Advanced Synthesis of Premium (1R,2S)-Methyl Dihydrojasmonate for Global Flavor Industries
Advanced Synthesis of Premium (1R,2S)-Methyl Dihydrojasmonate for Global Flavor Industries
The global demand for high-quality fragrance ingredients continues to drive innovation in synthetic organic chemistry, particularly for molecules that define the olfactory profile of luxury cosmetics and fine perfumery. Patent CN113929575B introduces a groundbreaking preparation method for (1R, 2S)-methyl dihydrojasmonate, a compound widely recognized in the industry by the trade name Hedione. This specific stereoisomer is prized for its intensely floral, jasmine-like scent which is significantly more potent and diffusive than its counterparts. The patent details a sophisticated three-step catalytic sequence that overcomes historical limitations in stereoselectivity and reaction severity. By leveraging a combination of acid-catalyzed enolization, copper-mediated dehydrogenation, and finally, rhodium-catalyzed asymmetric hydrogenation, this technology offers a robust pathway to achieving high optical purity. For R&D directors and procurement specialists, understanding the nuances of this synthetic route is essential for securing a reliable supply chain of this high-value flavor and fragrance intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
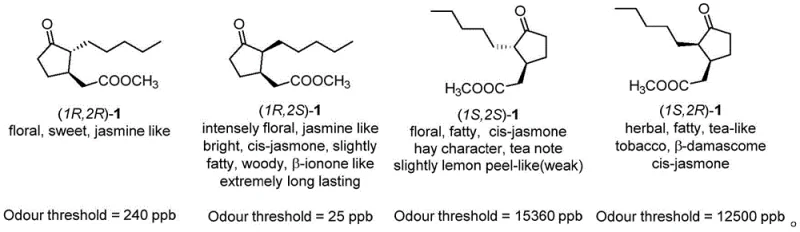
Historically, the industrial production of methyl dihydrojasmonate has faced significant challenges regarding stereochemical control and process efficiency. Traditional synthetic routes often rely on the resolution of racemic mixtures or non-selective hydrogenation processes that yield a complex mixture of all four possible optical isomers. As illustrated in the structural analysis, the trans-(1R,2R) isomer possesses an undesirable earthy note, while the (1S,2S) and (1S,2R) enantiomers exhibit very weak fragrance intensity. Conventional methods frequently necessitate harsh reaction conditions, such as high temperatures and pressures, which not only increase energy consumption but also pose safety risks in a manufacturing environment. Furthermore, many prior art methods utilize catalysts that are either prohibitively expensive, difficult to source in bulk quantities, or challenging to remove from the final product, leading to increased purification costs and potential contamination issues that compromise the quality of the final fragrance oil.
The Novel Approach
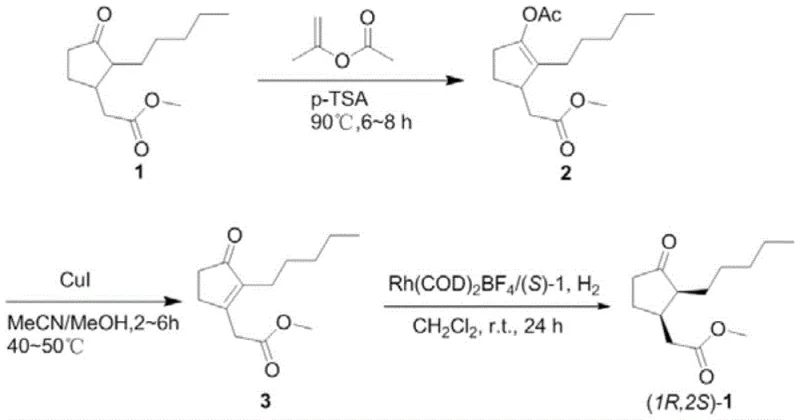
The methodology disclosed in patent CN113929575B represents a paradigm shift towards greener and more efficient manufacturing. The core innovation lies in a streamlined three-step sequence that begins with the conversion of methyl dihydrojasmonate into an enol acetate intermediate using isopropenyl acetate and p-toluenesulfonic acid. This is followed by a mild dehydrogenation step using copper iodide to generate a key alpha,beta-unsaturated ketone precursor. The culmination of the process is an asymmetric catalytic hydrogenation that selectively produces the desired (1R,2S) cis-isomer. This approach eliminates the need for extreme conditions, operating instead at moderate temperatures ranging from 40°C to 95°C across the different stages. The ability to reuse solvents and catalysts after separation further aligns this process with modern sustainability goals, offering a distinct competitive advantage over older, waste-intensive technologies.
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
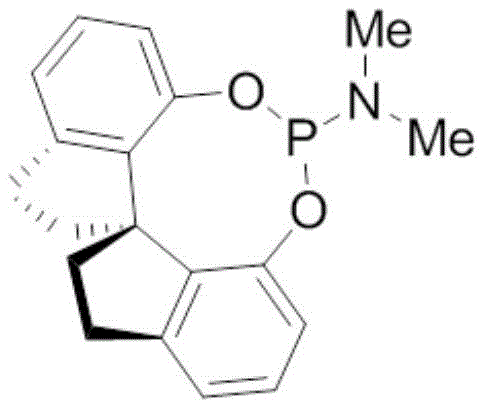
The heart of this synthetic strategy is the final asymmetric hydrogenation step, which dictates the optical purity and olfactory quality of the final product. This transformation utilizes a specialized rhodium catalyst system, specifically Rh(COD)2BF4 complexed with a chiral spirocyclic aminophosphine ligand, designated as (S)-1. The unique three-dimensional architecture of this ligand creates a highly specific chiral environment around the rhodium center. When the prochiral alpha,beta-unsaturated ketone substrate coordinates to the metal, the bulky groups on the ligand sterically direct the approach of molecular hydrogen. This ensures that hydrogen addition occurs predominantly from one face of the double bond, thereby establishing the two chiral centers with high fidelity. Experimental data from the patent indicates that this system can achieve enantiomeric excess (ee) values up to 83%, demonstrating the efficacy of the ligand design in controlling stereochemistry without requiring cryogenic temperatures or ultra-high pressures.
Impurity control is another critical aspect managed by this mechanistic design. In traditional hydrogenation, over-reduction or isomerization of the double bond can lead to off-odors or reduced fragrance intensity. The specific electronic properties of the aminophosphine ligand modulate the electron density at the rhodium center, optimizing it for the selective reduction of the carbon-carbon double bond while leaving the ester and ketone functionalities intact. Moreover, the mild reaction conditions (room temperature, 0.5-1.0 MPa H2) minimize thermal degradation pathways that often generate polymeric byproducts or decomposition species. This results in a cleaner reaction profile, simplifying the downstream workup and reducing the burden on purification units, which is a key consideration for maintaining high throughput in a commercial setting.
How to Synthesize (1R,2S)-Methyl Dihydrojasmonate Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and selectivity. The process begins with the activation of the starting material through enolization, followed by oxidation to the unsaturated ketone, and concludes with the stereoselective reduction. Each step has been optimized to balance reaction rate with product stability, ensuring that intermediates do not degrade before the next transformation. The use of common organic solvents like ethyl acetate, acetonitrile, and dichloromethane facilitates easy handling and solvent recovery. For technical teams looking to adopt this route, attention to the catalyst loading and hydrogen pressure in the final step is paramount to achieving the target optical purity. Detailed standardized operating procedures for scaling this chemistry from gram to kilogram scale are outlined in the technical documentation below.
- Catalytic enolization of methyl dihydrojasmonate with isopropenyl acetate using p-TSA at 85-95°C to form the enol acetate intermediate.
- Copper iodide-mediated dehydrogenation of the enol acetate in acetonitrile/methanol at 40-50°C to generate the alpha,beta-unsaturated ketone.
- Asymmetric hydrogenation using Rh(COD)2BF4 with a chiral spirocyclic aminophosphine ligand under 0.5-1.0 MPa H2 pressure at room temperature.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere technical feasibility. The shift towards milder reaction conditions directly translates to reduced operational expenditures, as there is less demand for high-energy heating or cooling systems and specialized high-pressure equipment. The ability to recycle solvents and catalysts significantly lowers the raw material cost per kilogram of finished product, addressing the constant pressure to reduce costs in flavor and fragrance manufacturing. Furthermore, the reliance on commercially available reagents like copper iodide and p-toluenesulfonic acid mitigates supply risk, ensuring that production schedules are not disrupted by the scarcity of exotic chemicals. This robustness makes the supply chain more resilient against market volatility.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the implementation of catalyst recycling protocols lead to substantial cost savings. By avoiding the need for expensive cryogenic cooling or ultra-high pressure reactors, capital expenditure for new production lines is minimized. Additionally, the high selectivity of the asymmetric hydrogenation reduces the formation of unwanted isomers, meaning less material is wasted during purification. This efficiency gain allows for a more competitive pricing structure for the final high-purity ingredient, providing a clear margin advantage in the marketplace.
- Enhanced Supply Chain Reliability: The synthetic route utilizes commodity chemicals and standard solvents that are readily available from multiple global suppliers. This diversification of the raw material base reduces dependency on single-source vendors for critical reagents. The mild nature of the reactions also implies lower maintenance requirements for processing equipment, resulting in higher plant availability and fewer unplanned downtime events. Consequently, manufacturers can offer more reliable lead times and consistent delivery schedules to their downstream customers in the cosmetic and detergent industries.
- Scalability and Environmental Compliance: Designed with green chemistry principles, this process generates less hazardous waste compared to traditional methods. The ability to recover and reuse solvents like dichloromethane and ethyl acetate aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. The scalability of the three-step sequence has been demonstrated through varied batch sizes in the patent examples, indicating that the chemistry translates well from pilot plant to full commercial production without significant loss of efficiency or selectivity, ensuring a stable long-term supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of (1R,2S)-methyl dihydrojasmonate. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing portfolios. The answers reflect the specific advantages of the catalytic system and the operational parameters required for success.
Q: What distinguishes the (1R,2S) isomer of methyl dihydrojasmonate from other stereoisomers?
A: The (1R,2S) cis-isomer possesses the strongest and purest jasmine fragrance with long-lasting diffusion properties, whereas the trans-isomers often exhibit earthy notes and the other enantiomers have significantly weaker olfactory impact, making high stereochemical purity critical for premium applications.
Q: How does this new catalytic method improve upon traditional synthesis routes?
A: Unlike conventional methods that often require harsh reaction conditions or difficult-to-source catalysts, this patented route utilizes mild temperatures (40-95°C) and commercially available reagents like copper iodide and rhodium complexes, enabling a simpler three-step process with potential for catalyst recycling.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is designed with green chemistry principles in mind, utilizing standard solvents like ethyl acetate and dichloromethane which can be recovered, and operating at near-atmospheric pressures for the hydrogenation step, facilitating safer and more cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl Dihydrojasmonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of delivering high-purity fragrance intermediates that meet the exacting standards of the global perfume and flavor industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN113929575B can be successfully translated into robust manufacturing operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (1R,2S)-methyl dihydrojasmonate delivers the consistent, powerful jasmine aroma that your formulations require. Our commitment to quality assurance ensures that the complex stereochemistry of this molecule is preserved throughout the production lifecycle.
We invite you to collaborate with us to leverage this advanced synthetic technology for your product lines. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments that demonstrate how we can optimize your supply chain for this high-value ingredient. By partnering with us, you gain access to a reliable source of premium synthetic flavors backed by cutting-edge chemical engineering and a dedication to sustainable manufacturing practices.
