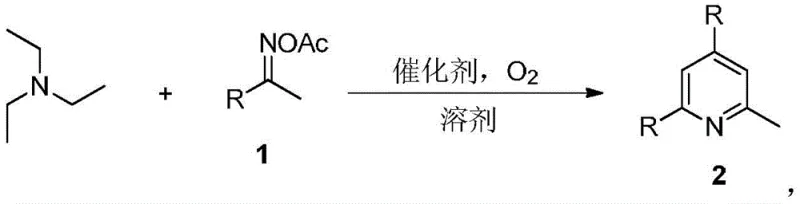
Scalable Synthesis of 2-Methylpyridine Compounds via Triethylamine Cyclization
The chemical landscape for synthesizing heterocyclic compounds is constantly evolving, driven by the need for greener, more efficient processes that align with modern sustainability goals. A significant breakthrough in this domain is detailed in Chinese Patent CN108314642B, which discloses a novel synthetic method for 2-methylpyridine compounds. This technology represents a paradigm shift from traditional high-energy processes to a streamlined, transition metal-free approach. The core innovation lies in the utilization of triethylamine and oxime acetate compounds as starting materials, reacting under an oxygen atmosphere with an iodine-based catalyst. This method not only simplifies the operational workflow but also addresses critical environmental concerns associated with heavy metal contamination and excessive energy consumption. For R&D directors and procurement specialists in the fine chemical sector, understanding this pathway is essential for optimizing supply chains for key intermediates used in sulfonamides, antihistamines like chlorpheniramine, and various agrochemicals.
2-Methylpyridine derivatives serve as pivotal building blocks in the pharmaceutical and agrochemical industries, yet their production has historically been fraught with inefficiencies. Conventional synthesis routes, such as the acetaldehyde method, acetylene method, ethylene method, and acrylonitrile method, typically demand harsh reaction environments characterized by high temperatures and high pressures. These legacy processes often suffer from low yields and a proliferation of side reactions, which necessitate complex purification steps and generate substantial waste. The economic burden of these inefficiencies is compounded by the safety risks associated with high-pressure reactors and the environmental cost of disposing of hazardous byproducts. Consequently, there is a pressing industrial demand for alternative synthetic strategies that can deliver high-purity intermediates with reduced operational complexity and lower overall production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The traditional manufacturing landscape for 2-methylpyridine compounds is dominated by petrochemical-derived pathways that are increasingly untenable in a regulatory environment focused on green chemistry. Methods relying on acetaldehyde or acrylonitrile often require rigorous control of exothermic reactions and specialized equipment capable of withstanding extreme pressures, which inherently limits scalability and increases capital expenditure. Furthermore, the selectivity of these reactions is often poor, leading to a mixture of pyridine isomers that are difficult and expensive to separate. The reliance on multi-step sequences in some variations further exacerbates the issue, as each additional step introduces potential yield losses and requires intermediate isolation and purification. This accumulation of inefficiencies results in a final product that carries a high cost basis and a significant environmental footprint, making it less attractive for cost-sensitive applications in generic drug manufacturing or large-volume agrochemical production.
The Novel Approach
In stark contrast, the methodology outlined in patent CN108314642B offers a sophisticated yet operationally simple solution through a one-pot tandem reaction. By employing triethylamine, a readily available and inexpensive commodity chemical, alongside oxime acetates, the process bypasses the need for complex precursor synthesis. The reaction proceeds smoothly in common organic solvents such as toluene, acetonitrile, or 1,4-dioxane, eliminating the need for exotic or hazardous reaction media. Crucially, the system operates under an oxygen atmosphere, utilizing molecular oxygen as the terminal oxidant, which is both atom-economical and environmentally benign. The absence of transition metal catalysts like palladium or platinum removes the risk of heavy metal residues in the final API intermediate, a critical quality attribute for pharmaceutical clients. This approach effectively consolidates multiple bond-forming events into a single operational step, drastically reducing the time and resources required to produce high-value 2-methylpyridine scaffolds.
Mechanistic Insights into Iodine-Catalyzed Oxidative Cyclization
The mechanistic underpinning of this transformation involves an iodine-catalyzed oxidative cyclization that leverages the nucleophilicity of triethylamine and the electrophilic nature of the oxime acetate. The reaction initiates with the activation of the oxime acetate by the iodine catalyst, generating a reactive nitrilium or iminium species that is susceptible to nucleophilic attack. Triethylamine, serving as both a reactant and a base, undergoes dealkylation and subsequent cyclization to construct the pyridine ring system. The presence of molecular oxygen is vital for the re-oxidation of the iodine species, sustaining the catalytic cycle and driving the reaction to completion without the need for stoichiometric oxidants that generate waste. This catalytic cycle is robust and tolerant of various functional groups, allowing for the synthesis of a diverse array of substituted 2-methylpyridines. The mild thermal conditions, typically ranging from 120°C to 160°C, ensure that sensitive functional groups on the aromatic rings remain intact, preserving the integrity of complex molecular architectures required for advanced drug candidates.
Impurity control is inherently superior in this one-pot design compared to multi-step linear syntheses. By avoiding the isolation of unstable intermediates, the process minimizes exposure to conditions that could lead to decomposition or polymerization. The use of iodine as a catalyst, rather than a stoichiometric reagent, ensures that residual metal content in the final product is negligible, simplifying the downstream purification process. Standard workup procedures involving aqueous quenching, extraction with ethyl acetate, and washing with sodium thiosulfate effectively remove iodine residues. Final purification via silica gel chromatography yields products with high purity profiles, as evidenced by the consistent NMR and HRMS data reported across numerous examples in the patent. This level of purity is essential for meeting the stringent specifications of reliable pharmaceutical intermediate suppliers and ensures that the material is suitable for direct use in subsequent coupling reactions or biological assays.
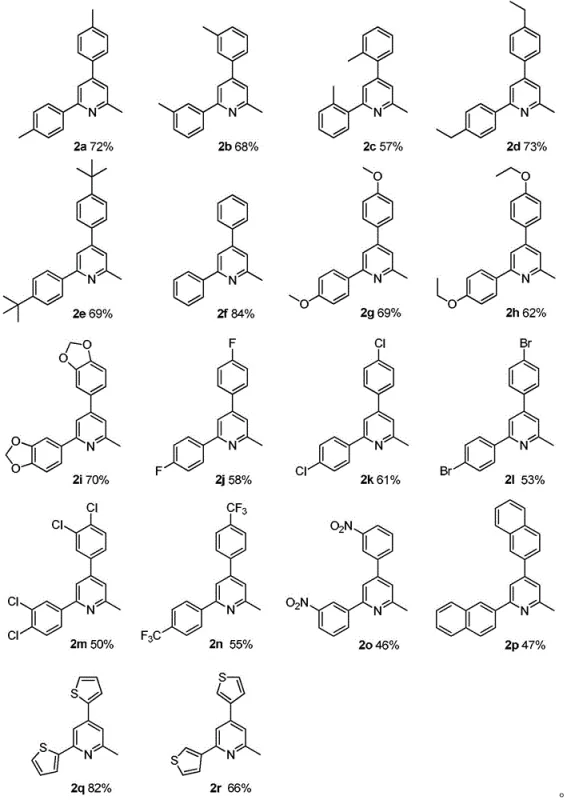
How to Synthesize 2-Methyl-4,6-diarylpyridines Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and reproducibility. The patent provides extensive experimental data demonstrating the robustness of the method across a wide range of substrates, including those with electron-donating and electron-withdrawing groups. The standard protocol involves dissolving the reactants in toluene, adding the catalyst, and heating under an oxygen balloon or flow. While the general procedure is straightforward, optimization of the catalyst loading and reaction temperature may be necessary for specific sterically hindered substrates. The detailed standardized synthesis steps for producing these high-purity intermediates are outlined below, providing a clear roadmap for technical teams looking to adopt this technology.
- Dissolve triethylamine and the specific oxime acetate compound in a solvent such as toluene, acetonitrile, or 1,4-dioxane within a reaction vessel.
- Add an iodine-based catalyst (Iodine, N-iodosuccinimide, or Ammonium Iodide) to the mixture and establish an oxygen atmosphere.
- Heat the reaction mixture to a temperature between 120°C and 160°C (optimally 140°C) for approximately 12 hours, followed by aqueous workup and silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The shift from high-pressure, multi-step processes to a mild, one-pot reaction translates into tangible operational efficiencies. By eliminating the need for expensive transition metal catalysts, the process removes a significant cost driver and mitigates the supply risk associated with precious metals. Furthermore, the simplified workup and purification sequence reduce the consumption of solvents and consumables, contributing to a leaner manufacturing operation. These factors combine to create a more resilient supply chain capable of delivering consistent quality at a competitive price point, which is crucial for maintaining margins in the highly competitive fine chemical market.
- Cost Reduction in Manufacturing: The utilization of triethylamine as a primary feedstock represents a strategic cost-saving measure, as it is a bulk chemical with a stable global supply and low price volatility compared to specialized organometallic reagents. The elimination of transition metals not only saves on the initial cost of the catalyst but also removes the downstream expense of metal scavenging and validation testing for heavy metal residues. Additionally, the one-pot nature of the reaction reduces labor costs and reactor occupancy time, allowing for higher throughput in existing facilities. These cumulative savings enable a substantial reduction in the cost of goods sold (COGS) for 2-methylpyridine derivatives, enhancing profitability for manufacturers and offering better pricing flexibility for buyers.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like triethylamine and common solvents like toluene ensures that raw material availability is not a bottleneck for production. Unlike processes dependent on scarce or geographically concentrated reagents, this method leverages a supply chain that is robust and diversified. The mild reaction conditions also reduce the risk of unplanned shutdowns due to equipment failure or safety incidents, ensuring consistent delivery schedules. For supply chain planners, this reliability is invaluable, as it minimizes the need for excessive safety stock and allows for more agile response to fluctuations in market demand. The ability to scale this process from gram to ton quantities without fundamental changes to the chemistry further secures the long-term continuity of supply.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the simplicity of this oxidative cyclization facilitates a smooth transition from bench to plant. The absence of high-pressure requirements lowers the barrier for equipment qualification and reduces the capital investment needed for scale-up. From an environmental standpoint, the use of oxygen as an oxidant and the generation of minimal waste align with increasingly strict global environmental regulations. This compliance reduces the administrative burden of waste disposal and permitting, accelerating the time to market for new products. Companies adopting this green chemistry approach can also leverage it for sustainability reporting, enhancing their corporate image and meeting the ESG criteria demanded by modern investors and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. They are derived from the specific experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this method for their own production needs. Understanding these details is crucial for making informed decisions about process adoption and supplier selection.
Q: What are the primary advantages of this triethylamine-based method over traditional acetaldehyde routes?
A: Unlike traditional methods requiring high temperature and pressure with low yields, this patent describes a mild, one-pot reaction at 120-160°C that avoids transition metals and simplifies purification, significantly reducing resource waste.
Q: Which catalysts are effective for this oxidative cyclization?
A: The patent specifies iodine element, N-iodosuccinimide (NIS), or ammonium iodide as effective catalysts, with elemental iodine often providing optimal yields in toluene solvent.
Q: Is this process suitable for large-scale pharmaceutical intermediate manufacturing?
A: Yes, the use of cheap raw materials like triethylamine, combined with mild reaction conditions and simple workup procedures involving standard extraction and chromatography, makes it highly suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methylpyridine Supplier
The technological potential of this iodine-catalyzed oxidative cyclization is immense, offering a pathway to high-quality intermediates that meet the rigorous demands of the global pharmaceutical industry. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust manufacturing processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We understand that consistency is key in API intermediate manufacturing, and our infrastructure is designed to deliver that reliability consistently.
We invite you to explore how this advanced synthesis method can optimize your supply chain and reduce costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. By partnering with us, you gain access to not just a product, but a comprehensive solution that enhances your competitive edge in the market through superior chemistry and supply chain excellence.
