Advanced Synthesis of Chlorantraniliprole Key Intermediate for Scalable Agrochemical Production
Introduction to Patent CN116332906A
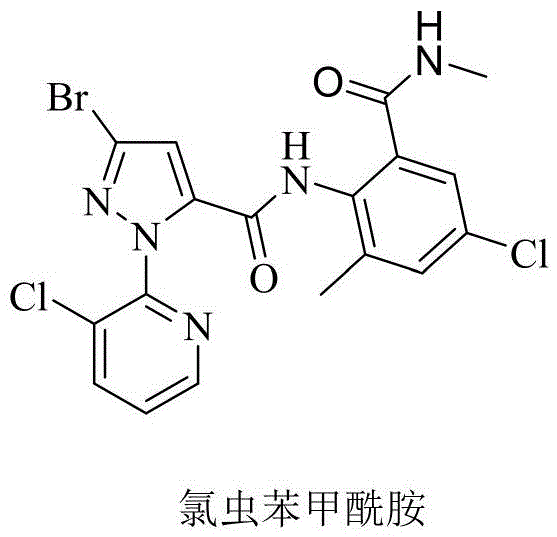
The agricultural chemical sector is constantly evolving towards more sustainable and efficient manufacturing processes, a trend exemplified by the technological breakthroughs detailed in patent CN116332906A. This patent discloses a novel method for synthesizing a critical intermediate for chlorantraniliprole, specifically 2-(3-bromo-1-(3-chloro-2-pyridyl)-1H-pyrazole-5-carboxamido)-5-chloro-3-methylbenzoic acid, alongside the final insecticide itself. Unlike traditional pathways that rely on complex cyclization steps, this innovation streamlines the synthesis by avoiding the formation of benzoxazinone ring intermediates. The methodology emphasizes mild reaction conditions, operational simplicity, and the use of cost-effective reagents, addressing key pain points in current agrochemical production. For R&D directors and supply chain managers, this represents a significant opportunity to optimize purity profiles while simultaneously reducing the environmental footprint associated with sulfur-containing waste streams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of chlorantraniliprole has relied heavily on pathways described in patents such as CN1678192A and WO2006062978, which necessitate the formation of benzoxazinone intermediates. These conventional routes typically involve reacting pyrazole carboxylic acids with anthranilic acid derivatives under the influence of tertiary amines and methanesulfonyl chloride to create a cyclic oxazine structure. This approach presents several significant drawbacks for large-scale manufacturing, primarily due to the requirement for excessive amounts of methanesulfonyl chloride and expensive organic solvents like tetrahydrofuran and acetonitrile. Furthermore, the generation of highly polluted sulfur-containing organic waste liquids poses severe environmental compliance challenges and increases disposal costs. The operational complexity is further compounded by cumbersome feeding and post-treatment procedures, often resulting in lower molar yields of the critical intermediates, which negatively impacts overall process economics.
The Novel Approach
In stark contrast, the method disclosed in CN116332906A introduces a paradigm shift by bypassing the oxazine ring formation entirely, opting instead for a direct acylation strategy that is both chemically elegant and industrially robust. This new route utilizes readily available reagents to convert the pyrazole carboxylic acid directly into an acyl chloride, which then couples with the anthranilic acid derivative under mild basic conditions. By eliminating the need for the benzoxazinone cyclization step, the process significantly reduces the number of unit operations and simplifies the purification workflow. The reaction conditions are notably温和 (mild), typically proceeding at temperatures between 0°C and 40°C, which minimizes thermal degradation and side-product formation. This streamlined approach not only enhances the overall yield but also aligns with green chemistry principles by reducing solvent usage and hazardous waste generation, making it an ideal candidate for modern, sustainable agrochemical manufacturing.
Mechanistic Insights into Direct Acylation and Amide Coupling
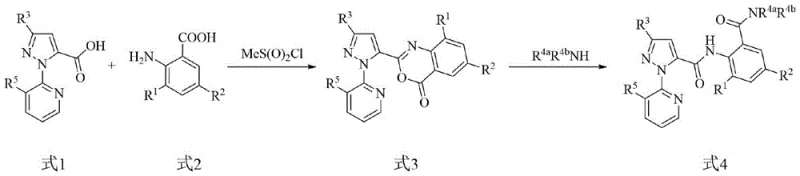
The core of this innovative synthesis lies in the precise control of the acylation and subsequent nucleophilic substitution mechanisms, which dictate the purity and yield of the final product. In the initial step, the carboxylic acid group of the pyrazole precursor is activated using acylating agents such as thionyl chloride or triphosgene, transforming it into a highly reactive acyl chloride species. This activation is crucial as it lowers the energy barrier for the subsequent nucleophilic attack by the amino group of the anthranilic acid derivative. The reaction is carefully managed in the presence of a base, such as triethylamine or sodium hydroxide, which serves to neutralize the generated acid and drive the equilibrium towards product formation. The use of phase transfer catalysts in aqueous systems further enhances the efficiency of this coupling by facilitating the interaction between organic and aqueous phases, ensuring complete conversion of the starting materials.
Impurity control is meticulously addressed through the optimization of stoichiometric ratios and temperature profiles throughout the synthetic sequence. For instance, maintaining the coupling reaction temperature between 15°C and 25°C is critical to preventing the hydrolysis of the sensitive acyl chloride intermediate while ensuring sufficient kinetic energy for the amide bond formation. Additionally, the selection of solvents plays a pivotal role; solvents like dichloromethane or toluene provide an optimal medium that balances solubility with reaction rate, allowing for easy separation of the product via simple washing and evaporation techniques. The avoidance of harsh cyclization conditions inherently reduces the formation of polymeric byproducts and structural isomers, resulting in a cleaner crude product that requires minimal downstream purification. This mechanistic clarity provides R&D teams with a robust framework for scaling the process while maintaining stringent quality specifications.
How to Synthesize Chlorantraniliprole Efficiently
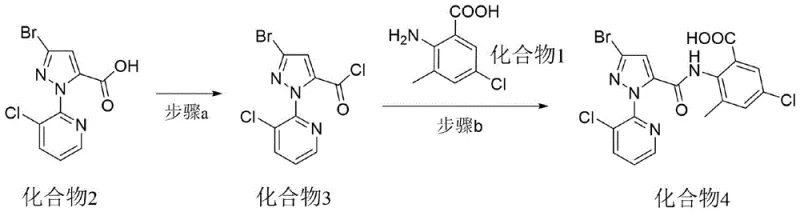
The synthesis protocol outlined in the patent offers a clear, step-by-step guide for producing the key intermediate and the final active ingredient with high efficiency. The process begins with the activation of the pyrazole acid, followed by a controlled coupling reaction to form the diamide backbone, and concludes with a final amidation step to install the methylamide functionality. Each stage is designed to maximize atom economy and minimize waste, leveraging standard chemical engineering unit operations that are familiar to plant operators. The detailed experimental examples provided in the patent demonstrate the reproducibility of the method across different scales and solvent systems, confirming its viability for commercial adoption. For a comprehensive understanding of the specific parameters, including exact molar ratios and workup procedures, please refer to the standardized synthesis guide below.
- Convert 3-bromo-1-(3-chloro-2-pyridyl)-1H-pyrazole-5-carboxylic acid to its corresponding acyl chloride using thionyl chloride or triphosgene at 50-100°C.
- React the resulting acyl chloride with 2-amino-5-chloro-3-methylbenzoic acid carboxylate salt in the presence of a base at 0-40°C to form the key intermediate Compound 4.
- Activate the carboxylic acid group of Compound 4 using an acylating agent to generate the reactive acyl chloride species (Compound 5).
- Perform nucleophilic substitution with methylamine or other active hydrogen compounds at 20-50°C to yield the final chlorantraniliprole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers substantial strategic advantages for procurement and supply chain stakeholders looking to optimize their agrochemical portfolios. The elimination of expensive and hazardous reagents like excessive methanesulfonyl chloride directly translates to a reduction in raw material costs and a simplification of the supply chain logistics. By relying on commodity chemicals such as thionyl chloride and common organic solvents, manufacturers can mitigate the risks associated with price volatility and supply disruptions of specialized reagents. Furthermore, the simplified post-treatment process, which often involves basic washing and solvent recovery rather than complex chromatographic separations, significantly reduces processing time and utility consumption. This operational efficiency allows for faster batch turnover times, enhancing the overall responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the reaction pathway and the use of low-cost, widely available reagents. By avoiding the formation of the benzoxazinone intermediate, the process eliminates the need for specific cyclization catalysts and the associated expensive solvents like THF, leading to significant savings in material costs. Additionally, the ability to recover and recycle excess acylating agents, such as thionyl chloride, further contributes to cost optimization by minimizing raw material waste. The high yield of the key intermediate ensures that less starting material is required per unit of final product, effectively lowering the cost of goods sold (COGS) and improving profit margins for large-scale producers.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the reliance on generic, non-proprietary raw materials that are readily accessible from multiple global suppliers. Unlike specialized intermediates that may have limited sources, the precursors used in this method, such as 2-amino-5-chloro-3-methylbenzoic acid, are established commodities with stable supply lines. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent choices, reduces the risk of batch failures due to minor process deviations. This reliability ensures consistent production schedules and helps maintain steady inventory levels, preventing stockouts and ensuring continuous availability for downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing reaction conditions that are easily transferable from laboratory glassware to industrial reactors without significant engineering hurdles. The mild temperatures and atmospheric pressure requirements reduce the need for specialized high-pressure or cryogenic equipment, lowering capital expenditure for capacity expansion. From an environmental standpoint, the reduction in sulfur-containing waste and the use of recyclable solvents align with increasingly stringent global environmental regulations. This compliance reduces the burden of waste treatment and disposal, mitigating regulatory risks and enhancing the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production lines or for sourcing high-quality intermediates from external partners. The insights provided here aim to clarify the operational benefits and technical nuances of the new route compared to legacy methods.
Q: How does this new synthesis method reduce environmental impact compared to traditional routes?
A: The novel method eliminates the formation of benzoxazinone intermediates, which traditionally require excessive methanesulfonyl chloride and generate significant sulfur-containing organic waste. By utilizing direct acylation with recyclable reagents like thionyl chloride, the process drastically reduces hazardous waste discharge and simplifies post-treatment protocols.
Q: What are the yield advantages of the Compound 4 intermediate synthesis?
A: Experimental data indicates that optimizing the molar ratios of amine, acyl chloride, and base allows for significantly higher yields compared to conventional methods. The mild reaction conditions (15-25°C) minimize side reactions, ensuring high purity and reducing the need for complex purification steps like column chromatography.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for industrial scalability. It utilizes cheap, easily obtainable reagents and avoids harsh conditions or expensive catalysts. The simple workup procedures, such as solvent recovery and filtration, facilitate seamless transition from laboratory scale to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chlorantraniliprole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthesis technologies like the one described in CN116332906A for the agrochemical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that monitor every stage of production, guaranteeing that our chlorantraniliprole and its intermediates meet the highest international standards. We understand the critical importance of consistency and reliability in the supply of active ingredients and intermediates for global crop protection markets.
We invite you to collaborate with us to leverage these technological advancements for your specific business needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can enhance your bottom line. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics. Together, we can drive efficiency and sustainability in the production of next-generation insecticides.
