Advanced Palladium-Catalyzed Bis-Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-Ketone Manufacturing
Introduction to Next-Generation Pyrrol-2-One Synthesis
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a privileged scaffold in medicinal chemistry, serving as the core backbone for numerous bioactive natural products and synthetic pharmaceuticals. As highlighted in recent intellectual property disclosures, specifically patent CN112694430B, innovative methodologies are emerging to construct this heterocyclic system with unprecedented efficiency. The significance of this chemical architecture is underscored by its presence in potent antibacterial agents like Althiomycin, effective hypoglycemic drugs such as Glimepiride, and anticancer candidates like Isomalyngamide A. 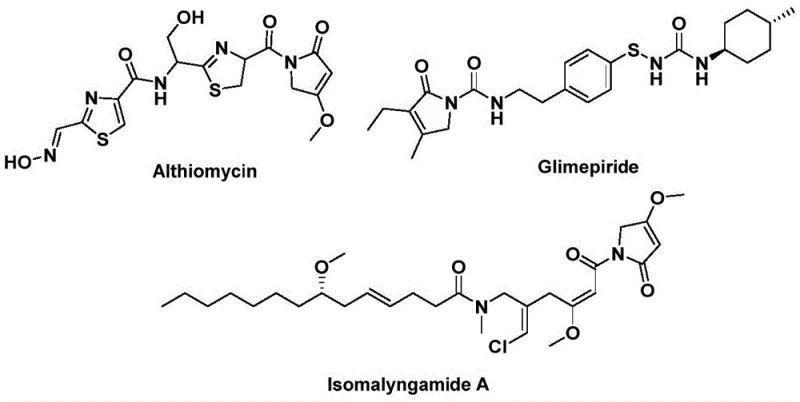 . For research and development directors seeking to optimize lead generation, access to a robust, scalable synthesis of this core is critical. The disclosed technology offers a transformative approach by utilizing a palladium-catalyzed bis-carbonylation strategy that bypasses traditional limitations, positioning it as a key enabling technology for reliable pharmaceutical intermediate supplier networks aiming to streamline API production pipelines.
. For research and development directors seeking to optimize lead generation, access to a robust, scalable synthesis of this core is critical. The disclosed technology offers a transformative approach by utilizing a palladium-catalyzed bis-carbonylation strategy that bypasses traditional limitations, positioning it as a key enabling technology for reliable pharmaceutical intermediate supplier networks aiming to streamline API production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one ring system has relied on multi-step sequences involving harsh reaction conditions or the use of unstable intermediates. Traditional carbonylation reactions often necessitate the direct use of carbon monoxide gas, which poses significant safety hazards due to its high toxicity and the requirement for specialized high-pressure equipment. Furthermore, conventional routes frequently suffer from poor atom economy and limited substrate scope, particularly when dealing with sensitive functional groups that may degrade under vigorous thermal or acidic conditions. These factors collectively contribute to increased operational costs and extended lead times, creating bottlenecks in cost reduction in API manufacturing. The reliance on gaseous CO also complicates the logistics of supply chain management, as it requires stringent safety protocols and infrastructure that many standard organic synthesis laboratories lack, thereby hindering the rapid iteration required in modern drug discovery programs.
The Novel Approach
In stark contrast, the methodology described in patent CN112694430B introduces a sophisticated one-pot synthesis that leverages a solid carbon monoxide surrogate, specifically 1,3,5-tricarboxylic acid phenol ester (TFBen). This innovation fundamentally alters the risk profile of the reaction by eliminating the need for pressurized gas cylinders, allowing the transformation to proceed safely in standard glassware at atmospheric pressure. The process utilizes readily available starting materials, namely propargyl amines and benzyl chlorides, which are coupled efficiently in the presence of a palladium catalyst and a bidentate phosphine ligand. 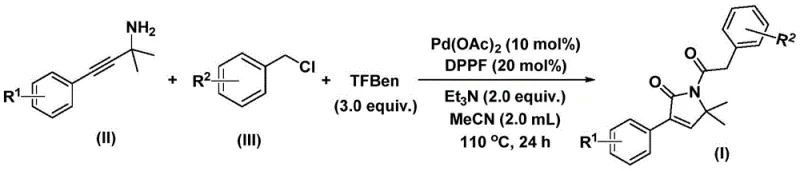 . This novel approach not only simplifies the operational workflow but also demonstrates exceptional functional group tolerance, accommodating a wide array of substituents without compromising yield. By consolidating what was previously a multi-step sequence into a single, high-efficiency transformation, this method significantly enhances the commercial scale-up of complex pharmaceutical intermediates, offering a streamlined pathway for industrial adoption.
. This novel approach not only simplifies the operational workflow but also demonstrates exceptional functional group tolerance, accommodating a wide array of substituents without compromising yield. By consolidating what was previously a multi-step sequence into a single, high-efficiency transformation, this method significantly enhances the commercial scale-up of complex pharmaceutical intermediates, offering a streamlined pathway for industrial adoption.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The catalytic cycle underpinning this transformation is a masterpiece of organometallic chemistry, initiated by the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate. This step generates a reactive benzyl-palladium intermediate, which is subsequently trapped by carbon monoxide released in situ from the thermal decomposition of the phenol ester surrogate. The insertion of the first CO molecule forms an acyl-palladium species, setting the stage for the crucial cyclization event. The nucleophilic attack of the propargyl amine onto this acyl intermediate, followed by migratory insertion of the alkyne moiety, constructs the five-membered nitrogen-containing ring. A second carbon monoxide insertion then occurs, expanding the coordination sphere before the final reductive elimination releases the desired 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active palladium catalyst. This intricate dance of ligands and substrates ensures high turnover numbers and minimizes the formation of side products, which is essential for maintaining high purity specifications in downstream processing.
From an impurity control perspective, the use of the solid CO surrogate provides a steady, controlled release of carbon monoxide, preventing the local concentration spikes that often lead to polymerization or over-carbonylation side reactions in gas-phase methods. The choice of the DPPF ligand (1,1'-bis(diphenylphosphino)ferrocene) is particularly strategic; its large bite angle stabilizes the palladium center throughout the catalytic cycle, preventing catalyst deactivation via aggregation into palladium black. This stability is paramount for achieving the reported high yields across diverse substrates, as seen in the successful synthesis of derivatives bearing electron-withdrawing trifluoromethyl groups and electron-donating methoxy groups alike. 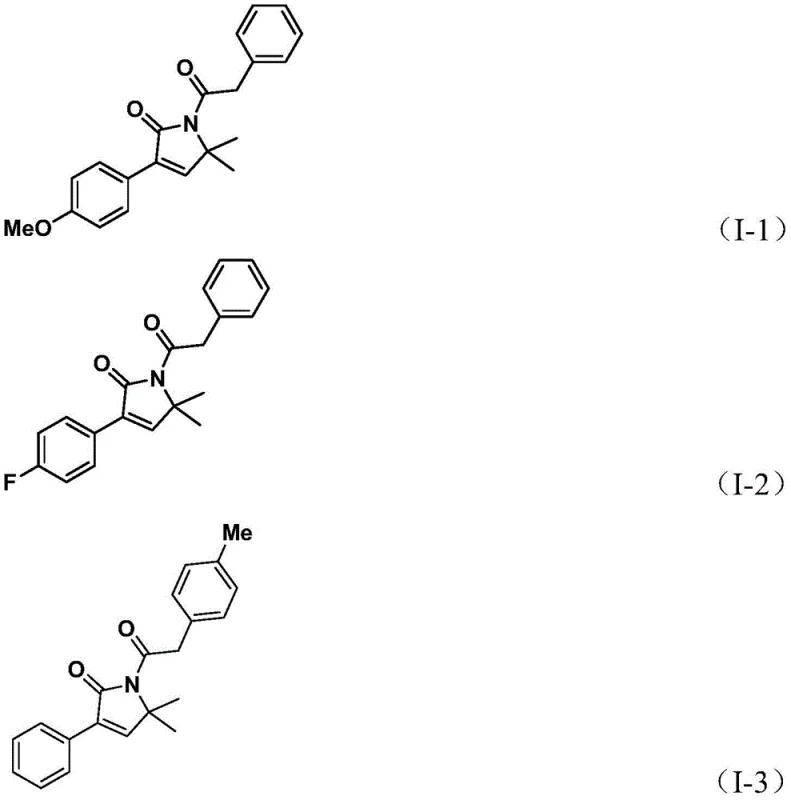 . For quality assurance teams, this mechanistic robustness translates to a cleaner crude reaction profile, reducing the burden on purification units and ensuring consistent batch-to-batch reproducibility essential for regulatory compliance.
. For quality assurance teams, this mechanistic robustness translates to a cleaner crude reaction profile, reducing the burden on purification units and ensuring consistent batch-to-batch reproducibility essential for regulatory compliance.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-Ketone Efficiently
The practical execution of this synthesis is designed for ease of operation, requiring standard laboratory equipment and commercially available reagents. The procedure involves charging a reaction vessel with the palladium catalyst, ligand, base, and the solid CO source in acetonitrile, followed by the addition of the amine and halide substrates. The detailed standardized synthesis steps see the guide below.
- Charge a reaction vessel with palladium acetate catalyst, DPPF ligand, triethylamine base, and the solid carbon monoxide surrogate (1,3,5-tricarboxylic acid phenol ester) in acetonitrile solvent.
- Add the propargyl amine substrate and benzyl chloride derivative to the mixture under inert atmosphere conditions.
- Heat the reaction mixture to 110°C and stir for 24 hours to ensure complete conversion, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The shift from gaseous carbon monoxide to a solid surrogate drastically simplifies logistics, removing the need for specialized gas handling infrastructure and reducing the regulatory burden associated with toxic gas storage. This operational simplification directly correlates to reduced overhead costs and enhanced facility safety, making the process viable for a broader range of manufacturing sites. Furthermore, the high reaction efficiency and short reaction times (24 hours) mean that reactor occupancy is minimized, allowing for higher throughput and better asset utilization without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive, commodity-grade starting materials such as benzyl chlorides and propargyl amines, which are widely available from global chemical suppliers. By eliminating the need for high-pressure reactors and specialized gas delivery systems, the capital expenditure (CAPEX) required for implementation is significantly lowered. Additionally, the high yields reported (up to 92%) imply less raw material waste and lower solvent consumption per kilogram of product, driving down the variable costs of production. The simplified work-up procedure, involving basic filtration and chromatography, further reduces labor and utility costs associated with complex isolation protocols.
- Enhanced Supply Chain Reliability: Dependence on specialized reagents can be a single point of failure in a supply chain; however, this method relies on robust, shelf-stable solids and liquids that are easily sourced from multiple vendors. The use of a solid CO surrogate ensures that the critical carbonylating agent is always available on-site without the risk of supply interruptions common with industrial gases. This resilience is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream API manufacturers. The broad substrate scope also allows for flexibility in sourcing different substituted benzyl chlorides, providing procurement teams with the leverage to negotiate better pricing based on market availability.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this protocol is inherently designed for scalability. The absence of high-pressure gas eliminates the risk of catastrophic leaks, and the use of acetonitrile, a common solvent with established recovery protocols, facilitates waste management. The high atom economy of the bis-carbonylation reaction means less chemical waste is generated per unit of product, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. This alignment with sustainability goals is increasingly important for multinational corporations aiming to meet their corporate social responsibility targets while expanding production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: What is the primary advantage of using 1,3,5-tricarboxylic acid phenol ester in this synthesis?
A: It serves as a safe, solid carbon monoxide surrogate, eliminating the need for handling hazardous high-pressure CO gas cylinders while maintaining high reaction efficiency.
Q: Does this method tolerate diverse functional groups on the benzyl chloride substrate?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as fluoro, chloro, bromo, and trifluoromethyl.
Q: What represents the typical yield range for this palladium-catalyzed transformation?
A: The patented process achieves high isolated yields, typically ranging from 70% to 92% across various substituted derivatives, indicating robust reaction performance suitable for industrial scaling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and commercialization. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless. We are committed to delivering high-purity 1,5-dihydro-2H-pyrrole-2-ketone intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking to secure their supply chains for next-generation therapeutics.
We invite you to explore how this innovative palladium-catalyzed technology can enhance your project's efficiency and cost-effectiveness. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals.
