Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly quinazolinones, which serve as privileged scaffolds in drug discovery. Patent CN112480015B introduces a groundbreaking multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones, addressing critical bottlenecks in traditional synthetic routes. This technology leverages a palladium-catalyzed carbonylation cascade that transforms inexpensive nitro compounds and trifluoroethylimidoyl chlorides into high-value intermediates under relatively mild conditions. For R&D directors and procurement managers, this represents a significant opportunity to streamline the supply chain for antifungal, antiviral, and anticancer agents, as the trifluoromethyl group is known to enhance metabolic stability and lipophilicity in bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied on methodologies that impose severe constraints on industrial scalability and operational safety. Traditional routes often necessitate the use of high-pressure carbon monoxide gas, requiring specialized autoclaves and rigorous safety protocols that drastically increase capital expenditure and operational complexity. Furthermore, many existing catalytic systems depend on expensive ruthenium or platinum catalysts, or require pre-activated substrates such as 2-bromoformylaniline, which adds multiple synthetic steps and generates substantial chemical waste. These conventional approaches frequently suffer from narrow substrate tolerance, meaning that introducing diverse functional groups often leads to poor yields or complete reaction failure, limiting the chemical space available for medicinal chemists to explore during lead optimization phases.
The Novel Approach
In stark contrast, the methodology disclosed in CN112480015B utilizes a transition metal palladium catalytic system that operates efficiently in a one-pot fashion, dramatically simplifying the process workflow. By employing Molybdenum Hexacarbonyl (Mo(CO)6) as a solid carbon monoxide substitute, the reaction avoids the hazards associated with handling toxic CO gas, thereby reducing the engineering controls required for safe manufacturing. The use of cheap and readily available nitro compounds as starting materials, coupled with a robust PdCl2/dppp catalyst system, ensures high reaction efficiency and broad functional group compatibility. This approach not only lowers the raw material costs but also minimizes the environmental footprint by reducing the number of isolation and purification steps, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The elegance of this synthesis lies in its intricate yet efficient catalytic cycle, which seamlessly integrates reduction, coupling, and cyclization events. The reaction initiates with the reduction of the nitro compound to an amine species by Mo(CO)6, which simultaneously serves as the carbonyl source. This in-situ generated amine then undergoes a base-promoted intermolecular carbon-nitrogen bond coupling with the trifluoroethylimidoyl chloride to form a trifluoroacetamidine derivative. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the imidoyl chloride moiety, forming a divalent palladium intermediate. As the temperature is maintained at 120°C, Mo(CO)6 releases carbon monoxide, which inserts into the carbon-palladium bond to generate an acyl-palladium species.
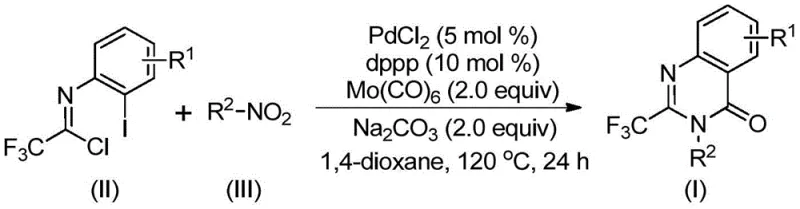
Following the CO insertion, the base facilitates the formation of a palladium-nitrogen bond, closing the ring to create a seven-membered cyclic palladium intermediate. The final step involves a reductive elimination that releases the desired 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium catalyst. This mechanistic pathway is highly advantageous for impurity control, as the one-pot nature minimizes the exposure of reactive intermediates to external contaminants. For quality assurance teams, understanding this mechanism confirms that the process is self-contained and less prone to side reactions compared to multi-step sequences, ensuring a cleaner impurity profile and higher overall purity for the final high-purity pharmaceutical intermediate.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and minimize byproduct formation. The protocol dictates the use of an aprotic organic solvent, with 1,4-dioxane being the preferred medium due to its ability to dissolve all reagents effectively and promote high conversion rates. The reaction is typically conducted at 120°C for a duration of 16 to 30 hours, a timeframe that balances reaction completeness with operational efficiency. Detailed standard operating procedures regarding reagent stoichiometry, specifically the molar ratios of palladium chloride, ligand, and base, are critical for reproducibility. For a comprehensive guide on executing this transformation in your facility, please refer to the standardized synthesis steps outlined below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 120°C and maintain stirring for 16 to 30 hours to allow the carbonylation cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented technology offers compelling advantages that directly impact the bottom line and supply chain resilience. The shift from high-pressure gas reactions to a solution-phase process using solid CO surrogates significantly lowers the barrier to entry for contract manufacturing organizations (CMOs) who may lack specialized high-pressure infrastructure. This accessibility translates to a broader supplier base and reduced risk of supply disruption. Moreover, the utilization of commodity chemicals like nitro compounds and simple imidoyl chlorides ensures that raw material costs remain stable and predictable, shielding the project from the volatility often seen with exotic or custom-synthesized building blocks.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the use of earth-abundant palladium catalysts instead of precious metals like ruthenium or platinum lead to substantial capital and operational expenditure savings. The one-pot design reduces solvent consumption and labor hours associated with intermediate isolations, driving down the overall cost of goods sold (COGS) without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Since the starting materials are widely available commercially and the reaction conditions are robust, the risk of batch failure is minimized. This reliability ensures consistent delivery schedules, which is crucial for maintaining continuous API production lines. The ability to source generic nitro compounds from multiple vendors further mitigates the risk of single-source dependency.
- Scalability and Environmental Compliance: The process has been demonstrated to be scalable from milligram to gram levels with consistent efficiency, indicating strong potential for ton-scale production. The use of Mo(CO)6 as a CO source is inherently safer and generates less hazardous waste compared to gaseous CO streams, simplifying waste treatment protocols and ensuring compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope and operational requirements. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development discussions.
Q: What is the primary advantage of using Mo(CO)6 over gaseous CO in this synthesis?
A: Using Molybdenum Hexacarbonyl (Mo(CO)6) as a solid carbon monoxide substitute eliminates the need for high-pressure gas equipment, significantly enhancing operational safety and simplifying the reaction setup for industrial scale-up.
Q: What is the substrate scope for the nitro compound in this reaction?
A: The method demonstrates excellent compatibility with various substituents on the nitro compound, including alkyl groups (methyl, n-propyl), halogens (F, Cl, Br), and electron-withdrawing groups like trifluoromethyl, allowing for diverse derivative synthesis.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the patent explicitly states that the method can be expanded to the gram level and beyond, utilizing cheap and readily available starting materials which supports cost-effective commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology for the next generation of therapeutic agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle complex organometallic chemistry safely, and our rigorous QC labs enforce stringent purity specifications to meet the exacting standards of global regulatory bodies.
We invite you to collaborate with us to leverage this innovative synthesis route for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can become your trusted partner in delivering high-quality quinazolinone intermediates efficiently and reliably.
