environmental Compliance: As regulatory pressure on the agrochemical industry intensifies, the environmental profile of a synthesis route becomes a key differentiator. This method excels in sustainability metrics by minimizing waste generation. The absence of protection-deprotection cycles significantly reduces the E-factor (mass of waste per mass of product). Furthermore, the use of aprotic solvents like methylene dichloride, while requiring careful handling, allows for efficient recovery and recycling through distillation, aligning with green chemistry principles. The process is inherently scalable, as demonstrated by the successful execution of the reaction in standard laboratory glassware which translates well to stirred-tank reactors. The straightforward workup procedures, involving simple aqueous washes and solvent removal, facilitate easy scale-up from kilogram to tonne quantities, ensuring that the technology can meet the demands of global commercial production without encountering unforeseen engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Azoxystrobin synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN100564362C, providing a factual basis for decision-making. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios. The answers highlight the specific advantages in terms of yield, purity, and operational simplicity that distinguish this method from prior art.
Q: How does the new Lewis acid catalyzed method improve upon traditional Azoxystrobin synthesis routes?
A: Traditional methods often require complex protection and deprotection steps for phenolic hydroxyl groups or the synthesis of benzofuranone intermediates. The novel method described in patent CN100564362C utilizes a direct Lewis acid catalyzed formylation, which significantly simplifies the production technique, reduces the number of reaction steps, and improves overall yield by avoiding unnecessary derivatization.
Q: What are the critical reaction conditions for the formylation step in this process?
A: The formylation reaction is optimally conducted in an aprotic solvent, preferably a halogenated alkane like methylene dichloride, at temperatures ranging from -20°C to 50°C. The process employs a Lewis acid such as titanium tetrachloride (1.0-3.0 molar equivalents) and a formylation reagent like trimethyl orthoformate, followed by the addition of a tertiary amine base to drive the reaction to completion.
Q: Is this synthesis method scalable for industrial production of agrochemical intermediates?
A: Yes, the method is highly suitable for industrial scale-up. It utilizes commercially available reagents such as titanium tetrachloride and methyl sulfate, operates under manageable temperature conditions (-20°C to 100°C), and eliminates the need for expensive or hazardous protection group chemistry, thereby enhancing process safety and economic viability for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azoxystrobin Supplier
The technological advancements detailed in patent CN100564362C represent a significant leap forward in the manufacture of high-value agrochemical intermediates. However, translating patented chemistry into commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that ensure every batch of Azoxystrobin meets the highest international standards. We understand that for R&D directors and supply chain heads, consistency is key, and our state-of-the-art facilities are designed to deliver exactly that, leveraging advanced catalytic technologies to optimize efficiency and reduce costs for our global clientele.
We invite you to explore how our optimized synthesis routes can enhance your supply chain resilience and drive cost efficiencies in your fungicide portfolio. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable Azoxystrobin supplier dedicated to fostering long-term growth and innovation in the agrochemical sector.
