Advanced Synthesis of Chlorantraniliprole Intermediates for Commercial Scale-Up
Advanced Synthesis of Chlorantraniliprole Intermediates for Commercial Scale-Up
The global demand for high-efficiency, low-toxicity insecticides has positioned Chlorantraniliprole as a cornerstone molecule in modern crop protection strategies. As the industry seeks more sustainable and cost-effective manufacturing routes, recent intellectual property developments offer significant breakthroughs in process chemistry. Specifically, patent CN111423431B discloses a preparation method that fundamentally restructures the synthesis of the key benzoxazinone intermediate and the final active ingredient. This technical insight report analyzes the proprietary methodology to provide R&D directors, procurement managers, and supply chain heads with a clear understanding of its commercial viability. By leveraging a novel mixing sequence and eliminating energy-intensive drying steps, this route addresses critical bottlenecks in the production of high-purity agrochemical intermediates. The following analysis details how this technology can be integrated into existing supply chains to enhance reliability and reduce operational expenditures without compromising product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Chlorantraniliprole intermediates has been plagued by complex operational procedures that hinder industrial scalability and cost efficiency. Conventional methods, such as those disclosed in earlier DuPont patents like WO2003/024222, typically involve dissolving methanesulfonyl chloride in acetonitrile and cooling it to low temperatures before adding it to a pre-cooled slurry of reactants. This process requires the sequential addition of multiple acetonitrile solutions and distinct portions of organic bases, leading to a fragmented and labor-intensive workflow. Furthermore, the post-reaction processing in these traditional routes necessitates multi-step leaching, extensive drying, and complex purification protocols to isolate the intermediate. These additional unit operations not only extend the production cycle time significantly but also introduce opportunities for product degradation and yield loss. The molar yield in such conventional processes is often reported to be as low as 54 percent based on the limiting reagent, which represents a substantial loss of valuable raw materials and increases the overall cost of goods sold for the final active ingredient.
The Novel Approach
In stark contrast to the fragmented conventional workflows, the novel approach described in patent CN111423431B introduces a streamlined mixing strategy that simplifies the reaction engineering and enhances overall throughput. The core innovation lies in the pre-mixing of 2-amino-5-chloro-3-methylbenzoic acid and 3-bromo-1-(3-chloro-2-pyridyl)-1H-pyrazole-5-carboxylic acid in the presence of an organic base and solvent to form a unified material prior to activation. This pre-formed material is then reacted with methanesulfonyl chloride under controlled low-temperature conditions to generate the benzoxazinone intermediate directly. This modification eliminates the need for complex sequential additions and allows for a more homogeneous reaction environment, which is critical for controlling exotherms and impurity formation. The resulting intermediate precipitates as a filter cake that requires no further treatment, drying, or purification before being charged into the subsequent aminolysis step. This telescoping of steps drastically reduces the number of unit operations, minimizes solvent handling, and significantly improves the total yield and product quality compared to legacy methods.
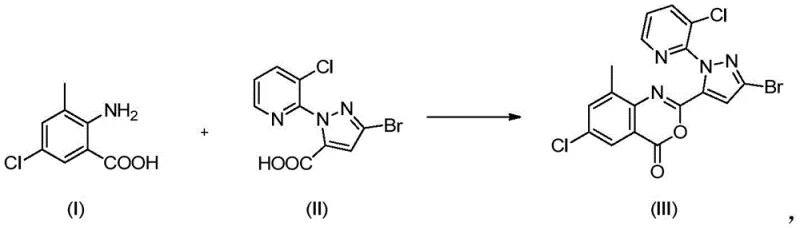
Mechanistic Insights into Benzoxazinone Cyclization
The chemical transformation at the heart of this process involves the formation of a 2-[3-bromo-1-(3-chloro-2-pyridyl)-1H-pyrazol-5-yl]-6-chloro-8-methyl-4H-3,1-benzoxazine-4-one intermediate, commonly referred to as BCPP. The mechanism relies on the activation of the carboxylic acid moiety by methanesulfonyl chloride to form a mixed anhydride or acyl sulfonate species in situ, which is then susceptible to intramolecular nucleophilic attack by the ortho-amino group. The selection of 3-methylpyridine as the base plays a dual role in this mechanism: it acts as a proton scavenger to drive the equilibrium forward and enhances the solubility of the starting materials in acetonitrile. Maintaining the reaction temperature between 0°C and 5°C is critical during the addition of the activating agent to suppress side reactions such as over-acylation or decomposition of the sensitive pyrazole ring. The precise control of stoichiometry, with a molar ratio of base to acid ranging from 5 to 6, ensures complete conversion while minimizing the formation of basic impurities that could complicate downstream processing. This mechanistic understanding allows for robust process control, ensuring that the intermediate is generated with high chemical purity suitable for direct conversion to the final insecticide.
Impurity control is further enhanced by the unique post-treatment protocol which leverages the physical properties of the reaction mixture. Unlike traditional methods that require drying the intermediate, this process utilizes the wet filter cake directly, thereby avoiding thermal stress that could generate degradation products. The filtrate obtained from the isolation of the intermediate contains a significant amount of acetonitrile and dissolved byproducts, which can be recycled as the solvent for the next batch. This mother liquor recycling strategy not only reduces the consumption of fresh solvent but also helps in maintaining a consistent impurity profile across batches by establishing a steady state of minor components. The subsequent reaction with aqueous methylamine proceeds efficiently on the wet cake, where the ring-opening of the benzoxazinone and the formation of the diamide structure occur under mild conditions. The final desalting refining step using a mixed solvent of acetonitrile and water ensures that inorganic salts and residual organic impurities are effectively removed, yielding a refined product with purity levels exceeding 97 percent.
How to Synthesize Chlorantraniliprole Efficiently
The implementation of this synthesis route requires careful attention to the mixing order and temperature profiles to maximize the benefits of the patented methodology. The process begins with the dissolution of the two key acid components in acetonitrile, followed by the addition of the organic base to form a homogeneous solution or slurry. Once the system is cooled to the optimal range of 0°C to 5°C, methanesulfonyl chloride is added slowly to control the exotherm and ensure selective activation. The reaction is then allowed to warm to room temperature to complete the cyclization, after which the solid intermediate is isolated by filtration. The detailed standardized synthesis steps, including specific charging rates, stirring speeds, and safety precautions for handling methanesulfonyl chloride, are critical for successful technology transfer and scale-up.
- Mix 2-amino-5-chloro-3-methylbenzoic acid and 3-bromo-1-(3-chloro-2-pyridyl)-1H-pyrazole-5-carboxylic acid with organic base in acetonitrile.
- Add methanesulfonyl chloride at 0-5°C to form the benzoxazinone intermediate filter cake.
- React the wet filter cake directly with aqueous methylamine to obtain crude Chlorantraniliprole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this novel synthesis route offers substantial advantages that directly address the pain points of procurement managers and supply chain directors in the agrochemical sector. The elimination of the drying step for the intermediate represents a significant reduction in energy consumption and equipment occupancy time, allowing for faster batch turnover and increased plant capacity. By enabling the direct use of the wet filter cake, the process removes a major bottleneck that often limits throughput in multi-purpose chemical manufacturing facilities. Furthermore, the ability to recycle the filtrate as a solvent for subsequent batches drastically reduces the volume of fresh acetonitrile required, leading to lower raw material costs and reduced waste disposal expenses. These operational efficiencies translate into a more competitive cost structure for the final active ingredient, providing buyers with a more stable and cost-effective supply source in a volatile market environment.
- Cost Reduction in Manufacturing: The streamlined process significantly lowers manufacturing costs by eliminating energy-intensive drying operations and reducing solvent consumption through mother liquor recycling. The removal of complex leaching and purification steps for the intermediate reduces labor costs and minimizes the loss of valuable materials during transfer operations. Additionally, the high yield achieved by this method ensures better utilization of expensive starting materials, further driving down the variable cost per kilogram of the final product. These cumulative savings allow for a more aggressive pricing strategy while maintaining healthy margins, making the supply chain more resilient to fluctuations in raw material prices.
- Enhanced Supply Chain Reliability: The simplicity of the operation enhances supply chain reliability by reducing the number of potential failure points in the manufacturing process. Fewer unit operations mean less equipment is required, lowering the risk of mechanical breakdowns and maintenance-related downtime. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing, ensures consistent batch-to-batch quality, reducing the risk of out-of-specification products that could disrupt supply. This reliability is crucial for long-term supply agreements where consistent delivery schedules are paramount for the planning of downstream formulation and distribution activities.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, with reaction conditions that are easily manageable in large-scale reactors without requiring exotic equipment or extreme pressures. The reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations, reducing the burden on waste treatment facilities and lowering compliance costs. The use of acetonitrile, a common industrial solvent with established recovery infrastructure, further facilitates scalable implementation. This environmental efficiency not only mitigates regulatory risk but also enhances the sustainability profile of the supply chain, which is a growing priority for global agrochemical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions provided in the patent literature to ensure accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of adopting this route for commercial production or sourcing intermediates manufactured via this method.
Q: How does this method improve yield compared to conventional DuPont processes?
A: The novel approach avoids complex multi-step leaching and drying, achieving yields around 90% compared to the conventional 54% by utilizing direct filtration and mother liquor recycling.
Q: Is the intermediate stable enough for direct use without drying?
A: Yes, the benzoxazinone intermediate (Formula III) is generated as a filter cake that can be directly charged into the next aminolysis reaction without drying, significantly reducing processing time.
Q: What solvent system is recommended for industrial scalability?
A: Acetonitrile is the preferred solvent, with the capability to recycle the filtrate from the intermediate step, drastically reducing overall solvent consumption and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chlorantraniliprole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, possessing the technical expertise to translate complex patent methodologies into robust commercial processes. Our R&D team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Chlorantraniliprole intermediate meets the exacting standards required by global agrochemical leaders. Our commitment to quality and process safety makes us an ideal partner for companies looking to secure a stable supply of high-performance crop protection intermediates.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain. By partnering with us, you gain access to a reliable source of high-purity agrochemical intermediates that are produced using state-of-the-art, cost-efficient methodologies. Contact us today to discuss how we can support your product development and commercialization goals with our advanced synthesis solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
